Abstract
Mammography screening trials have shown the decreased mortality benefit of early diagnosis. Nonetheless there is certainly room for improvement with new screening methods. This chapter will discuss the relative efficacy of breast screening using mammography (including digital tomosynthesis), hand-held and automated-ultrasound and MRI. Results from the breast screening trials using single or a combination of these various modalities are reviewed. In recent years, studies of high-risk women have shown that, when compared with mammography and ultrasound, dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI) is the most sensitive method for detecting breast abnormalities and is an excellent screening tool. Data concerning recent trials using MRI for women at moderate risk (15-20%), women with a prior history of breast cancer and women with mammographically dense breasts, have shown excellent results with increased cancer detection rates when compared with other modalities.
Further discussed in this chapter is the importance of not only consideration of the number of cancers detected by various methods, but also consideration of the biology of screen-detected cancers, specifically those with adverse pathologic profiles. It is increasingly evident that identification of small aggressive cancers at screening will have the greatest likelihood of achieving improved breast cancer mortality reduction, and in this respect, MRI has a distinct advantage.
2 Screening MRI: Who Should Be Screened?
2.1 Background
Breast cancer is the most common type of malignancy found among women in both developed and developing countries and remains the second leading cause of cancer death in women. According to estimates from the International Agency for Research on Cancer (IARC) there will be about 1.2 million new breast cancer cases worldwide in 2018, accounting for almost 1 in 4 cancer cases among women, and about 626 thousand breast cancer deaths. 1 Approximately one in eight women (about 12%) in the United Sates will develop invasive breast cancer over the course of her lifetime. In 2017, the American Cancer Society (ACS) estimates that 252,710 new cases of invasive cancer and 63,410 cases of in situ disease will be diagnosed in women in the United States. 2 , 3 The lifetime risk of breast cancer in men is about 1 in 1,000, with the ACS estimating that 2,470 new cases of invasive breast cancer in men will be diagnosed in 2017. Despite a decrease in breast cancer death rates since 1989, about 40,610 women in the United States are still expected to die from this disease in 2017. The decrease in death rates is likely the result of earlier cancer detection through screening, increased awareness, and improved treatment. As of March 2017, there are more than 3.1 million women living with a personal history of breast cancer in the United States, including women currently undergoing treatment and those who have completed treatment. 2
There is known to be a close association between the stage of breast cancer at diagnosis and cancer survival, even when discounting the confounding impact of various histologic tumor types and treatment regimens. Women with small cancers, less than 15 mm in size, are highly curable with 10-year survival rates greater than 90%. Women with regional disease confined to the breast and axillary nodes can expect a 10-year survival of about 80%; however, survival rates for women with metastatic disease are greatly reduced. Earlier detection of breast cancer through mammography screening results in a significant decrease in the number of advanced breast cancers, better disease-specific survival, relapse-free survival, and overall survival. Indeed, early detection allows a higher frequency of breast-conserving surgery (BCS) and fewer patients requiring severe forms of adjuvant therapy.
2.2 Mammographic Screening
Mammography has been the primary large-scale screening method for breast cancer detection in the general female population over the past five decades. The goal and expectation that early detection of breast cancer will decrease breast cancer death has been validated in multiple randomized clinical trials. 4 , 5 , 6 , 7 Mammography screening as shown in the Swedish trials resulted in a decrease in breast cancer mortality by about 30%, detecting small node-negative cancers before their clinical presentation, with the additional benefit of improving patient treatment options allowing less aggressive therapy. 4 A 30% breast cancer mortality decrease has been achieved not only in randomized controlled mammography trials but also in observational and service studies as well, with meta-analyses confirming that a decrease in breast cancer mortality begins about 5 to 7 years after the institution of screening. 5 , 8 It is important to note, however, that of the mammography screening trials, none were specifically directed toward screening of women at a high risk for breast cancer.
Despite these important gains in reducing breast cancer death, mammography has limitations, notably decreased sensitivity in women with dense breast tissue. Mammographic technique creates radiographic images of the breast that produce two-dimensional X-ray projection images that do not penetrate dense breast tissue effectively. Cancers may thus be obscured by dense overlapping tissue. The sensitivity of mammography in young high-risk women with dense breasts is low, as shown in the ACRIN-DMIST trial. Almost 50,000 women were recruited into this investigation, which was designed to compare the clinical performance of film-screen mammography with that of digital mammography. 9 The overall sensitivity of screening mammography for women with dense breasts, ACR category (c) heterogeneously dense and (d) extremely dense, ranged between 36 and 38%.
Multiple retrospective and prospective screening studies have demonstrated the limitations of mammography in high-risk patients, especially in those with a BRCA mutation. Compared to the sporadic breast cancers identified in women of average risk, cancers detected in high-risk women, those with a genetic predisposition, are usually more difficult to detect at mammography. These cancers exhibit unique imaging features and pathologic profiles; often presenting as masses at a younger age (premenopausal), with rapid growth and a tendency to present a more benign appearance by exhibiting “pushing” rather than spiculated or irregular margins. These aggressive lesions are generally noncalcified, high grade, receptor negative, and frequently favor a posterior location in the breast where detection is more difficult. 11 The majority of these cancers are larger than 1 cm in size at diagnosis, with a 20 to 56% incidence of metastatic axillary node involvement. 12 , 13
2.2.1 Breast Density
Breast density has been shown to be an independent risk factor for breast cancer. A meta-analysis in 2006 showed that women with dense breasts were at a four- to fivefold increased risk compared with other women. 14 Evidence concerning the limitations of mammography has prompted the majority of U.S. states to pass national breast density legislation. These laws generally require that women be informed if they are known to have heterogeneously dense or extremely dense breasts on mammography and that consideration of adjunctive imaging screening methods should be considered. 15 These laws impact a very large number of women because it is estimated that more than 50% of women fall into the dense breast category. Although mammography is an effective screening test for many women, the shortcomings of mammography, particularly for those women at high risk and with dense breasts, have led to an interest in pursuing adjunctive supplementary screening methods. The requirement for any alternative or supplemental screening method must be validated by studies, which document their ability to detect small node-negative cancers. Current candidate methods for adjunctive breast cancer screening include digital breast tomosynthesis (DBT), ultrasound (US), and magnetic resonance imaging (MRI).
Digital Breast Tomosynthesis Screening
DBT is a Food and Drug Administration (FDA)-approved mammographic technique. Several large clinical studies have shown an added cancer yield of about 1.25 per 1,000 women, an average 30% increase in breast cancer detection, when DBT is compared to screening with standard full-field digital mammography (FFDM). A study reported in 2019 from the Oslo Tomosynthesis Screening Trial found that addition of DBT to digital mammography resulted in significant gains in sensitivity and specificity. Additionally, synthetic mammography in combination with DBT had similar sensitivity and specificity to digital mammography in combination with DBT. 2 DBT also has the added important advantage of reducing recall rates and improving the positive predictive value (PPV) of recall and biopsy recommendations. 16 , 17 , 18 Central to the outcome of a screening program is the consideration not only the number of cancers detected, but also the biologic profile of those detected. Although more research is needed, the studies show that DBT has a propensity to identify cancers that are associated with well-differentiated lower-grade disease, when compared with small higher grade cancers that are found with screening MRI.
Ultrasound Screening
Studies have consistently shown that whole breast ultrasound (WBUS) will detect an additional 2 to 4 cancers per 1,000 women screened beyond those detected at mammography. 19 , 20 , 21 Supplemental WBUS in addition to mammography is now the most commonly used method for adjunctive screening of women with dense breasts. Multiple prospective ultrasound screening studies have found an increased cancer detection rate of about 4 per 1,000 women compared with mammographic screening in the same group of individuals, with a concomitant reduction in the interval cancer rate.
Handheld Ultrasound Screening
The additive value of handheld ultrasound screening (HHUS) in women with heterogeneously dense or extremely dense breast tissue and at least one other high or intermediate risk factor was evaluated in the ACRIN-sponsored 6666 trials. 19 Berg and colleagues investigated 2,662 women who underwent three rounds of annual mammography and HHUS screenings between April 2004 and February 2006. Cancer yield overall was 111, 33 (30%) detected on mammography only, 32 (29%) by ultrasound only, and 26 (23%) by both mammography and ultrasound; 11 (12%) were not detected by either screening method. Supplemental screening ultrasound identified 3.7 cancers/1,000 screenings. Of the cancers found only at ultrasound, 94% were invasive with a median size of 10 mm and 96% were node negative. The reported interval cancer rate was low at 8%. However, rates of biopsy for findings seen only on ultrasound were high (5%) on incidence screens, with a low malignancy rate (7.4%). It is important to note that after three negative consecutive mammographic and ultrasound screening rounds, a subset of 612 women underwent a single MRI screening examination. Cancer yield among these women was 14.7/1,000, a significant increase when compared to the 4.2/1,000 detection rate in the same cohort of women who received mammographic and ultrasound screening in the prevalent round. Of the MRI-detected cancers, 89% were invasive, median size was 8.5 mm, and 100% (of those staged) were node negative.
Automated Breast Ultrasound Screening
A large multicenter study comparing mammography screening with automated breast ultrasound screening (ABUS) was conducted between 2009 and 2011 and included a total of 15,318 women with heterogeneously or extremely dense breasts; 112 cancers were identified, 82 on mammography and 30 on ultrasound. The additional cancer yield with ultrasound was 1.9/1,000 women screened; 62.2% of the mammography-detected cancers were invasive compared with 93.3% of the cancers identified at ABUS. Ultrasound increased the recall rate from 15 to 28.5% (285/1000), mammography yielding 1 cancer for every 28.1 recalls, and ultrasound yielding 1 cancer for every 68.7 recalls. An additional 552 biopsies were performed to identify 30 “US-only” cancers. 20
Limitations of Screening with Ultrasound
Although current screening practice for women with dense breasts now consists of annual mammography supplemented by WBUS, there are several limiting factors. These are principally due to the high biopsy rates and unwieldy short-term follow-up rates of screening with WBUS and increased radiologist interpretation time. 19 , 20 Other limiting factors include the time to conduct the examination; on average, a bilateral ultrasound screening study in the 6666 ACRIN took just under 20 minutes to complete. The low PPV of WBUS-generated biopsies and the high rates of short-term interval follow-up recommendations result in increased downstream costs. 21 Sprague et al estimated the cost per QALY gained by screening women with mammography and supplemental ultrasound to be $320,000, a significant burden on health care costs. 22
Because of the limitations of WBUS outlined above, alternative supplemental screening tests have been investigated to screen women with dense breasts, notably abbreviated breast MRI (AB-MR). A short MRI scan time, reduced to less than 10 minutes, has been shown to be equivalent in time and cost to the standard combination of mammography and WBUS. Early studies involving interpretations of an abbreviated MRI protocol (AB-MR) have shown equivalent sensitivity for cancer detection when compared to a full standard MRI, with only minimal decrease in specificity. 23 , 24 The growing evidence for an abbreviated MR protocol suggests a strong benefit for the expanded use of breast MRI for screening of a larger section of the female population. The abbreviated MRI technique will be discussed further in Chapter 3.
2.3 MRI Screening for High-Risk Women
In recent years, studies have shown that, compared with mammography and ultrasound, dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI) is the most sensitive method for detecting breast abnormalities and is an excellent screening tool. MRI uses magnetic fields to produce cross-sectional images of soft-tissue structures. The contrast between normal breast tissue consisting of adipose and fibroglandular structures, and breast lesions, depends on the mobility and magnetic environment of the hydrogen atoms in the water and fat of these tissues. Gadolinium-based contrast agents (GBCA) are injected intravenously during MRI to improve detection of cancers and other lesions. 25 , 26 , 27 The advantages of MRI include its high sensitivity for detection of invasive breast cancers and most in situ cancers and unlike mammography, the sensitivity of MRI is not limited by breast density, postsurgical or postradiation changes or the presence of breast implants.
The superior sensitivity of breast MRI compared to other breast imaging methods has been shown in women with a familial increased risk for breast cancer. This evidence has resulted in recommendations from the ACS, the National Comprehensive Cancer Network (NCCN), and joint recommendations from the Society of Breast Imaging (SBI) and the American College of Radiology (ACR) that MRI be used as an adjunct to mammography screening to improve breast cancer detection in high-risk women. 28 , 29 , 30 , 31 , 32 Supplemental screening with breast MRI is recommended for those women who are carriers of BRCA1, BRCA2, mutation, for their first-degree relatives with a BRCA mutation (tested or untested), for those with a lifetime risk of 20 to 25% or greater, and for those with a clinical history of chest irradiation between ages 10 and 30 years. 31 , 33 Annual screening with MRI and mammography is also recommended for women with less common, specific genetic mutations such as Li–Fraumeni syndrome, Cowden’s and Bannayan–Riley–Ruvalcaba syndromes (TP53 gene mutations) or their first-degree relatives.
2.3.1 Women with a Genetic Predisposition for Breast Cancer
Clinical features indicating that a woman may be at high risk for breast cancer caused by a high-penetrance gene include close relatives with a history of breast or ovarian cancer (two or more) occurring before age 50. Harmful mutations of the BRCA tumor suppressor genes result in a greater lifetime risk of malignancy particularly for breast and ovarian cancers, and are estimated to account for 5 to 10% of all newly diagnosed breast cancers. 34 The BRCA gene mutation can be inherited from either parent and passed on to both daughters and sons in an autosomal dominant pattern of transmission. Each first-degree relative of a BRCA mutation carrier has a 50% chance of also being a carrier of the mutated gene. Women with no known risk factors have an average lifetime breast cancer risk of 12.3%, whereas women with a BRCA mutation have a 55 to 65% (BRCA1) and a 45% (BRCA2) risk of developing breast cancer by age 70. An increased risk of up to 63% for development of a second ipsilateral cancer or a contralateral breast cancer is found in women with a BRCA1-associated breast cancer, the highest risk conferred in women with a primary cancer diagnosed before age 40. 35 , 36 , 37 , 38 A woman’s risk nearly doubles if a first-degree relative (mother, sister, and daughter) is known to have a personal history of breast cancer. Genetic counseling should be recommended for women who are found to be at increased risk of carrying a BRCA gene mutation.
Fewer than 15% of women diagnosed with breast cancer have a history of breast cancer in a family member. Breast cancer is more common in African-American women than white women, when diagnosed under age 45, and have an increased likelihood of dying from breast cancer. Asian, Hispanic, and Native American women have a lower risk of developing and dying from breast cancer. Women with known genetic mutations are associated with a high lifetime risk of breast cancer estimated at 50 to 80%. Supplemental MRI screening of BRCA mutation carriers should begin at an early age and is essential for early and accurate cancer diagnosis. Management options for women with a BRCA mutation include enhanced screening with MRI, prophylactic mastectomy, and/or oophorectomy, and chemoprevention. Treatment with tamoxifen has been shown to reduce the risk of receptor-positive tumors for women who are known BRCA1 or BRCA2 mutation carriers.
The BRCA1 Gene
The BRCA1 gene located on chromosome 17 is thought to be responsible for the hereditary breast and ovarian cancer syndrome (HBOC). BRCA1-associated breast cancers account for 50% of familial breast cancers and 5 to 8% of all breast cancers. The associated cancer risk is known to decrease with increasing age. These cancers are usually high grade and invasive, differing from sporadic cancers in that they are often aneuploid and of the basal type triple negative molecular subtype, 39 , 40 and exhibit 19% prevalence for the medullary subtype of invasive ductal carcinoma (IDC), a subtype rarely diagnosed in women with sporadic cancers (< 1%). BRCA1-associated breast cancers constitute 15% of medullary cancers in the general female population.
The BRCA2 Gene
The BRCA2 gene located on chromosome 13 accounts for approximately 35% of familial breast cancers. BRCA2 carriers have been known to confer a high risk for other cancers including prostate, colon, bladder, pancreatic, fallopian tube, and male breast. BRCA2-associated cancers are estrogen receptor positive (> 75%) and triple negative (16%). 41 They have been found to be of a higher histologic grade than tumors identified in age-matched control studies compared to sporadic cancers 42 and their rates of associated ductal carcinoma in situ (DCIS) are similar to rates found in sporadic breast cancers. 43 , 44 No increase in frequency of the medullary IDC subtype is found in these lesions. Mutations in other less common genes, such as the TP53 and PTEN genes, are also known to confer a high risk for breast cancer, and women with these gene mutations should benefit from supplemental MRI screening as well.
An important metric for judging the success of breast cancer screening is not only the sensitivity of cancer detection and cancer size at diagnosis, but also the interval cancer rate. For average-risk women, the interval cancer rate at mammography screening is generally between 30 and 50%, the rate is about 20% in breast ultrasound screening and 0 to 6% rate for MRI screening of high-risk women. The low interval cancer rate at MRI screening is especially important, given that these cancers tend to be rapidly growing and aggressive. Rapid tumor growth, especially in young BRCA mutation carriers, accounts for the high interval cancer rate found at mammography screening, double that of nonmutation carriers. 13 , 45
Women with a Lifetime Risk of 20 to 25% or Greater
How do we identify women with a risk of 20 to 25% or greater? The majority of high-risk women selected to undergo supplemental screening with MRI do not have an identified genetic mutation, but rather a strong family history of breast cancer. Estimation of breast cancer risk is usually calculated by applying models that primarily evaluate family history. The prediction model of breast cancer is a mathematical equation designed to quantify the risk that an individual woman would develop breast cancer within a defined period. Risk-prediction models that are commonly used in clinical practice include the Gail, Claus, and Tyrer–Cuzick BOADICEA and BRCRAPRO models. 46 , 47 , 48 , 49 , 50 Among these prediction models, various factors are incorporated to quantify breast cancer risk and they differ from one model to another. For example, the Gail model factors in only a first-degree relative, 46 whereas the Tyrer–Cuzick model includes both family history and a history of a high-risk lesion diagnosis (lobular carcinoma in situ [LCIS] and atypical ductal hyperplasia [ADH]), not included in other risk-prediction models. 48 There are limitations to the consistency and accuracy of breast cancer risk-prediction models as shown in a recent meta-analysis that included 18 prediction models and 7 validating studies. The authors found only poor-to-fair discriminatory accuracy in internal and external validation and recognized an important need for development of a new reliable risk-prediction model. 51
Nonetheless, although the current risk models are indeed imperfect, they remain the standard means of assessing breast cancer risk for the majority of the female population; indeed, most of the women who participated in the large prospective screening studies that evaluated breast MRI sensitivity and specificity were evaluated by these models. The 2000 and 2005 National Health Interview Survey and the National Cancer Institute Breast Cancer Risk Assessment Tool (Gail model 2) estimate that 880,063 (1.09%) of U.S. women aged 30 to 84 years have a lifetime absolute breast cancer risk of 20% or more, and are thus eligible for MRI screening. 52
MRI Screening Trials of High-Risk Women
An early MRI screening study published by Kuhl et al in 2000 evaluated a cohort of 192 women with known or suspected carriers of a breast cancer gene mutation and found that of the 9 cancers diagnosed, 6 were visible only on MRI and were occult at mammography and ultrasound. 53 One of the largest MRI screening trials published by Kriege and colleagues in 2004 divided the patient cohort of 1,909 women into mutation carriers (50–85% lifetime risk), and women at high risk of developing breast cancer due to family history (20–29% lifetime risk) and the moderate risk group (15–20% risk). The overall rate of detection for all breast cancers both invasive and in situ was 9.5 per 1,000 woman-years at risk, with the highest rate, 26.5 per 1,000, in women with a known genetic mutation. The overall sensitivity of MRI was 79.5% compared to 17.9% for clinical breast examination and 33% for mammography. 28
Multiple prospective high-risk screening studies followed these early reports, all aimed to compare the diagnostic accuracy of MRI with screening mammography with or without ultrasound (Table 2‑1). The risk factors for women recruited to these later studies varied widely, including women with a known, or likely, genetic mutation, women with a strong family or personal history of breast cancer, and women with a prior diagnosis of a high-risk lesion (LCIS or ADH). 29 , 30 , 54 , 55 , 56 Despite the differences in the entry risk criteria, reports yielded concordant results, finding that MRI screening was consistently more accurate than screening with mammography and ultrasound. The sensitivity of breast MRI in all studies was approximately 90% range (71–100%), significantly higher than that of mammography range (13–59%). When data from 11 studies were combined in a meta-analysis, it was found that there was an overall sensitivity of 77% for MRI alone, 94% for a combination of MRI and mammography, and 39% for mammography alone. 57 The relatively low sensitivity (71–77%) observed for MRI in the early screening trials can be explained in part by false-negative MRI diagnoses of DCIS. This problem is likely due to older technology with limited spatial resolution and lack of knowledge concerning the interpretive criteria necessary for recognition of the subtle nonmass findings seen in DCIS. A criticism of MRI in the early years of high-risk screening trials concerned the lower specificity of MRI than mammography. In recent years, the specificity of MRI has improved as radiologist experience has increased, and the current PPV3 of biopsy rates and short interval follow-up rates for MRI are comparable to those of mammography.
Author (year) | Cancer yield | MG | US | MR | Interval cancers | Cancer yield MR only |
Sardanelli (2011)89 | 52/501 10% | 50% | 52% | 91% | 3 | 16/501 3.2% |
Rijnsburger (2010)90 | 97/2,157 4% | 41% | – | 71% | 13 | 44/2,157 2% |
Hagen (2007)91 | 25/491 5% | 50% | – | 86% | 5 | 8/491 1.6% |
Lehman (2007)92 | 6/171 3.5% | 33% | – | 100% | N/A | 4/171 2.3% |
Kuhl (2005)29 | 43/529 8% | 33% | 40% | 91% | 1 | 19/529 3.6% |
Leach (2005)30 | 35/649 5% | 40% | – | 77% | 2 | 19/649 2.9% |
Kreige (2004)28 | 51/1,909a 3% | 40% | – | 71% | 4 | 22/1909 1.2% |
Warner (2004)93 | 22/266 9% | 36% | 33% | 77% | 1 | 7/236 3% |
a24. 1 of 51 cancers detected was non-Hodgkin’s lymphoma. | ||||||
The EVA trial, published in 2010, studied intermediate- to high-risk women, investigating the respective contributions of screening with mammography, ultrasound and MRI, to the individual diagnosis of breast cancer. 58 The sensitivity of mammography alone was about 33%, similar to the findings in the ACRIN-DMIST trial 9 for digital mammography screening of women with BIRADS category (C) and (D) breast tissue density. The addition of ultrasound to mammography screening yielded an overall sensitivity of 48%, whereas MRI alone yielded a sensitivity of 93%. Three low-grade DCIS cancers in this trial were undetected at MRI. Nonetheless, the authors concluded that MRI could be used as a stand-alone imaging method because the missed cancers in this trial were low-grade lesions, considered “insignificant,” thus preventing overdiagnosis of lesions that, if followed, would not harm the patient. The high sensitivity of MRI for biologically significant disease in this study could be viewed as a method of avoiding the problem of underdiagnosis, by identifying and treating small node-negative aggressive lesions that would harm the patient if left undetected.
Riedl et al conducted a prospective, nonrandomized comparison study in 2015, reporting on 559 women with 1,365 complete imaging rounds (2.45 rounds per woman) who underwent annual high-risk screening with mammography, ultrasound, and MRI. 59 Women entered into the study were high risk, either BRCA1 or 2 mutation carriers (28%) (n = 156) or those found to have a greater than 20% lifetime risk based on family history. MRI sensitivity (90%) was significantly higher than the sensitivity of mammography (37.5%), ultrasound (37.5%), and mammography combined with ultrasound (50%), (p < 0.001 for all methods). MRI identified all 14 cases of DCIS, mammography and ultrasound each detected 5 cases (35.7%), and a combination of mammography and ultrasound methods detected 7 cases (50%). No cancers were detected by ultrasound alone. The authors concluded that MRI allows early detection of breast cancer in high-risk patients regardless of patient’s age, breast density, or risk status, and also proffered the opinion that when MRI screening is utilized, the added value of mammography is limited and there is no added value of ultrasound.
Today, the recommended screening protocol for women at high lifetime risk for breast cancer is a combination of annual mammography beginning at age 30 and MRI screening beginning at age 25. The two annual examinations can be scheduled at 6-month intervals. 60 Adding ultrasound to this protocol has not shown to increase cancer detection rates. 58 With increasing breast MRI expertise and technologic advances, routine use of screening mammography in women undergoing screening breast MR imaging is again under question. A clinical service study by Lo and colleagues in Canada, published in 2017, sought to evaluate the added value of cancer detection of mammography when high-risk women undergo screening with MRI. 61 The cancer detection rate for MRI was 21.8 cancers per 1,000 examinations (95% confidence interval [CI]: 15.78, 29.19) and that for mammography was 7.2 cancers per 1,000 examinations (95% CI: 3.92, 11.97; p = 0.001). MRI detected smaller cancers than those detected on mammography. Sensitivity and specificity of MRI were 96 and 78% respectively, and those of mammography were 31% and 89%, respectively (p = 0.001). PPV for imaging recalls was 9.3% for MRI and 6.5% for mammography. The authors found no mammography screening benefit, that is, mammography depicted no MRI-missed cancers and concluded that the routine use of screening mammography in women undergoing screening breast MRI warrants reconsideration.
2.4 Women with a History of Chest Irradiation
Breast cancer is the leading cause of death in long-term survivors of mediastinal and chest irradiation, many of whom were treated for Hodgkin’s lymphoma at a young age. The risk of subsequent development of breast cancer is greatest in those women who were treated between the ages of 10 and 30 years, because radiation sensitivity is highest in this age group. 62 , 63 Breast cancer risk has been shown to be highest among women treated during the time period from the early 1960s to the mid-1970s when higher radiation doses were used compared to subsequent years. 64 Studies have shown that the incidence of breast cancer in women with a history of irradiation is similar to that of women with a BRCA mutation, breast cancer occurring in about 13 to 20% of women by the age of 40 to 45. The ACS, ACR, and the Children’s Oncology Group recommend annual screening MRI as an adjunct to mammography in women with a history of chest irradiation. 31 The screening protocol for women who have been treated with moderate- to high-dose mediastinal and/or chest irradiation includes annual screening with mammography and MRI beginning at 8 years following completion of treatment but not before age 25. 65 , 66 The cumulative radiation risk depends on the dose and volume of the radiation field and the time interval since therapy was completed. 62
Two retrospective studies reported on the utility of supplemental breast MRI screening in women with a history of mediastinal/chest irradiation. Sung and colleagues performed a review of 247 screening examinations in 91 women and reported a 4.4% incremental cancer detection rate increase with MRI when compared to mammography. Ten cancers were identified in the study period. Among these, four cancers were seen on MRI alone (early-stage T1 invasive cancers) and three with mammography alone (DCIS and DCIS with microinvasion). The authors concluded that MRI is a useful adjunct to mammography screening in women with this history. 67 Other investigators reviewed the medical records of 98 patients with a prior history of chest radiation therapy; all had undergone both screening mammography and MRI between January 2004 and July 2010. 68 Analysis of 558 screening examinations (296 mammograms and 262 MRI) yielded an incremental cancer detection rate of 4.1% (95%, CI 1.6–10%). Of the 13 cancers detected, 12 (92%) were found on MRI and 9 (69%) by mammography. The median latency from completion of radiation to detection of the breast cancer was 18 years (range, 8–37 years). The authors concluded that both MRI and mammography should be used to screen women in this high-risk group.
Despite the recommendation that women with a history of chest irradiation benefit from screening with mammography and MRI, many women in this category are not adequately screened. Lack of compliance with MRI screening recommendations is evident in a study of women participating in the Childhood Cancer Survivor Study (CCSS), a North American cohort of long-term survivors diagnosed from 1970 to 1986. 66 In this group of women, 63.5% of those aged 25 through 39 years, and 23.5% of those aged 40 through 50 years, failed to be screened with mammography during the prior 2 years. Screening MRI was not included in the study design.
2.5 Women at Moderate Risk (15–20%)
Women who fall into this category include women with a personal history of invasive or in situ breast cancer, women with a prior biopsy or excision yielding a high-risk lesion such as ADH or lobular neoplasia, and women with heterogeneously dense or extremely dense breasts on mammography. Recent literature supports the use of screening MRI in addition to mammography in patients with a moderate risk of developing breast cancer. The 2018 ACR Practice Parameters for Breast MRI state that screening MRI may be considered as a supplement to mamography to screen women at intermediate (moderately elevated) risk of breast cancer (15%-20%). Annual screening MRI is also recommended for women with a personal history of breast cancer, those with dense tissue at mammography, or those women diagnosed with breast cancer under the age of 50. 32 Current guidelines also recommend that individual facilities in conjunction with referring clinicians may be in the best position to decide whether to screen these patients. 69 The NCCN and SBI/ACR guidelines also state that screening MRI should be considered in these moderate-risk patients.
2.5.1 Women with a Personal History of Breast Cancer
Women with a personal history of breast cancer are generally classified as moderate risk; the current recommendations by ASCO, the ACS, and the NCCN for surveillance of women with a personal history of breast cancer are clinical examination and mammography. 31 , 33 , 70 , 71 These women are at a higher risk of developing a breast cancer recurrence or a second breast cancer, depending on their age at cancer diagnosis and the presence of other risk factors. A retrospective review of breast MRI examinations in 144 women with a personal history of breast cancer, without a family history, evaluated the stage at diagnosis of recurrent lesions. Cancer was detected in 12% of women (n = 18) and of these, 10/18 cancers were solely identified on MRI. Of the 10 “MRI-only” cancers, 4/10 were noninvasive and 7/10 were minimal cancer lesions, defined as DCIS or node-negative invasive cancer less than 1 cm in size. The PPV of MRI screening was 39%. 72 Another study conducted between January 2008 and March 2012 in Korea followed 607 consecutive women (median age, 48 years; age range, 20–72 years) who had undergone BCS. Preoperative MRI was performed in 92% of the study cohort. All women had negative mammography and US findings and underwent posttreatment surveillance MRI. Of the cancers detected at MRI screening, 11 additional cancers were found, (18.1 cancers per 1,000 women), 8 invasive, 3 DCIS (median invasive size, 0.8 cm, range, 0.4–1.4 cm), all node negative. Results specified a PPV for recall of 9.4%, PPV for biopsy of 43.5%, sensitivity 91.7%, and specificity 82.2%. 73 Other beneficial supporting evidence for MRI screening in this group is found in a study that showed recurrent tumor to be more likely to be mammographically occult if the primary tumor before treatment was not visible at mammography. 74 A recent report on breast MRI screening in women (n = 1,521), by Lehman and colleagues, 75 found equally high cancer detection rates in women with a personal history (PH) but no known genetic or family history (GFH) of breast cancer, compared to the detection rates of patients of breast cancer in the GFH group. The overall MRI sensitivity was 79.4% for all cancers and 88.5% for invasive cancers. False-positive examinations were lower in the PH versus the GFH group (12.3 vs. 21.6%, p < 0.001), specificity was higher (94 vs. 86%, p < 0.001). Sensitivity and cancer detection rate were not statistically different (p > 0.99). More than half of the cancers detected were invasive, the latency period being 5 years after treatment. The authors concluded that the diagnostic performance of screening MRI is superior in women with a personal history of breast cancer compared with women with a genetic or family history of breast cancer.
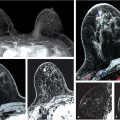
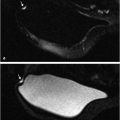
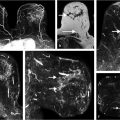
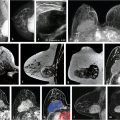
The MRI screening results conducted on women with a personal history of breast cancer as shown above provide convincing evidence that MRI benefits these patients by detecting breast cancers while they are still small, node negative, and undetectable by other imaging methods (Fig. 2‑1).

Patient age 37, without a family history of breast cancer, was treated for right breast DCIS 2 years ago, undergoing mastectomy and reconstruction with a silicone implant. MIP image (a, arrow) shows a 4-mm irregular enhancing mass in the left breast, without a correlate on the T2w series. A right breast silicone implant is noted (b). Postcontrast T1w image (c) and subtracted image (d) show margin irregularity with persistent kinetics noted on angiomap (e). Reformatted sagittal and coronal images are shown. Histology: DCIS intermediate/high grade at MRI-guided biopsy (f, g).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree