7 Detection and Management of Unexpected Incidental Pulmonary and Nonpulmonary Findings
Summary
This chapter focuses on some of the more commonly encountered incidental pulmonary and nonpulmonary findings encountered on lung cancer screens, highlighting typical CT (computed tomography) imaging findings as well as the potential clinical implications of such. The qualitative visual or ordinal scoring of the extent of centrilobular emphysema, coronary artery calcifications, and osteoporosis with compression fractures are discussed in detail with illustrative case examples.
Keywords: incidental finding, unexpected finding, COPD, coronary artery disease, osteoporosis, pulmonary Langerhans’ cell histiocytosis, respiratory bronchiolitis, desquamative interstitial pneumonia, acute eosinophilic pneumonia, thyroid, ordinal score, Agatston
7.1 Introduction
As any radiologist can attest, incidental findings are quite prevalent on radiologic exams and can often distract from the original clinical intent, leaving both the radiologist and referring clinician unsure of the next appropriate step in management. Whereas most screening tests only include information about one particular organ system (e.g., mammography: breast; prostate-specific antigen [PSA]: prostate; colonoscopy: colon), lung cancer screening (LCS) with low-dose computed tomography (LDCT) provides a great deal of information about other organ systems outside the realm of possible lung cancer. Erb et al reported the rate of non-nodule incidental findings was as high as 67% in their series. The detection of incidental findings, however, should not necessarily be regarded as a downside of LCS CT. Besides assessing for the presence or absence of lung nodules, LDCT screening is an opportunity to screen for other conditions that can adversely impact a given individual’s health including but not limited to chronic obstructive pulmonary disease (COPD), coronary artery disease, and osteoporosis. This may in fact further increase its cost-effectiveness and provide better global outcomes. The interpreting radiologist and referring physician must be prepared to recognize, address, and know how to handle the additional “incidental” information afforded by LCS. To further complicate matters, LCS also often reveals challenging lung abnormalities specific to the current and former smoking population that can mimic more ominous diseases, including potential lung cancer. The characterization of other nonpulmonary lesions (e.g., thyroid, adrenal, etc.) may also prove difficult on LDCT due to quantum mottle, excessive noise, and lack of intravenous iodinated contrast media. This chapter will focus on some of the more commonly encountered incidental pulmonary and nonpulmonary findings encountered on lung cancer screens, highlighting typical CT imaging findings as well as potential clinical implications.
7.2 Incidental Pulmonary Findings: Smoking-Related Lung Diseases
7.2.1 Chronic Obstructive Lung Disease
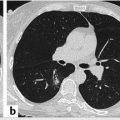
COPD, including chronic bronchitis and emphysema, is a major cause of morbidity and mortality in LCS populations and the fourth leading cause of death. Twenty percent of smokers develop COPD, so it is vital we identify those screened individuals at increased risk of such and quantify or at least qualify the extent of emphysema. The low-dose technique of LCS CT is designed to depict high-attenuation structures such as lung nodules, not low-attenuation process such as emphysematous lung. Additionally, the iterative reconstruction algorithms employed to reduce radiation dose may also “hamper” the depiction of emphysematous lung. Therefore, accurate quantification of emphysema may be somewhat limited on these studies. However, qualitative assessment, especially of moderate and extensive emphysema is possible and may be used as a potential risk stratification tool. At our institution, we visually score the extent of centrilobular emphysema and rate it on a scale of 0 to 3: 0 (none)—no centrilobular emphysema appreciated; 1 (mild)—centrilobular emphysema involving less than 25% of the visualized lung parenchyma; 2 (moderate)—centrilobular emphysema involving greater than 25% but less than 50% of the visualized lung parenchyma; and 3 (severe)—centrilobular emphysema involving greater than 50% of the visualized lung parenchyma. Although not mandated, we chose to report emphysema scores greater than 1 as significant findings (“S” modifier) in addition to our Lung CT Screening Reporting and Data System (Lung-RADS) score (▶ Table 7.1). Other institutions may opt to report the degree of emphysema in other manners. Some representative case examples applying this qualitative emphysema scoring system are illustrated in ▶ Fig. 7.1 , ▶ Fig. 7.2 , ▶ Fig. 7.3.



Extent of emphysema | Score |
No visualized emphysema | 0 |
Emphysema involving < 25% of the visualized lung parenchyma | 1 |
Emphysema involving > 25%, but < 50% of the visualized lung parenchyma | 2 |
Emphysema involving > 50% of the visualized lung parenchyma | 3 |
In addition to emphysema, several additional lung parenchymal abnormalities directly result from tobacco abuse. Examples include pulmonary Langerhans’ cell histiocytosis (PLCH) and the spectrum of interstitial pneumonias ranging from respiratory bronchiolitis-interstitial lung disease (RB-ILD) to desquamative interstitial pneumonia (DIP). Acute eosinophilic pneumonia (AEP) has also been linked to cigarette smoking in some studies, although smoking is likely only one of numerous causes and not a definitive causative agent alone. The basis for all smoking-related lung diseases is predominately an inflammatory response. The inflammation is caused by the numerous noxious antigens found in cigarette smoke followed by the body’s attempt at repairing the resultant damage. Cigarette smoke produces a powerful inflammatory response stimulating an increased influx of macrophages and other anti-inflammatory cells into the lungs. This ultimately results in destruction of lung tissue, followed by attempted reparation through the deposition of collagen and fibrous tissue. The latter is directly proportional to the amount of cigarette smoking incurred by the individual. Eventually, with continued smoking, irreversible fibrosis occurs in the alveolar walls, causing the syndrome now known as combined pulmonary fibrosis and emphysema. The latter is often complicated by pulmonary hypertension. These smoking-related lung diseases have typical CT imaging features the radiologist can apply to establish the appropriate diagnosis. Many of the histopathologic and imaging findings associated with these various smoking-related diseases improve with smoking cessation and abstinence.
7.2.2 Pulmonary Langerhans’ Cell Histiocytosis
PLCH is a rare smoking-related disorder typically seen in young adults in the third to fourth decade of life. Symptomatic patients may present with dyspnea, nonproductive cough, and fatigue. Secondary spontaneous pneumothorax occurs in about 15% of individuals. A small percentage of individuals are entirely asymptomatic and diagnosed solely because of incidentally detected imaging findings. Histologically, PLCH is characterized by granulomatous infiltration of the distal bronchioles. Although the diagnosis may be suggested with high accuracy on CT, the evolving nature and wide variety of imaging findings associated with PLCH can sometimes prove challenging. When imaging is inconclusive, tissue biopsy may be necessary for a definitive diagnosis.
As with most smoking-related lung disorders, the mid and upper lung zones are predominately affected in PLCH. Early in the disease process, the most common imaging manifestation is ill-defined micronodularity in a peribronchial and centrilobular distribution. Typically, the nodules measure less than 1.0 cm in size and some may have subtle surrounding ground glass or central cavitation. As active inflammation decreases, the nodular pattern dissipates and is replaced by more thick-walled cysts. These cysts gradually lose wall thickness, increase in size, coalesce, and become more bizarre and irregular in shape and morphology as the disease progresses. End-stage fibrosis is a rare long-term complication. Remember PLCH is an evolving disease and as such a constellation of these imaging findings may coexist in any given patient. When CT reveals both nodules and bizarre shaped cysts in a young smoker, the diagnosis is highly suggested. In the early nodular phase, PLCH may be indistinguishable from RB or endobronchial spread of infection. In the cystic phase, the differential diagnosis might include Pneumocystis pneumonia or LAM (lymphangioleiomyomatosis), depending on the clinical scenario. Serial imaging exams and appropriate history are invaluable in establishing the correct diagnosis. More than 50% of PLCH patients experience complete spontaneous resolution or stabilization without treatment; even better results are reported with smoking cessation. The latter is the recommended first line of management for PLCH. Corticosteroids may be of some benefit in the early stages of the disease. In a minority of cases, the disease progresses to end-stage fibrosis, at which time lung transplantation may be the only viable option.
7.2.3 Respiratory Bronchiolitis (Also Known as “Smoker’s Bronchiolitis”)
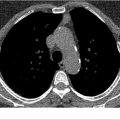
RB is histopathologically characterized by an accumulation of pigmented macrophages and inflammation without fibrosis involving the respiratory bronchioles. It affects nearly all cigarette smokers. Interestingly, RB is typically asymptomatic and only discovered incidentally on imaging, if at all. Rarely, heavier smokers, with more than 30 pack-years of abuse, may develop symptomatic RB-ILD. Individuals with RB-ILD are usually young and complain of chronic cough and progressive dyspnea over 1 to 2 years. Bibasilar, end-inspiratory crackles or crackles throughout inspiration and sometimes into early expiration may be auscultated. RB-ILD is occasionally diagnosed in former smokers and in persons exposed to secondhand smoke or fumes. Pulmonary function tests (PFTs) may be normal but alternatively may reveal an increased residual volume, mixed obstructive-restrictive pattern, mild hypoxemia, and mild to moderate reduction in the diffusion capacity for carbon monoxide (DLCO). On CT, the imaging findings of RB and RB-ILD are inseparable. In most instances, CT will be normal. In others, CT may demonstrate poorly defined centrilobular nodules (71%) and ground-glass opacities (67%; ▶ Fig. 7.4). Although more profuse in the upper lung zones (53%), the mid and lower lung zones may be affected (20%). Bronchial wall thickening is seen in nearly all persons with RB-ILD (90%). Most persons also have concomitant mild centrilobular emphysema (57%). Air trapping is not uncommon on expiratory images. Fibrotic changes manifest by intralobular linear opacities and honeycombing is only seen in about 25% of persons diagnosed with RB-ILD. RB-ILD may coexist with clinical and imaging findings of DIP, another smoking-related disease, discussed later in this chapter. Other potential diseases that may have a similar CT imaging appearance include subacute hypersensitivity pneumonitis, nonspecific interstitial pneumonitis (NSIP), and early PLCH. First-line treatment of RB-ILD entails smoking cessation, which typically results in either improvement or stabilization of the symptoms. RB-ILD does not typically progress to end-stage pulmonary fibrosis in most persons.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree