KEY POINTS
The most frequent craniofacial malformations are facial clefts. Different varieties exist. These conditions can be corrected surgically with good results. However, they are frequently associated with other malformations and syndromes that may have a major influence on the prognosis.
In expert hands, facial clefts can be accurately identified and categorized with sonography since early gestation. Three-dimensional (3D) ultrasound (US) may be helpful, and magnetic resonance imaging (MRI) can also be employed. However, the diagnosis is not simple, and indeed in standard sonographic examinations it is frequently missed.
In addition to facial clefts, other craniofacial anomalies can be identified sonographically. The list is long and includes ocular anomalies, such as microphthalmia, cataracts, micrognathia, and craniosynostosis. However, the diagnosis is generally difficult and is hampered by their progressive development.
Modern US equipment and 3D sonography in particular reveal many details of fetal craniofacial anatomy and may allow the diagnosis of even subtle dysmorphism. Evaluation of the fetal face is important in the assessment of fetuses with extracraniofacial anomalies because it may provide important clues to the diagnosis of syndromes.
Craniofacial anomalies are also discussed in Chapters 2 and 7.
Craniofacial anomalies include a wide spectrum of malformations. They may be clinically relevant as such, but they may also be associated with other congenital anomalies or be part of a syndrome. Evaluation of the face is indeed an important part of the clinical genetic examinations performed postnatally. Therefore, any time a fetal anomaly is identified during an antenatal US scan, the diagnostic workup should include a detailed examination of the fetal face. Apart from obvious malformations, a prenatal sonogram may also identify subtle dysmorphisms that may be crucial for a definitive diagnosis.
Prenatal sonographic diagnosis of craniofacial anomalies is possible in early gestation.1,2,3,4,5 The diagnostic accuracy of referral centers in the investigation of selected patients at increased risk is quite high,2,3,4,5,6 whereas the sensitivity of standard examinations of low-risk patients is low, in the range of 20% to -40%, with a general tendency to recognize facial malformations associated with other anomalies and to miss the isolated ones.7,8,9,10 However, until a few years ago, most national guidelines for the standard examination of fetal anatomy suggested only demonstration of the orbits and the eyes. More recently, the recommendations include visualization of the nose, lips, and chin, with an expected positive impact on the detection of craniofacial anomalies.
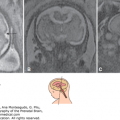
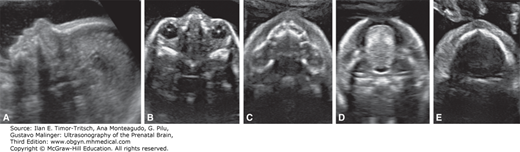
With two-dimensional (2D) US, a combination of planes must be used to assess facial anomalies.1,3,6 Midgestation is probably the most favorable time to evaluate the fetal face. However, many details of facial anatomy can be identified as early as 11 postmenstrual weeks. In the third trimester, the examination fails frequently because of intrauterine crowding and unfavorable fetal position. The median plane allows the visualization of the profile (Figure 16–1) with the forehead, nose, and jaw, which are readily appreciated in this view. Axial or coronal planes are used to assess the integrity of the eyes and lips. Nomograms for binocular distance, interocular distance, and ocular diameter are available (see Chapter 3). By moving the transducer caudally, the anterior lip and palate can be visualized. Further caudal shift of the probe will display the tongue inside the oral cavity and the mandible.
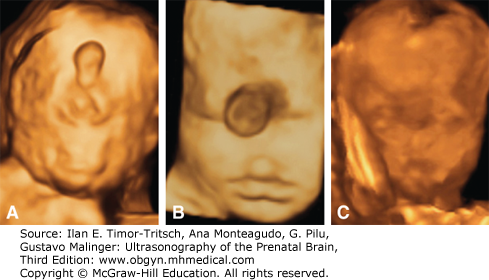
Figure 16–1.
Two-dimensional (2D) sonographic evaluation of the fetal face at midgestation. (A) Median view demonstrating the fetal profile. (B)–(E) Axial scans demonstrating the eyes, the maxilla, the tongue within the oral cavity, and the mandible. (Reproduced, with permission, from Visual Encyclopedia of Ultrasound in Obstetrics and Gynecology, www.isuog.org.)
The advantages of 3D US over conventional 2D US include the visualization of a panoramic view of the face, as well as a clear visualization of sutures and fontanelles (Figure 16–2). The limitations of the technique are the same as for 2D sonograms. If the fetal face is not accessible, or if there is no pocket of amniotic fluid separating the face from the surrounding structures, 3D will be of little help. However, in expert hands, a full and complete exam is possible in the majority of cases in the second to early third trimester. The relative value of 2D over 3D US has been debated.4,6,11 In expert hands, 2D US is effective in identifying and categorizing craniofacial malformations. The authors have not found any significant diagnostic advantage over 3D US.6 However, special 3D techniques may allow visualization of the posterior palate, which is seldom if ever possible with 2D US (Figures 16–3 and 16–4).11,12,13,14,15 It has also been suggested that 3D US has other potential benefits, such as offering the parents an understandable image of the anomaly, and allowing better communication with and counseling by the specialists involved in the management of the neonate. Most of the experience with 3D US has been derived from referral centers. The impact of this technique in routine anatomy scans has yet to be assessed. In general, 3D US is complex and probably beyond the scope of a basic routine evaluation of fetal anatomy in low-risk pregnancies. It is possible that real-time 3D US (also referred to as 4D), which allows a continuous, rapid, and accurate visualization of the external surface of the fetus in motion, could prove useful for assessment of facial anatomy.
Figure 16–2.
Three-dimensional (3D) sonographic examination of the fetal face at midgestation. (A), (B) Surface rendering from a lateral and frontal perspective. (C)–(E) Maximum rendering demonstrating the skull and the sutures and fontanels interposed among the different bones (1, coronal suture; 2, bregmatic fontanelle; 3, metopic or frontal suture; 4, sagittal suture). (Reproduced, with permission, from Pilu G. Atlas of Obstetric Ultrasound, The Global Library of Women’s Medicine, www.glowm.com.)
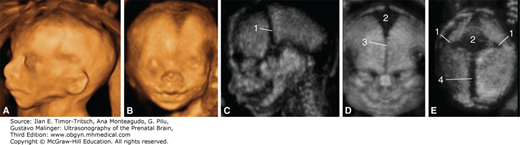
Figure 16–3.
3D sonographic evaluation of the posterior palate. (A) The palate is insonated with a 45° angle. (B) The volume that is obtained is rotated 45° counterclockwise to bring the palate in a vertical position. (C) The corresponding axial section effectively demonstrates the full length of the posterior palate; compare this image with the axial section that is obtained with a direct scan in Figure 16–1C. (Reproduced, with permission, from Pilu G, Segata M. A novel technique for visualization of the normal and cleft fetal secondary palate: angled insonation and three-dimensional ultrasound. Ultrasound Obstet Gynecol 2007;29[2]:166–169.)
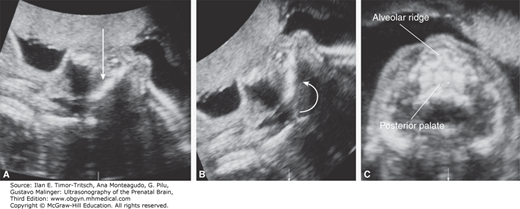
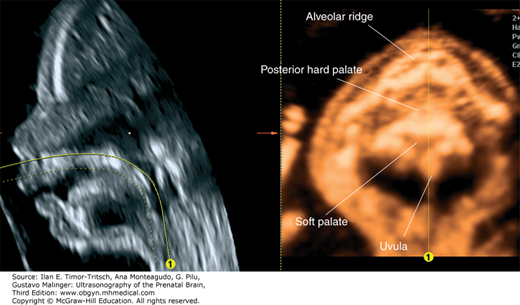
Figure 16–4.
3D sonography of the fetal palate. The mouth of the fetus is slightly open, and some fluid is found between the palate and the tongue. By using a curvilinear cut, it is possible to demonstrate both the hard and the soft palate, including the uvula. (Reproduced, with permission, from Pilu G. Atlas of Obstetric Ultrasound, The Global Library of Women’s Medicine, www.glowm.com.)
Facial clefts are the most frequent craniofacial anomalies and the second most common congenital malformation, accounting for 13% of all anomalies. The incidence is ~1.4 cases per 1000 live births.7,18 The anatomy and pathogenesis of these defects can be better understood in light of its embryological development. The fetal splanchnocranium derives from outgrowths of mesenchyma that surrounds the primitive oral cavity or stomodeum. These outgrowths (frontonasal prominence, maxillary prominence, and mandibular prominence) are separated by grooves that eventually undergo fusion and obliteration. The palate originates from the fusion of three palatine processes with the nasal septum, which divides the nasal cavities. The palate is commonly divided in three parts: the anterior or primary palate, the posterior or secondary palate, and the soft palate.
Most frequently, facial clefts derive from the persistence of the grooves between the frontonasal and maxillary prominences and involve the ideal line running between each nostril and the central part of the posterior palate. However, defects can occur in any part of the face. We will describe separately the typical cleft lip/palate, the cleft palate, and atypical facial clefts, as these categories differ in the etiology, clinical implications, and diagnostic approach.
The most frequent facial clefts are linear defects running between the ideal line connecting each nostril to the central portion of the palate. Roughly, in one-third of cases, there is an isolated defect of the lip (cleft lip). In the remaining two-thirds of cases, the opening extends variably into the palate (cleft lip/palate). The cleft may reach only the alveolar ridge, but in the majority of cases, it involves the secondary palate as well, reaching the floor of the nasal cavity or even the floor of the orbit. Cleft lip is bilateral in 20% of cases, whereas cleft lip/palate is bilateral in 25%.
In the vast majority of cases, cleft lip/palate has a multifactorial etiology. In some cases, however, it may be part of well-established genetic and nongenetic syndromes. The claimed risk associated with intake of diazepam and steroidal agents has not been confirmed in carefully controlled studies. Chromosomal abnormalities are rare in postnatal series but rather frequent in the prenatal ones.2,6,19 The discrepancy may be due to a high rate of intrauterine selection of aneuploid fetuses, as well as to the inclusion of an excess of atypical clefts.
Extrafacial anomalies are found in ~13% of infants with cleft lip/palate and up to 60% of fetuses with facial clefting.2,3,19,20 A long list of genetic and nongenetic syndromes is associated with facial clefts. The most frequent malformations are nervous, skeletal, and cardiovascular anomalies.
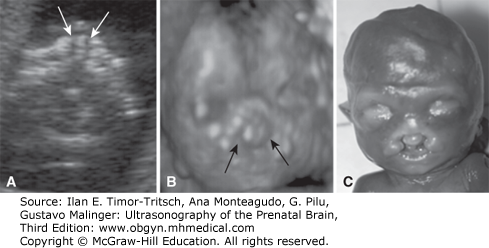
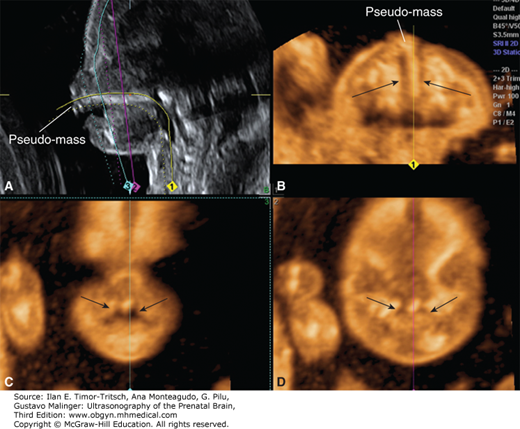
The sonographic diagnosis of cleft lip/palate in the fetus depends on demonstration of a groove extending from one of the nostrils into the lip and possibly into the alveolar ridge. With standard 2D US, facial clefts can be recognized and categorized by a combination of coronal and axial scans (Figures 16–5, 16–6, 16–7, and 16–8). In our experience, cleft lip is best visualized in an anterior coronal plane demonstrating a linear defect extending from one nostril to the oral rim, usually associated with distortion of both the upper lip and nose (Figure 16–5). Extension of the defect into the palate is better demonstrated in an axial scan of the maxilla (Figure 16–7). By angling the transducer or using the 3D approaches for the previously described posterior palate, it is usually possible to evaluate the degree of extension of the defect, that is, to identify whether the lesion is limited to the most anterior part of the palate, the alveolar ridge, or continues into the posterior palate. Indirect sonographic findings can also be noted that correlate with the type of defect. In the typical form of bilateral cleft lip/palate, the axial and sagittal views demonstrate a protrusion of the central portion of the palate and lip that is commonly referred to as the “maxillary pseudomass”2 (see Figure 16–8). We would like to emphasize that this maxillary pseudomass is generated by the “rolled up” tissue of the philtrum and appears as a hyperechoic protrusion pathognomonic on a median plane (profile) as well as a coronal or tangential plane of the upper lip. With bilateral cleft lip/palate there is a major distortion of facial anatomy and the pseudomass is a more obvious and reliable finding than the visualization of the clefts that is sometimes difficult. With unilateral cleft lip/palate or bilateral cleft lip the profile view is -usually unremarkable. Unilateral cleft lip is always associated with some degree of asymmetry and distortion of the tip of the nose and nostrils, which is maximal with cleft lip/palate. A few cases of bilateral cleft lip/palate are not associated with a pseudomass but rather to flat facies with nasal hypoplasia.21 This rare anomaly is probably closely related to median cleft lip, in that it is found with extreme flattening of the nose, usually in association with multiple anomalies and chromosomal aberrations, trisomy 18 in particular (Figure 16–9).
Figure 16–5.
Unilateral cleft lip in the third trimester of gestation. (A) Axial section of the palate demonstrating a defect in the upper lip (arrow), while the alveolar ridge appears intact. (B) Coronal section of the face demonstrating the cleft extending between the nostril and the oral cavity (arrow). (C) 3D sonogram with surface rendering in the same fetus. (Reproduced, with permission, from Visual Encyclopedia of Ultrasound in Obstetrics and Gynecology, www.isuog.org.)
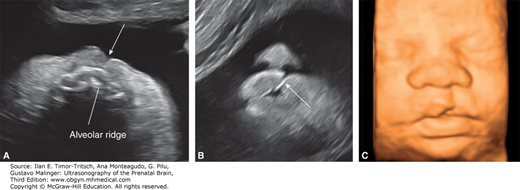
Figure 16–6.
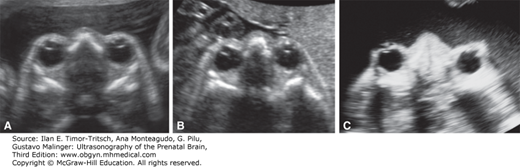
Bilateral cleft lip (arrows) in the second trimester. Coronal (A) and axial (B) 2D sonograms demonstrating the clefts in the upper lip and the intact alveolar ridge. (C) 3D sonogram with surface rendering in the same fetus. (Reproduced, with permission, from Visual Encyclopedia of Ultrasound in Obstetrics and Gynecology, www.isuog.org.)
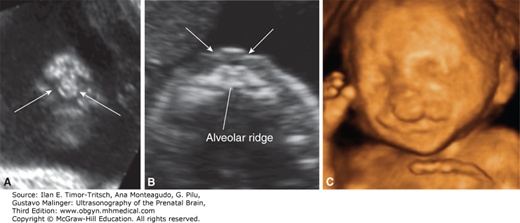
Figure 16–7.
Unilateral cleft lip/palate: multisectional analysis of an ultrasound (US) volume. (A–D) and 3D sonogram with surface rendering (E) demonstrate a square-shaped defect in the upper lip and major distortion of the nose. (Reproduced, with permission, from Pilu G. Atlas of Obstetric Ultrasound, The Global Library of Women’s Medicine, www.glowm.com.)
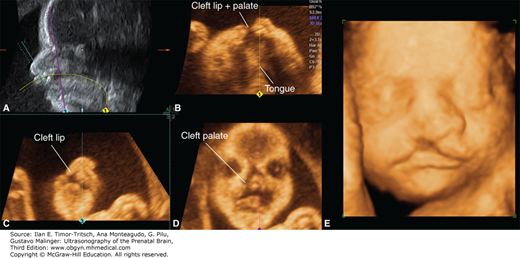
Figure 16–8.
Bilateral cleft lip/palate (arrows) demonstrated with multisectional analysis of a US volume. The anterior protrusion of the central portion of the maxilla in the sagittal (A) and axial (B) views results in a typical premaxillary pseudomass. Coronal sections (C–D) reveal the clefts extending into the palate (Reproduced, with permission, from Pilu G. Atlas of Obstetric Ultrasound, The Global Library of Women’s Medicine, www.glowm.com.)
At times, evaluation of the palate is difficult with sonography, particularly in advanced gestation. MRI is usually quite effective in depicting the normal and abnormal palate and may be used in selected cases16,17,22 (Figure 16–10).
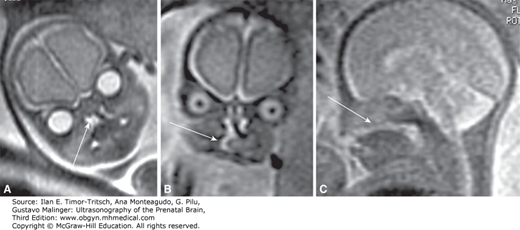
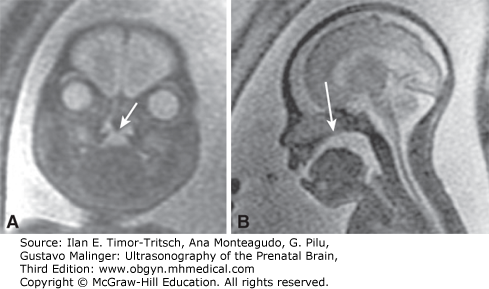
Figure 16–10.
Magnetic resonance imaging (MRI) demonstrating a fetal facial cleft. (A), (B) Coronal sections. (C) Median section demonstrating the central absence of the posterior palate. (Reproduced, with permission, from Ghi T, Tani G, Savelli L, Colleoni GG, Pilu G, Bovicelli L. Prenatal imaging of facial clefts by magnetic resonance imaging with emphasis on the posterior palate. Prenat Diagn. 2003;23[12]:970–975.)
The diagnosis of cleft lip/palate, mostly of the bilateral type, has been described even in early gestation, at 11 to 14 weeks, usually because of the demonstration of indirect signs of a premaxillary pseudomass or flat face.21,23
Prognosis depends primarily on the presence and type of associated anomalies. Mild clefts, such as lineal indentations of the lips or submucosal cleft of the soft palate, may not require surgical correction. Larger defects cause cosmetic, swallowing, and respiratory problems. Recent advances in surgical technique have produced good cosmetic and functional results. Cases that are associated with a defect in the posterior palate represent the greatest challenge for surgical correction, as the soft palate is involved in the process of swallowing and vocalizing. Furthermore, inner ear/tubal disorders may lead in time to acoustic problems and deafness.
The term cleft palate refers to a defect of the posterior portion of the palate (secondary palate) in the presence of a normal upper lip and anterior (primary) palate. Cleft lip/palate and cleft palate are two different anomalies. With exceedingly rare exceptions, recurrences are type specific. If the index case has cleft lip/palate, there is no increased risk for cleft palate, and vice versa. Roughly, 25% of facial clefts are cleft lip, 50% cleft lip/palate, and 25% cleft palate. In the vast majority of cases, cleft palate has a multifactorial etiology. Cleft of the palate may include defects of the hard palate, the soft palate, or both or the submucosal tissue.
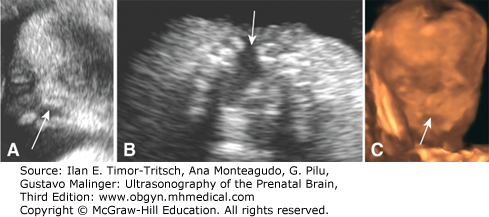
At present the US diagnosis of isolated cleft palate appears difficult. In general 2D US fails to demonstrate the condition.3,6 Specific 3D US techniques have been described to overcome the physical limitations of US (Figure 16–11), but very few accurate diagnoses have been reported thus far.24 Conversely, MRI does not suffer the physical limitations of US and is likely to be more effective than US (Figure 16–12).16,17,24
Figure 16–11.
3D sonographic diagnosis of cleft palate. (Reproduced, with permission, from Benacerraf BR, Sadow PM, Barnewolt CE, Estroff JA, Benson C. Cleft of the secondary palate without cleft lip diagnosed with three-dimensional ultrasound and magnetic resonance imaging in a fetus with Fryns’ syndrome. Ultrasound Obstet Gynecol. 2006;27[5]:566–570.)
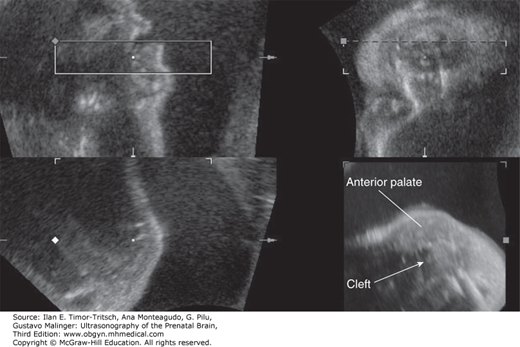
Figure 16–12.
Same case as in Figure 16–11. MRI effectively confirms the central defect in the posterior palate (arrows) both in the coronal plane (A) as well as sagittal plane (B). (Reproduced, with permission, from Benacerraf BR, Sadow PM, Barnewolt CE, Estroff JA, Benson C. Cleft of the secondary palate without cleft lip diagnosed with three-dimensional ultrasound and magnetic resonance imaging in a fetus with Fryns’ syndrome. Ultrasound Obstet Gynecol. 2006;27[5]:566–570.)
The prognosis of cleft palate depends primarily upon the presence of associated malformations. Surgical correction is frequently challenging as the soft palate is involved in the process of swallowing and vocalizing. Furthermore, tubal disorders may lead in time to acoustic problems and deafness.
About 3% of clefts occur in portions of the face different from the line joining the nostrils to the posterior palate. Their prevalence was reported to be much higher in prenatal studies due to both the high intrauterine fatality rate that is associated with some of these conditions and probably the higher detection rate due to the frequency of associated malformations.
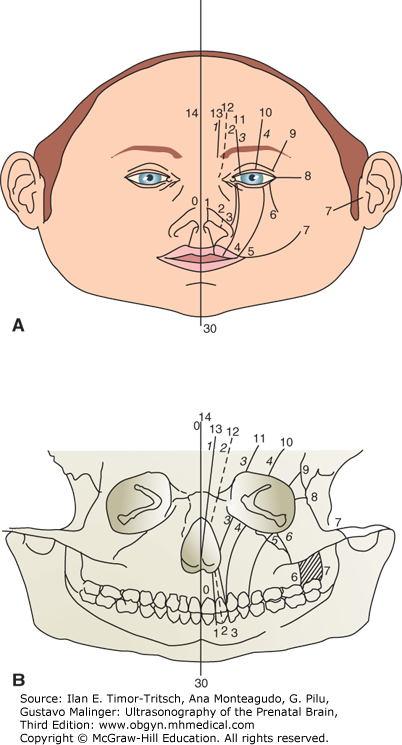
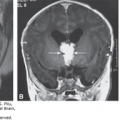
Tessier proposed a classification of these clefts that is demonstrated in Figure 16–13.25,26 The median cleft, or Tessier cleft 0, is the type most frequently reported in prenatal studies. This is a quadrangular defect in the central portion of the upper lip and palate, usually associated with a flattened nose (Figure 16–14). It accounts for 0.2% to 0.7% of all cases of cleft lip. It is considered to represent the consequence of underdevelopment of the frontonasal prominence, which normally joins the two maxillary prominences creating the philtrum. Development of the midface is induced by the prechordal mesenchyma, which is also responsible for the differentiation of the midline structures of the brain. This explains the frequent association of median clefts with orbital anomalies (Figure 16–15) and cerebral malformations. There is a striking correlation between severe holoprosencephaly and craniofacial malformations, including mostly cyclopia, median cleft lip with hypotelorism, absence of the nose, and the presence of a proboscis3,27,28 (Figure 16–16). Alobar and semilobar holoprosencephaly both have a very severe prognosis.27
Median cleft lip may also occur in association with hypertelorism (increased interorbital diatance), a combination that is pathognomonic of the median cleft face syndrome or frontonasal dysplasia.29,30 The pathogenesis is much different in these cases. The premaxilla is present, as well as the nose, which is usually bifid; however, the brain is normal in most cases.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree