5 Nuclear Medicine
Introduction
Nuclear medicine imaging comprises planar projection and tomographic image acquisition techniques to capture the distribution of radiolabeled substances. Acquisitions includes single time point imaging, gated and time-series imaging to map radiotracer biodistributions for evaluation of normal/abnormal function. Data collection is a photon starved process, that is, the total number of photons collected is substantially lower than any other X-ray-based modality leading to images of high noise. Moreover, attenuation and scatter within the patient and detector coupled with an imperfect photon detection process (detector energy resolution, collimation, and event processing) degrade the image contrast and spatial resolution. Evaluation of these factors, and improvements when possible, is key to the pursuit of improved image quality. Elements of good image quality in nuclear medicine include absence of image distortion and a noise distribution that is consistent with the expected radiotracer uptaket
Common Image Quality Problems
Patient motion: degrades spatial resolution due to object motion blurring.
Improper attenuation correction: leads to hypo- or hyperintense regions due to misalignment between the transmission and emission data in the reconstruction process.
High-Z materials: oral/intravenous contrast and implanted metallic devices are opaque to X-rays used in positron emission tomography/computed tomography (PET/CT) systems to collect transmission data leading to artifacts that propagate to the emission image.
Truncation: tissue lying outside the camera field of view (FOV) that is not incorporated properly into the reconstruction process.
5.1 Case 1: Degraded Resolution of a Whole-Body Planar 99mTc Methylene Diphosphonate Image
5.1.1 Background
Patient suspected of having neoplastic disease.
Delayed phase skeletal images obtained approximately 2 hours post injection.
Anterior/posterior planar contiguous bone imaging protocol with low-energy high-resolution (LEHR) collimators.
Counts from the first images starting at the head totaled approximately 2.3 million.
5.1.2 Findings
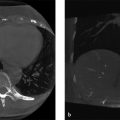
Counts are sufficient in each view (Fig. 5‑1).

Fig. 5.1 Anterior (left) and posterior (right) whole-body 99mTc methylene diphosphonate images.
A large visual difference in image resolution is observed between the anterior and posterior planar images.
The anterior image is of unacceptable diagnostic quality.
5.1.3 Discussion
Review of the acquisition setup showed that the anterior camera head was positioned further from the patient than the posterior camera head. The consequence was a sharp degradation in image quality observed in the anterior camera due to the strong dependence of resolution on the collimator distance from the source. The loss of resolution with collimator distance is a linear relationship with distance from the camera as shown in Fig. 5‑2.

Although the distance has a large effect on resolution, count rate is not strongly dependent on the source distance from the collimator. The counts collected at a single point decrease by 1/(source-to-collimator distance)2; however, the number of collimator holes that permit passage of photons is proportional to (source-to-collimator distance)2. 11 , 12 – 13 Therefore, the total number of counts, represented by the area under the curves in Fig. 5‑2 is essentially the same at all source distances with a parallel-hole collimator. This relationship applies to point, line, and uniformly distributed sources.
5.1.4 Resolution
Proper placement of the gamma camera heads relative to the patient is the responsibility of the camera operator. There are no software corrections to sharpen a blurred image, therefore the operator should rescan the patient with the gamma camera heads placed at the proper distances from the patient. A satisfactory bone scan is shown in Fig. 5‑3 with the anterior camera closer to the patient.

5.2 Case 2: Effect of Positron Range on Image Quality and Resolution
5.2.1 Background
A rubidium-82 (82Rb) chloride PET study is performed to assess myocardial perfusion.
Following the resting exam, an fluorodeoxyglucose ([18F]FDG) viability study is performed to assess myocardial metabolism.
Both studies are performed in the resting state on the same scanner with the same reconstruction parameters.
5.2.2 Findings
The resting [82Rb]Cl and [18F]FDG viability studies are of excellent quality exhibited by the high contrast (> 2:1) between the myocardium and blood pool.
There is a marked difference in resolution between the [18F]FDG and [82Rb]Cl images, where the [82Rb]Cl image appears to be blurred relative to the [18F]FDG study.
5.2.3 Discussion
A resting/viability protocol is a good example of how positron range affects image quality. Because both images were collected on the same instrument using the same reconstruction parameters, the difference in image quality can be attributed entirely to the position range effect (Fig. 5‑4). Note that some differences in image contrast exist due to the biochemical processes that govern the uptake and distribution of [82Rb]Cl and [18F]FDG.

Fig. 5‑5 details three main factors that affect image resolution in PET imaging: detector size, noncollinearity of the annihilation photons and position range.4,5 Briefly, resolution is approximately proportional to half the detector size for annihilation events originating in the center of the FOV and approximately equal to the detector size at the periphery. Noncollinearity in the annihilation photons occurs because of a small amount of residual momentum remaining at the time of positron annihilation. Since the PET system assumes collinear photons, the line along which the system assigned the coincidence event is in error compared to the true annihilation location. This error in positioning increases with FOV diameter. Lastly, the energy of positrons from beta decay have a continuous spectrum from zero to a maximum energy. As a result of this energy distribution, the range of a position in tissue can be described by an exponential function. The higher the maximum energy, the larger the positron range and distance of the annihilation event from the decay origin. This disparity between location of the radiotracer and annihilation event contributes to resolution loss independently of the detector design. Rb-82 has maximum positron energy of 3,400 keV compared to F-18 at 635 keV. Therefore, all things the same, the resolution achieved with Rb-82 will be inferior to that of F-18-labeled compounds.

5.2.4 Resolution
The resolution difference between [82Rb]Cl and [18F]FDG in this example is normal and attributed to positron range. Newer reconstruction algorithms now model the positron physics (among other resolution degrading factors) in the iterative reconstruction process, which can compensate for some of this effect.
5.3 Case 3: Standardized Uptake Value in Positron Emission Tomography (Noise Bias)
5.3.1 Background
Three difference whole-body [18F]FDG PET reconstructions are presented.
Regions of interest (ROIs) are drawn in the liver, which is assumed to be uniform.
Standardized uptake value (SUV) mean of the liver is a semiquantitative indicator of scanner calibration and may be checked periodically to ensure correct SUV calculation.
The three images are reconstructed with the same iterative algorithm but with bed times of 30, 60, and 90 seconds.
5.3.2 Findings
Although the ROIs are drawn in the same location for each reconstruction, extracted information differs.
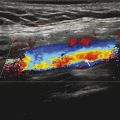
Fig. 5‑6 reports the SUV mean and max results.

Fig. 5.6 A whole-body [18F]FDG coronal slices reconstructed with imaging durations of 30, 60, and 90 seconds per bed position. The max and mean standardized update value are reported for a region of interest placed in the liver (red circles).
5.3.3 Discussion
The SUV is calculated by normalizing the activity concentration in the reconstructed image to the ratio of the administered activity and body weight.6,7

It has meaning similar to the pharmacological concept of distribution volume. That is, if the administered activity is uniformly distributed throughout the body, the SUV would be 1.0 g/mL everywhere. This concept is independent of the amount of FDG administered and is a method for removing variation between patients of [18F]FDG distribution that are related to administered amount and body weight. Assessment of the mean SUV in a large uniform area, such as the liver, can be a good marker for detecting potential imaging problems. In uniform normal liver, the mean SUV has been shown to vary with camera manufacturer but have an expected value ranging between 1.8 and 2.3 g/mL.
Clinically, the maximum SUV, not the mean, is used in the reporting and staging of cancer with [18F]FDG. SUVmax is the brightest (or highest) voxel in the ROI and has been shown to be a better predictor of outcomes. A number of factors affect the accuracy of SUV including presence of body fat, patient diet/fasting, reconstruction parameters, scan duration, partial volume effects, and others. In this case, we illustrate the change in SUV on scan duration by reducing the number of counts used in each reconstruction while maintaining all other variables constant. As the scan duration per bed is reduced, the noise in the reconstructed images increases. Noise is a high-frequency component in the image; therefore, SUVmax will always increase when noise increases. This can be thought of as a noise bias, where the mean value is less sensitive. Although the SUV mean is robust in the liver, it is less reproducible than SUV max in smaller features such as lesions.
5.3.4 Resolution
Image quality is largely subjective and based on the preference of the interpreting radiologist or nuclear medicine physician. It is critical that the patient preparation and PET camera used to collect data be kept the same for each scan in a patient’s cancer assessment. Therefore, any bias related to the instrument hardware or reconstruction is largely kept constant throughout a patient’s initial staging and clinical follow-up.
5.4 Case 4: The Impact of Attenuation Correction in PET
5.4.1 Background
An [18F]FDG PET/CT image is collected as part of cancer staging.
The overall image is unremarkable and considered of good technical quality.
5.4.2 Findings
A photopenic area is observed at the interface between the lung and liver in the attenuation-corrected [18F]FDG PET (Fig. 5‑7 a).

Fig. 5.7 (a) Attenuation corrected coronal [18F]FDG positron emission tomography (FDG PET) slices showing a photopenic area at the interface between the liver and lungs. (b) Nonattenuation-corrected coronal slice with no photopenic regions. (c) Corresponding coronal CT used for attenuation correction of (a). (d) Attenuation-corrected coronal FDG PET using an average CT protocol that better accounts for the liver position in the PET data.
The height of the liver in the PET scan does not match to that in the CT scan (Fig. 5‑7 a–c).
The nonattenuation-corrected (NAC) scan shows liver activity in areas that the attenuation-corrected scan does not, suggesting these photopenic areas are not physiological (Fig. 5‑7 d).
5.4.3 Discussion
The photopenic area observed in Fig. 5‑7 a is not present in the NAC images in Fig. 5‑4 b. Also, the height of the liver in the attenuation-corrected images and CT are the same suggesting that the attenuation correction was not accurate in that interface region. This attenuation-related artifact is common in whole-body [18F]FDG PET and is caused by a difference in temporal resolution between the acquisition of the PET and CT data.8 The result is that the PET represents a time-averaged position of all structures that move during the respiratory cycle while the CT captures a snapshot at a specific phase of the respiratory cycle. This leads to a mismatch in position of structures within the thoracic cavity, more easily observed where the motion extent is the greatest, near the lung and liver boundary.
Attenuation correction is the largest data correction step in the image reconstruction process. For a set of annihilation photons that originate at center of the body and travel to opposing detectors in the coronal plane of Fig. 5‑4, the correction factor can be as large as 18× for a total of 30 cm tissue. If the amount of tissue traversed by these photons were underrepresented by 40%, such as the presence of lung instead of liver tissues, then attenuation correction factor applied would be 5.5 or 3 times lower. An undercorrection in attenuation of this magnitude will alter image contrast leading to photopenic regions similar that described in Fig. 5‑7.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree