Classical anatomists have provided detailed description of the arterial collateral pathways found in the head and neck. The small branches building this intricate network are difficult to access. The arterial map inherited from the anatomists has been put to the test with detailed high-resolution vascular imaging. Superselective angiography has helped rediscover the complexity of the craniocervical arterial network. The concept of dangerous collaterals or dangerous anastomoses was born with the advent of endovascular therapy. Although dangerous anastomoses of the skull base are described in the literature, variations and collateral pathways have been overlooked or misunderstood. This article reviews normal orbital arterial vascularization and its principal variations.
Over the past 200 years, classical anatomists have provided a detailed description of the arterial collateral pathways that can be found in the head and neck. The small branches building this intricate arterial network often are difficult to access, as they are located near or within the skull base. They have been revealed, one at a time, at the price of precise but painstaking anatomic dissections. The arterial map inherited from the anatomists recently has been put to the test with detailed high-resolution vascular imaging. Superselective angiography, in particular, has helped rediscover the complexity of the craniocervical arterial network under normal and pathologic conditions. The concept of dangerous collaterals or dangerous anastomoses was born with the advent of endovascular therapy. Until then, the arterial network of the skull base had been seen as a safety mechanism bringing relief to areas that had lost their primary source of blood supply (eg, the classic cerebral collateral supply through the ophthalmic artery [OA] in case of proximal carotid occlusion). With the introduction of transarterial embolization, these beneficial pathways also have become a potential source of procedural complications, mostly by inadvertent passage of embolic material into branches supplying important structures, such as the eye or the brain. In addition to confirming the accuracy of anatomic knowledge, superselective angiography has shown that most of the so-called dangerous collaterals, although consistently present under normal and abnormal conditions, may become angiographically conspicuous only under particular hemodynamic circumstances. For example, a connection between the occipital artery and the vertebral artery may “appear” (ie, become detectable) during the course of an embolization dealing with a distal occipital artery lesion. Such a phenomenon emphasizes the need for strict embolization techniques and the necessity to master the complex anatomy of these dangerous connections. Although dangerous anastomoses of the skull base are well described in the literature, the variations and collateral pathways related to the orbital arteries often have been overlooked or misunderstood, in spite of their importance for safe extraorbital embolization procedures. This article presents a review of the normal orbital arterial vascularization and its principal variations, with particular emphasis placed on abnormal pathways involving residual segments of the stapedial artery.
Arterial vascularization of the orbit
Development
The arterial supply to the orbit combines, in its normal adult configuration, vascular elements taken from several primitive arterial systems. A few basic embryonic concepts are introduced to clarify the type of variations and collateral pathways existing in the orbital region. The following summary is based on works published by Padget and by Maillot and colleagues.
Early in the fetal life, the blood supply to the eye and optic nerve is provided by two primitive OAs. The primitive dorsal OA (PrDOA) appears first in the 4-mm embryo, followed by the primitive ventral ophthalmic artery (PrVOA) in the 9-mm embryo. The PrVOA is a branch of the cranial division of the internal carotid artery (ICA) (future anterior and middle cerebral arteries), whereas the PrDOA originates from the intracranial portion of the ICA, near its termination, distally to the final point of emergence of the adult OA. The PrDOA, therefore, is not a branch of the cavernous segment of the ICA; this misconception, disseminated in the literature, finds its source in a misreading of Padget’s splendid work on the embryology of the cranial arteries. In the 16- to 19-mm embryo, the temporociliary branch of the PrDOA and the nasociliary branch of the PrVOA fuse around the optic nerve, the PrDOA becomes dominant, and the PrVOA starts regressing. Concomitantly, the PrDOA migrates from its initial point of origin to a more proximal location consistent with the origin of the adult OA. The blood supply to the connective apparatus surrounding the optic elements comes, alternatively, from the primitive orbital artery, a branch of the ramus superior of the stapedial artery (ie, the future middle meningeal artery [MMA]). These various embryonic OA components are illustrated in Fig. 1 .
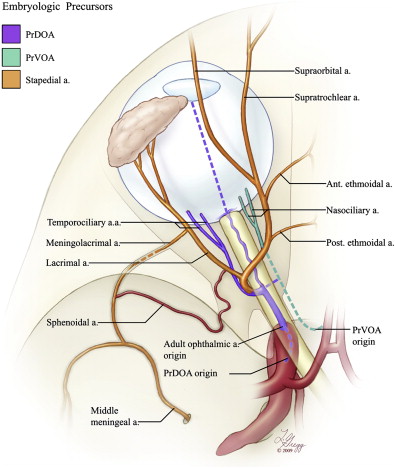
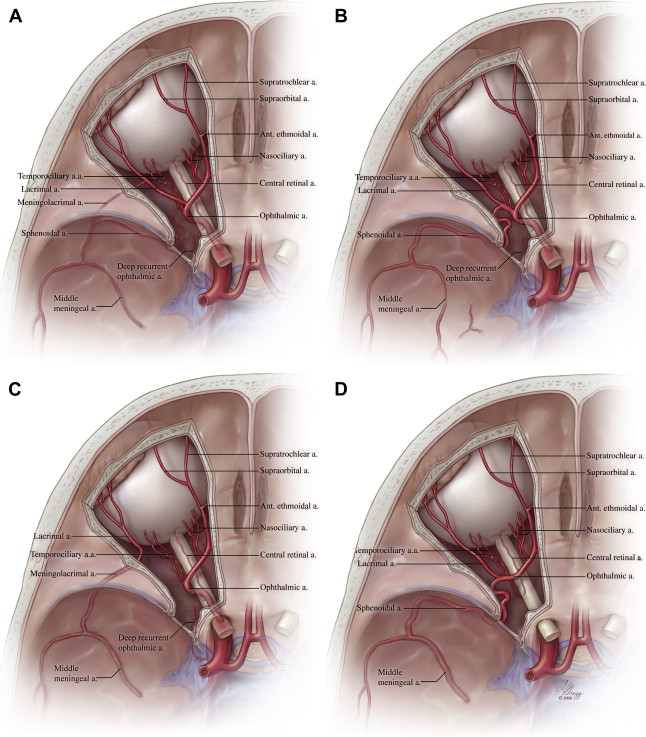
In order to reach its normal adult configuration, the OA establishes a connection with the primitive orbital artery and takes over most of the orbital blood supply. The proximal segment of the primitive orbital artery involutes to become an anastomosis between the anterior division of the MMA and the lacrimal artery. This anastomosis has classically received different names for its intraorbital and intracranial segments (ie, the recurrent meningeal branch of the lacrimal artery and the orbital branch of the anterior division of the MMA, respectively). This arterial network seems more complex than initially appreciated, however, and the existence of a double connection between the MMA and the lacrimal artery has been demonstrated by several investigators. One connecting branch is short and straight; it crosses the foramen of Hyrtl (or cranio-orbital foramen) and takes the name, meningolacrimal artery. The other is long and tortuous; it passes through the superior orbital fissure (SOF) and takes the name, sphenoidal artery. It remains uncertain if only one or both branches are derived from the stapedial artery ( Fig. 2 A ). A recent review of available phylogenetic and ontogenetic data seems to indicate that the meningolacrimal artery is the true vestige of the stapedial artery, whereas the sphenoidal artery is a neomorph that appears late in the ontogenic development and seems restricted to hominids (man and orangutans). Variations in the mode of connection of these various arteries and in their regression patterns are at the origin of the principal variants of the orbital supply and of several related dangerous collaterals. These variants (discussed later) include the origin of a part or of the whole MMA from the OA (see Fig. 2 B) and the origin of the OA or one of its branches from the MMA ( Fig. 2 C, D).
Anatomy
The ophthalmic artery and its branches
The nomenclature used in this section is based, whenever possible, on the 1998 edition of Terminologia Anatomica . The OA (arteria ophthalmica) is the first major branch of the ICA. As the site of origin of the OA usually is located at or close to the superior dural ring, it often is used as a point of demarcation between the intra- and extradural segments of the ICA. This landmark is somewhat imprecise: whereas the OA generally branches off the ICA immediately after the latter has pierced the roof of the cavernous sinus, in most cases within the subdural space, this site of origin can vary proximally from the cavernous sinus up distally to the ICA bifurcation. Rarely, the OA can originate within the two layers of the dural roof. The intracranial segment of the OA has a short course within the subdural space before it enters the optic canal. It is followed by the intracanalicular portion of the OA, which often can be identified by a slight decrease of its caliber throughout the length of the optic canal. The OA courses below the optic nerve in its intracranial and intracanalicular segments. The intraorbital segment of the OA has been divided in three parts by Hayreh and Dass. As it penetrates the orbital cavity through the common anular ring (tendon of Zinn), the OA first lies inferiorly and laterally to the optic nerve (first part). It then loops above (82.6%) or below (17.4%) the nerve to reach its superior-medial aspect (second part). This semicircular course around the nerve is a vestige of the connection between the nasociliary and temporociliary branches of the primitive OAs. From that point, the OA aims anteriorly and medially toward the orbital wall (third part), ending at the superior-medial aspect of the orbital opening, although its caliber often is already significantly reduced distal to the take off of the anterior ethmoidal artery.
A complete description of the branches of the OA is beyond the scope of this article. For more detailed reference, readers are directed to the classic work of Hayreh. It is convenient to classify the branches of the OA according to their topography. The ocular group, which includes the branches supplying the optic apparatus (central retinal, ciliary, and collateral arteries), are not discussed further. The lacrimal and muscular arteries remain within the confine of the orbital cavity and constitute the orbital group, along with various smaller branches supplying the surrounding connective structures. Finally, the extra-orbital group includes branches that exit the orbital cavity, such as the posterior and anterior ethmoidal, the medial palpebral, the dorsal nasal, and the supratrochlear arteries. Several branches of the lacrimal artery are part of this group, including the sphenoidal and meningolacrimal arteries. The first part of the OA also provides two small recurrent arteries. One of them exits the orbit through the medial aspect of the SOF and establishes a connection with the inferior-lateral trunk of the cavernous ICA (C4 segment). This branch, known as the deep recurrent OA, seems to be a stable feature of the OA anatomy and plays a significant role in several OA variations. The second recurrent branch, the superficial recurrent OA, seems less constant and represents one of the possible origins of the marginal tentorial artery.
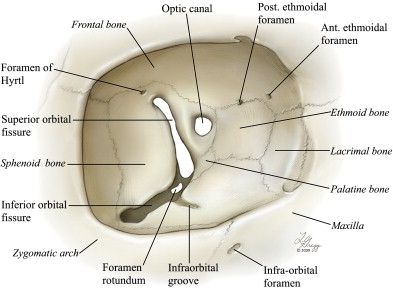
The orbital foramina
A brief description of some of the various foramina connecting the orbital cavity to its surroundings helps understand most of the variants and anastomoses (discussed later). These principal foramina are illustrated in Fig. 3 .
- 1.
The optic canal (canalis opticus) is contained within the base of the lesser wing of the sphenoid bone. The optic nerve and the OA enter the orbital cavity through the optic canal. In rare instances, the OA can penetrate a separate osseous canal that joins the optic canal near its orbital end.
- 2.
The SOF (fissura orbitalis superior) is delimited superiorly by the lesser wing and inferiorly by the greater wing of the sphenoid bone. It is pear-shaped, with a wide medial-inferior base and a narrow lateral-superior tail. The two edges of the fissure usually remain separate until they reach the frontal bone. A small foramen, the ophthalmomeningeal foramen of Hyrtl (or cranio-orbital foramen), can be seen laterally to the SOF, sometimes close enough to be confluent with its lateral extremity. When present, this foramen gives way to an anastomosis between the lacrimal artery and the MMA, the meningolacrimal artery, which is believed to represent the vestige of the superior ramus of the stapedial artery (ie, the primitive orbital artery). A second anastomosis between the lacrimal artery and the MMA, the sphenoidal artery (often confusingly identified as the recurrent meningeal branch of the lacrimal artery), is believed to be a neomorph present only in some hominids, including man. The sphenoidal artery passes through the lateral aspect of the SOF and is involved in several major OA variants. The middle portion of the SOF contains the superior ophthalmic vein, whereas the medial portion gives way to the inferior ophthalmic vein. The deep recurrent OA also passes through the medial portion of the SOF, crossing the tendon of Zinn.
- 3.
The inferior orbital fissure (IOF) (fissura orbitalis inferior) connects the orbital cavity with the infratemporal fossa and, through the latter, with the more medially located pterygopalatine fossa. Coming from the pterygopalatine fossa, the infraorbital artery (with its vein) enters the orbit via the IOF. It first follows the infraorbital groove that stems anteriorly from the IOF (sulcus infraorbitalis), then courses within the infraorbital canal (canalis infraorbitalis) and exits the orbit through the infraorbital foramen (foramen infraorbitale).
- 4.
The anterior ethmoidal foramen (foramen ethmoidale anterius) and the posterior ethmoidal foramen (foramen ethmoidale posterius) contain the anterior and posterior ethmoidal arteries, respectively. The anterior ethmoidal artery provides an important meningeal branch, the anterior meningeal artery, which enters the cranial cavity via the ethmoidal foramen of the cribriform plate.