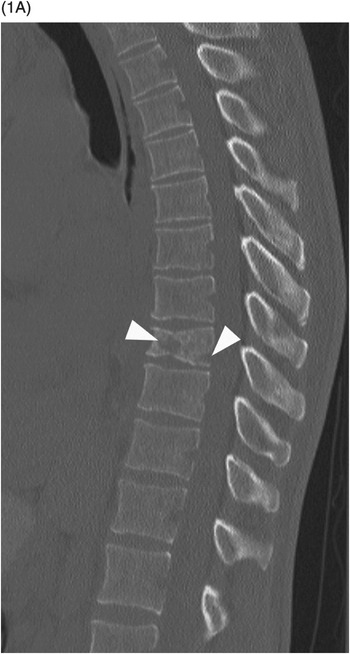
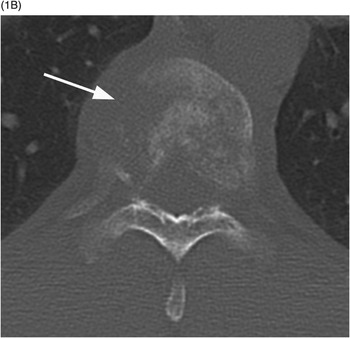
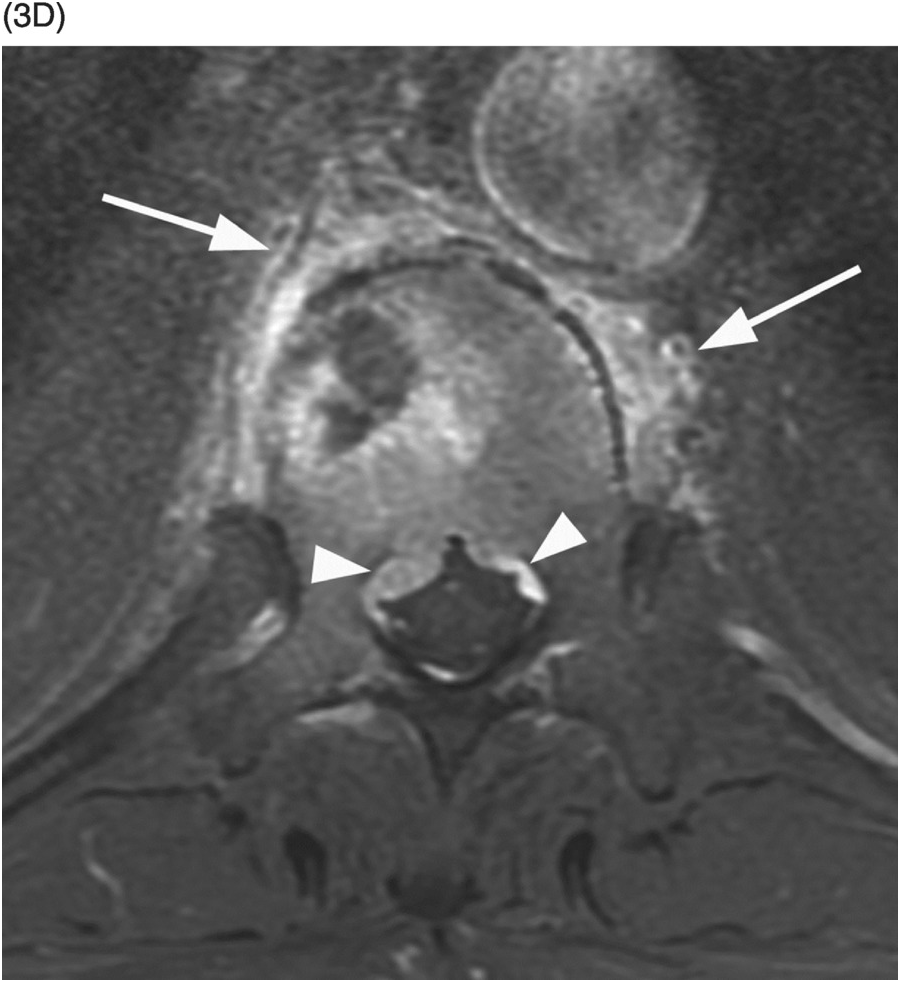
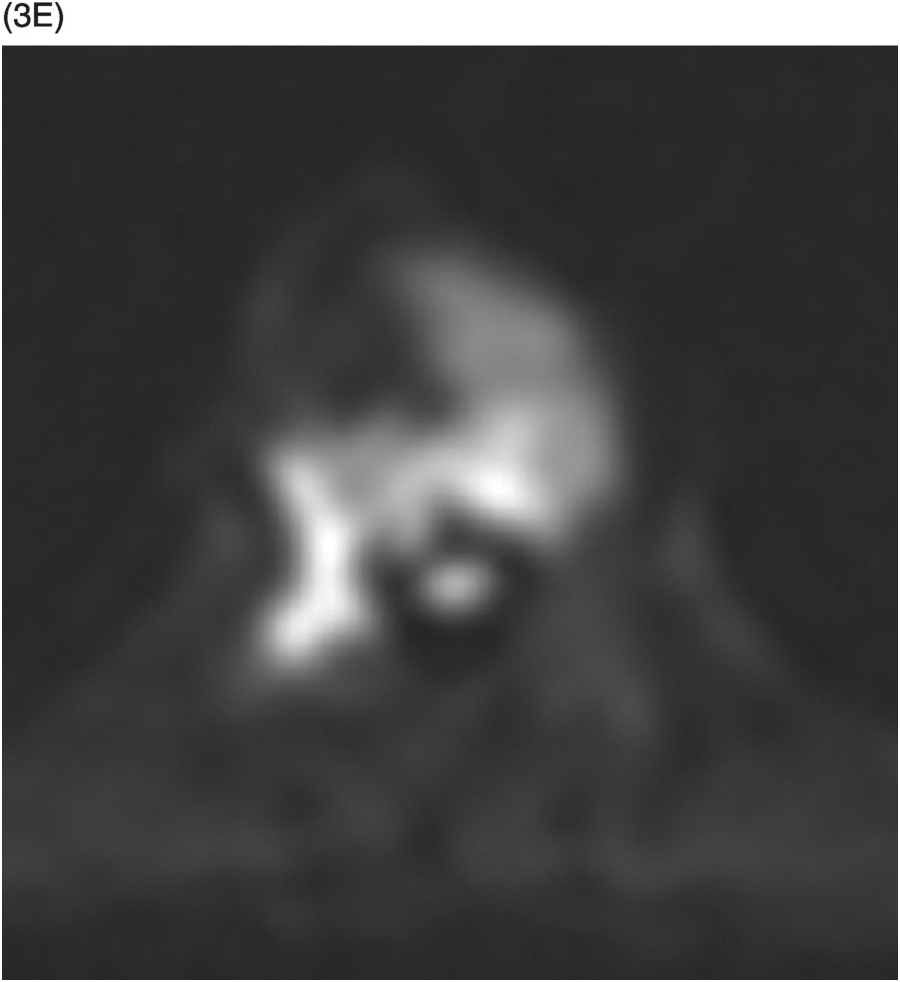
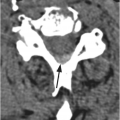
B) Axial CT through the fractured vertebra shows the altered trabecular texture with permeative lytic changes, more extensive on the right side (arrow).
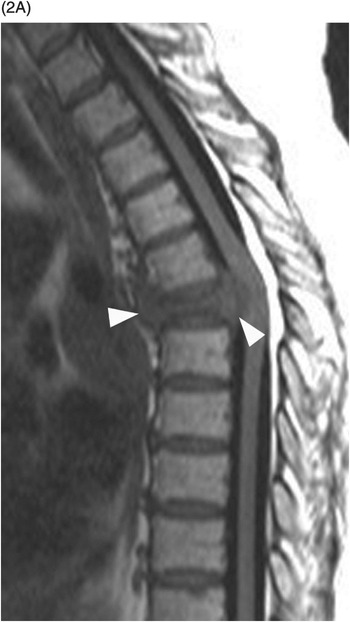
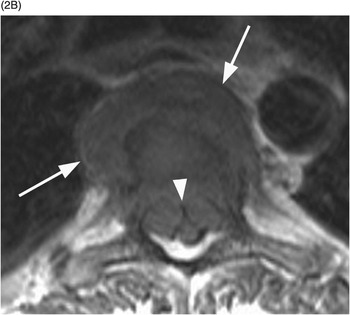
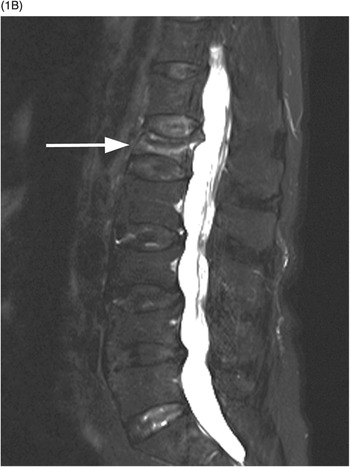
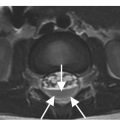
B) Axial T1w image confirms the prevertebral (arrows) and anterior epidural tissue, with a characteristic curtain sign (arrowhead).
Imaging Findings
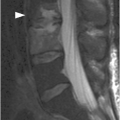
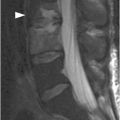
Malignant vertebral fractures are almost invariably characterized by a compression deformity. As with benign compression fractures, the vertebral body appears defomed, with a wedge, lateral wedge, biconcave, or crush deformity. MRI and CT are sensitive and accurate, and have complementary diagnostic roles. The posterior wall of the vertebral body can have a dorsally convex appearance, so-called bulging posterior wall, especially well seen on sagittal MR images. The normal signal of the bone marrow is replaced by hypointensity on T1w (darker than the disc) and hyperintensity on fat-suppressed T2w or STIR images, as well as enhancement, best seen on fat-suppressed post-contrast T1w sequences. These signal changes reflect a variable combination of neoplastic tissue infiltration, bone marrow edema, and hemorrhage. Non-fat-suppressed T2w and post-contrast T1w images without fat saturation may be completely unrevealing.
Sclerotic changes appear hypointense on T1w and T2w images, with mild, absent, or peripheral enhancement on fat-suppressed post-contrast T1w scans. The signal abnormalities can involve the vertebral body and posterior elements partially or entirely, often in a heterogeneous manner. Extra-osseous enhancing soft tissue, in continuity with the signal abnormalities within the bone, is characteristically seen in the epidural space and perivertebral regions. The anterior epidural enhancing neoplastic tissue frequently gives the “curtain sign” due to preserved median ligament. CT detects lytic lesions involving the trabecular and cortical osseous structures, with a diffuse permeative or focal pattern. Mixed pattern of lytic and sclerotic changes can also be present. In some cases there is diffuse osteopenia only, and in others no osseous structural changes are seen on CT while MRI can still be markedly abnormal. Abnormal MRI signal and CT osseous abnormalities can be present at multiple sites and levels.
Bright DWI signal, absence of signal drop on in-phase/out-of-phase imaging (both at least in part due to hypercellularity), and hyperperfusion on dynamic contrast-enhanced T1w images are frequently seen. MRI is able to detect the presence and severity of central canal and foraminal compromise, spinal cord and nerve roots compression, and signs of myelopathy, due to a variable combination of vertebral column deformity, osseous fragments’ encroachment, and epidural soft tissue component. Bone scan shows increased radiotracer uptake due to osteoblastic reaction adjacent to a lytic or sclerotic lesion, or prompted by a pathological fracture. Bone scans can be completely negative in multiple myeloma lesions due to lack of osteoblastic activation. PET (PET-CT) detects high levels of metabolic activity at the sites of malignant fractures due to presence of active neoplastic tissue.
In neoplastic spine involvement, CT and MRI, in a complementary way, detect lesions at risk of impending collapse: lytic lesions involving a large portion of the vertebral body (>30%), especially at the anterior third or the central region in the axial plane, are at greater risk of compression fracture.
Differential Diagnosis
Benign Fractures
Traumatic and osteoporotic fractures may at times be indistinguishable from malignant fractures; abnormal signal extending from the vertebral body into the pedicles, by some authors reported as a sign of malignant fractures, can be encountered in benign fractures as well; DWI, in phase/out-of phase imaging, and dynamic contrast enhancement have been reported as useful, but are not commonly used in clinical routine, and the findings and interpretation are not always unequivocal.
Band-like bone marrow edema, bone marrow sparing, fluid cleft within the fracture (fluid-like hyperintensity within the collapsed body on STIR images), and presence of healed fractures at other levels are all suggestive of a benign fracture.
Vertebral Venous Malformation (Hemangioma)
In fragility fractures due to benign vertebral hemangioma, wide intertrabecular vascular spaces might mimic lytic lesions on CT, and the typical T1 hyperintensity of hemangioma may be replaced by hypointensity due to edema associated with the recent fracture; T2 hyperintense, contrast-enhancing extra-osseous soft tissue can be seen in aggressive vertebral hemangiomas.
A diagnostic clue is the presence of coarse thickened sclerotic trabeculae interdispersed in the cancellous bone.
Spondylo-Discitis (Discitis-Osteomyelitis)
Vertebral body collapse can complicate spondylo-discitis; infectious process can be suggested by edematous changes and contrast enhancement involving the disc space and the subchondral region of adjacent vertebral body/bodies.
A very specific finding is the presence of peripherally enhancing perivertebral collections (abscess/inflammatory mass).
Clinical Findings, Implications, and Treatment
In contrast to benign osteoporotic fractures, the “crush and crumble” that very rarely cause spinal cord compression, malignant vertebral lesions due to the presence of intra and/or extra-osseous soft tissue components “crush and squeeze” so that neurological impairment is much more common. Different sets of criteria have been suggested to predict the risk of impending collapse, usually combining clinical and imaging data. The treatment goal of a malignant vertebral fracture can be radical resection, pain palliation, and/or decompression and/or stabilization. Malignant fractures or lesions at risk of collapse can be treated with many different approaches or combination thereof, including radiation, cement augmentation, decompressive laminectomy, posterior fixation, or 360-degree approaches requiring corpectomy or spondylectomy with posterior stabilization.
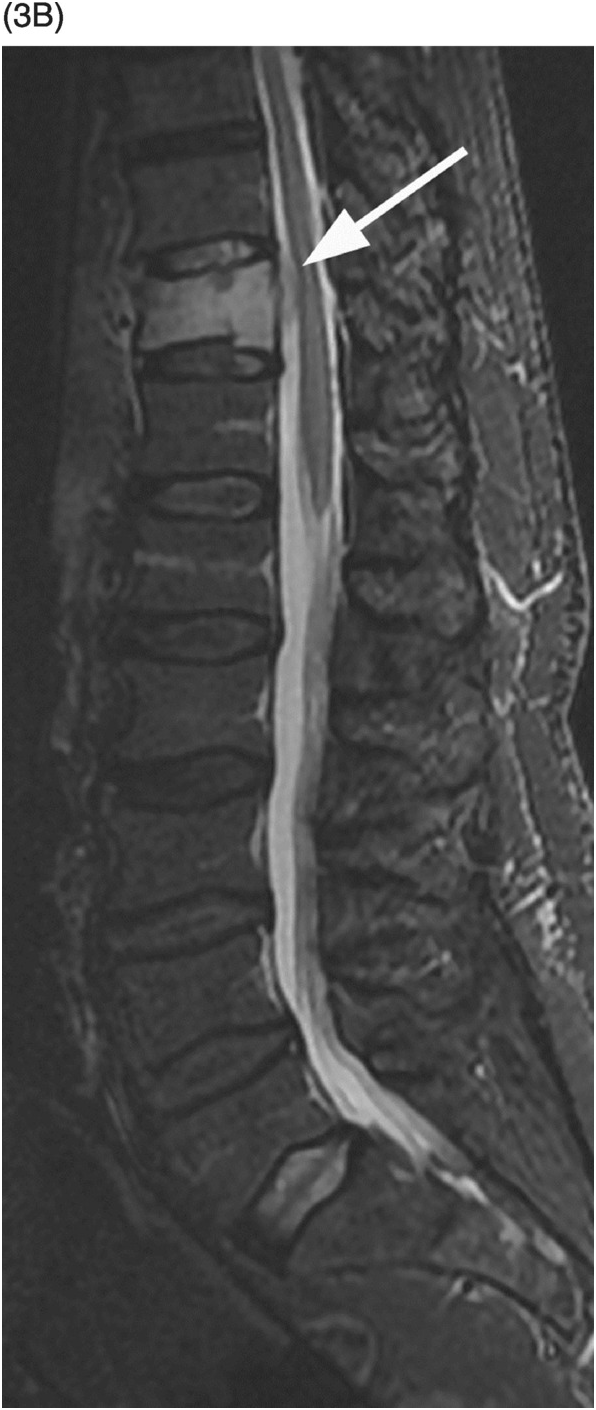
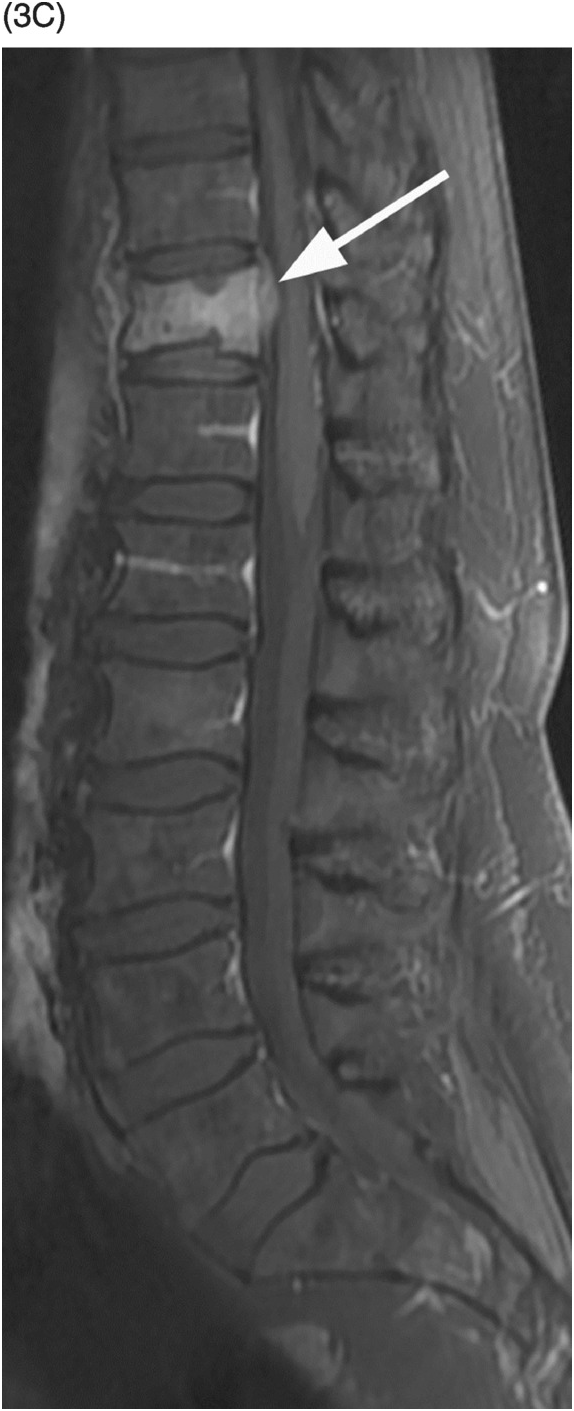
Figure 53.3 A) Sagittal reformatted CT of thoracolumbar spine shows a mild compression fracture of T11 body (arrow) in this non-osteopenic patient. Corresponding STIR (B) and post-contrast fat-suppressed T1-weighted (C) images demonstrate high signal and enhancement within the vertebral body, a nondistinctive finding, but also reveal enhancing epidural tissue (arrows), which is confirmed (arrowheads) on post-contrast fat-suppressed axial T1-weighted image (D). Note also the irregular perivertebral enhancing tissue (arrows). E) Corresponding DWI shows very bright signal within the vertebral body and right pedicle lesion, which is commonly seen in malignant fractures. Final diagnosis was lung cancer metastasis.
Additional Information
A percutaneous image-guided (fluoroscopy or CT) bone core-biopsy is often performed to differentiate a malignant fracture from a benign one, or to assess tumor type. Even in patients with a known malignancy, a vertebral fracture can have a benign etiology, for example due to osteoporosis from frequently implemented corticosteroid treatment. On the other hand, a fracture with benign imaging characteristics can still have a malignant cause, as in multiple myelomas presenting with diffuse osteopenia instead of multifocal discrete lytic lesions.
In addition to metastatic neoplasms and multiple myeloma, so-called pathological compression fractures may also be secondary to primary bone lesions, most commonly giant cell tumor, but also aneurysmal bone cyst and other etiologies.
References
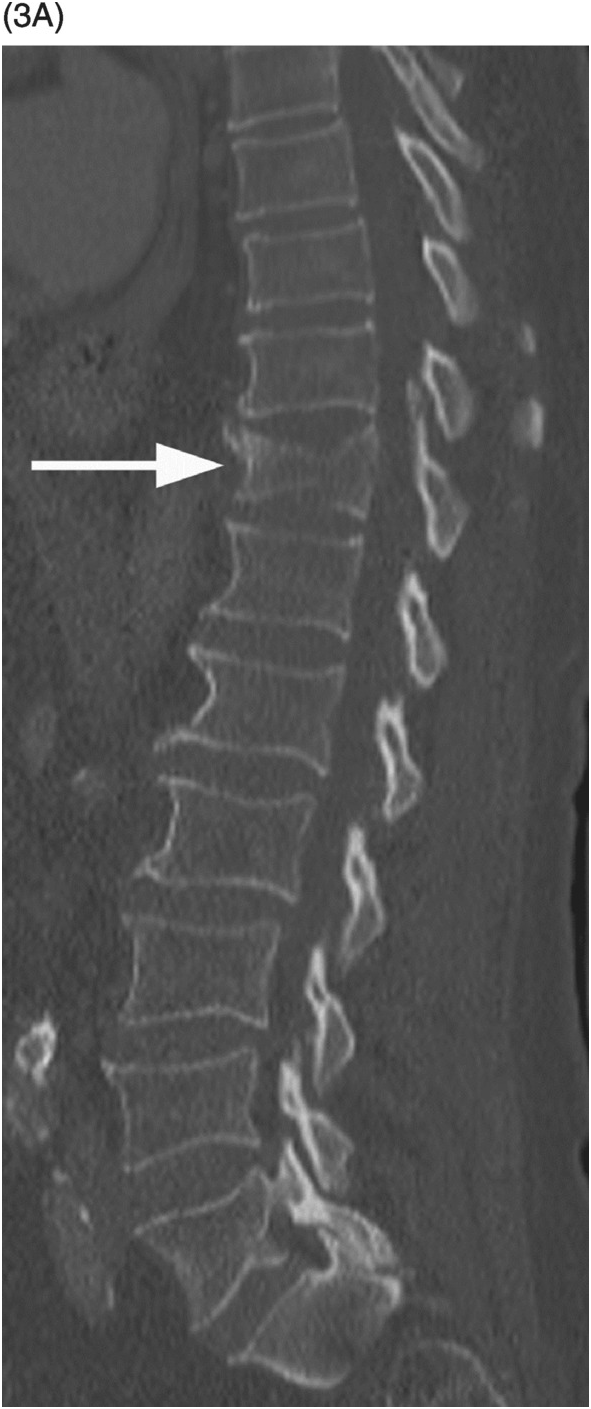
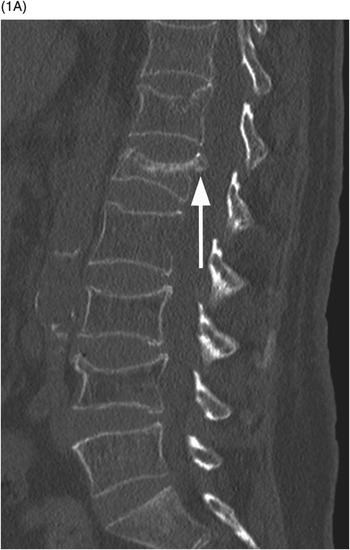
A) Sagittal lumbar spine CT shows compression fractures at multiple levels with wedging and biconcave shape, superior endplate being more commonly involved. The posterior wall is preserved, except for the typical retropulsion of the L1 superior posterior corner (arrow). The age of fractures cannot be determined on CT.