Key Points
- •
Focused cardiac ultrasound can improve medical decision making or alter time to initiation of appropriate treatment, especially in patients who are unstable, have heart failure, or have indeterminate volume status.
- •
After limited training, providers can accurately detect left ventricular enlargement and decreased systolic function, left atrial enlargement, right ventricular enlargement, pericardial effusion, and inferior vena cava collapsibility.
- •
Training in focused cardiac ultrasound should include three core components: didactic education, hands-on imaging practice, and image interpretation experience.
Background
Ultrasound is more portable and less expensive than other modalities (CT, MRI, nuclear perfusion) used to image the heart. Unlike modalities that expose patients to radiation, there are no known adverse effects of ultrasound used at diagnostic imaging intensities, allowing safe, serial evaluation of patients across the entire spectrum of patient care from in utero to the frail elderly patient. Ultrasound images are evaluated in real time at the bedside with no need for advanced image processing or image display on a dedicated workstation.
Miniaturized ultrasound platforms have significantly fewer “knobs and buttons,” which makes them easier to operate. The simplified operation and substantially smaller size and cost of these devices have appealed to physicians from diverse specialties, leading to the concept of focused cardiac ultrasound (FCU) exam.
Indications and Applications
Many physicians could potentially use FCU to augment bedside cardiac evaluation. Findings of an FCU examination should be interpreted in conjunction with other bedside measures, such as physical exam and laboratory data, to formulate a diagnostic impression and guide appropriate triage and management. While an FCU exam may become routine in all patients in the future, FCU exams are currently used in specific clinical settings to answer a directed clinical question to expedite patient care.
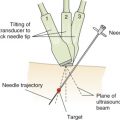
The specific cardiac views and images to acquire differ depending on clinical need. Device capabilities, image acquisition expertise, and interpretive skills of the provider are the major considerations when defining which cardiac abnormalities can be reliably detected. The primary cardiac views are obtained from the parasternal and subcostal windows, which is consistent with the published ultrasound exams, such as the cardiovascular limited ultrasound exam (CLUE) and extended focused assessment with sonography in trauma (EFAST). Imaging in the subcostal and parasternal windows requires minimal changes in patient position and is relatively easier to perform compared to other windows.
It is not possible to enumerate all of the specific clinical scenarios in which an FCU exam might prove useful. FCU may be used anytime it has potential to improve the provider’s assessment of a patient. FCU allows detection of findings that are occult to bedside physical exam (pericardial effusion) or simply difficult to detect with traditional bedside tools (left ventricle [LV]) dysfunction).
Ideally, FCU would be used in settings where it has been demonstrated to improve medical decision making or alter time to initiation of appropriate treatment. However, most studies evaluating FCU have focused on diagnostic accuracy to detect specific abnormalities compared to a gold standard. Future studies will identify the settings in which FCU is most beneficial and focus on demonstrating improved health outcomes.
Unstable Patients
FCU has demonstrated benefit in patients with hemodynamic instability, chest trauma, and pericardial tamponade. In these settings, FCU can be performed rapidly at the bedside and clearly adds to the bedside evaluation of patients. Assessment of critically ill patients after a sudden change in clinical status is an invaluable application of FCU, and in these critically ill patients, FCU has been shown to improve bedside assessment of volume status and LV systolic function. In hemodynamically unstable patients, FCU can guide management with regard to volume repletion and use of pressors, diuretics, and vasodilators. FCU may also be used to identify findings suggestive of pulmonary embolism (right ventricular enlargement). Cardiac arrest patients may benefit from FCU to evaluate for cardiac standstill when deciding to cease or continue resuscitative efforts. FCU can guide postarrest management by allowing rapid assessment of LV systolic function and volume status. In patients with chest trauma or findings suggestive of pericardial tamponade, providers can learn to identify the presence or absence of pericardial effusion after brief training.
Heart Failure
In patients with acute decompensated heart failure (ADHF), physicians who have performed 20–25 practice FCU exams can readily distinguish those with normal vs. reduced LV systolic function. It is clear that FCU is superior to physical examination, ECG, chest radiograph, and blood chemistries for detection of LV systolic dysfunction in patients with ADHF. While patients with heart failure should have a comprehensive echocardiogram, bedside FCU provides immediate knowledge of LV systolic function, which allows both initiation of appropriate therapies and avoidance of contraindicated therapies. Early data suggest that length of stay may be reduced when FCU is routinely integrated into care of hospitalized patients with ADHF.
Volume Status
Assessment of volume status is indicated in patients with acute dyspnea and hypotension. Identification of volume depletion in a hypotensive patient or volume excess in a dyspneic patient can facilitate diagnosis and treatment. Traditionally, providers have attempted to estimate right atrial pressure by examining the jugular venous pulse (JVP) as a surrogate of volume status. It is clear that body habitus limits evaluation of JVP, and the JVP is often poorly assessed, even when clearly visible. FCU assessment of the inferior vena cava (IVC) is both more feasible and accurate than physical exam to detect elevated central venous pressure. Knowing when to stop, intensify, or continue inpatient intravenous diuretic therapy is a common clinical question. IVC size and collapsibility correlates with central venous pressure and can assist in management. A pilot study of FCU in ADHF patients showed that patients discharged with a plethoric IVC, suggesting ongoing volume overload, were more likely to be readmitted within 30 days. Critically ill patients frequently have variations in volume status and LV or right ventricle (RV) function, and serial evaluation using FCU is readily applicable to guide management.
Screening
Because FCU can identify many cardiac abnormalities with higher accuracy than physical examination, it can potentially be used to screen for LV systolic dysfunction (LVSD). LVSD is ideal for screening because it is relatively prevalent (2-4%), readily detectable with FCU, and has effective therapy even in the preclinical stage, but is often asymptomatic and missed by physical examination. Several studies have demonstrated feasibility of FCU for identifying LV dysfunction in a variety of patient populations. A cost analysis study suggested that, rather than screening all patients directly with FCU, using FCU in patients with an abnormal BNP or ECG was the most cost-effective strategy for identifying asymptomatic LV dysfunction. In addition to LVSD, increased LV wall thickness and LA dimensions, which are associated with increased cardiovascular morbidity and mortality, can readily be detected with FCU, and their identification may have prognostic implications.
Literature Review
The ability of FCU to detect LV systolic function has been well studied. Physicians with limited ultrasound training can detect LV systolic dysfunction with sensitivities of 73–100% and specificities of 64–96%. In the assessment of LV systolic function, apical abnormalities may not be appreciated if imaging is performed only from the parasternal window. Several studies have demonstrated that parasternal imaging is adequate for the subjective assessment of LV function. For cardiac imaging, the parasternal and subcostal views are typically easier to master. Landmarks for these windows are more reliable, and these views consistently provide more interpretable images than apical views. Parasternal views are less dependent on patient positioning and less subject to interference from patient body habitus. The parasternal window is preferred for the assessment of LV systolic function by less experienced users. Nonexpert imagers obtain adequate images from the parasternal view nearly twice as often as from the apical views with an FCU device.
Other abnormalities that can be detected by FCU include LV enlargement, LV hypertrophy, LA enlargement, RV enlargement, pericardial effusion, and IVC size. FCU devices have shown reasonable accuracy when used by providers with limited training to quantify LVH, LA size, and LV dimensions.
Although the ability to detect abnormalities at the bedside with FCU is lower than a comprehensive echocardiogram, FCU allows detection of cardiac pathology more accurately than traditional bedside physical examination assessment. When used by physicians without formal echocardiographic training, FCU is superior to physical examination for the detection of cardiac abnormalities including LV enlargement, LV systolic dysfunction, LA enlargement, LV hypertrophy, pericardial effusion, and RA pressure elevation.
Limitations
Smaller ultrasound devices, while clinically useful, should not be expected to provide the same image quality as a full platform ultrasound machine (see Chapter 44, Equipment, Image Archiving, and Billing ). Instruments used for FCU, especially pocket and hand-carried devices, have been miniaturized to improve portability and lower cost. This compromise may result in loss of features, including spectral and tissue Doppler, which limits FCU for the assessment of valvular, ventricular, and pericardial hemodynamics. In an effort to simplify operation, some devices have image controls for depth and gain adjustment only, omitting image enhancement capabilities found on higher-end devices. In addition, display of images on a smaller screen with lower resolution limits interpretation.
Devices used for FCU imaging have a limited degree of measurement capability. In addition, as rapid bedside evaluation is one of the major strengths of FCU, pausing to take a measurement or make a calculation is typically not done. An FCU exam is geared to acquire and interpret images rapidly at the bedside to detect significant abnormalities, such as moderate or severe deviations from normal. Quantitative characterization of pathologies into severities of abnormality should not be expected with FCU.
FCU exams do not acquire images from all possible windows and views, which may limit detection of abnormalities. A well-designed FCU examination includes views that are readily obtained to detect cardiac pathologies that are pertinent to the clinical setting and within the provider’s level of expertise. Providers must recognize their individual skill level, in addition to limitations of the available ultrasound equipment. Common pitfalls, such as off-axis imaging and foreshortening, can lead to incorrect conclusions.
Complex or unusual cardiac disorders should not be expected to be diagnosed with FCU. In addition, some pathologies are subtle and difficult to recognize (LV wall motion, vegetations, hypertrophic cardiomyopathy, RV hypertrophy). Other abnormalities require assimilation of data from multiple views to correctly define (RV systolic function and size, cardiac masses, LV thrombus, aneurysms). Certain conditions, such as congenital heart disease, require extensive training in cardiovascular disease to accurately evaluate with cardiac ultrasound.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree