9 Venous Access
Summary
Central venous access procedures are frequently performed by interventional radiologists, and are most commonly considered straightforward. However, knowledge of the nuances of placement, management, and troubleshooting of central venous catheters is necessary for an effective and successful venous access practice. In particular, management of patients with limited access options is nearly completely within the purview of interventional radiology. This chapter will review guidelines relevant to the management of central venous access, and will present techniques for achieving access in patients with venous occlusions.
9.1 Introduction
Central venous access is a cornerstone procedure in the modern interventional radiology (IR) practice. Technical mastery of routine catheter placement is straightforward. The expertise provided by the interventional radiologist in both basic and advanced catheter management helps patients and referring physicians navigate the complexities of long-term central venous access such as optimizing catheter choice, managing catheter-related complications, and achieving optimal venous access. The aim of this chapter is to provide a construct for managing central venous access using evidence-based strategies and state-of-the-art techniques.
9.2 Case Vignette
9.2.1 Patient Presentation
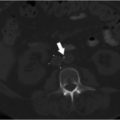
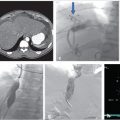
A 54-year-old woman with a history of recurrent pancreatic cancer status post Whipple procedure presented 2 months after placement of a left subclavian vein dual-lumen port with neck, facial, and bilateral upper extremity edema. Computed axial tomography (CT) of the chest was performed with contrast, revealing thrombus within the superior vena cava (SVC) surrounding the catheter tip (Fig. 9.1). Incidental note was made of small pulmonary emboli, from which she was asymptomatic. The patient was treated with a heparin drip as a bridge to oral anticoagulation. Facial and neck edema resolved completely, and at discharge there was minimal symmetric bilateral upper extremity edema.
A few months later the patient was referred to the IR department for evaluation of a malfunctioning port, with documentation of unsuccessful aspiration through both lumens and resistance to injection resulting in prolonged infusion times.
9.2.2 Physical Exam
Vital signs were within normal limits and the patient was in no acute distress. There was trace residual pitting edema noted in the bilateral forearms, right slightly greater than left. Small anterior chest wall collateral venous structures were noted. No head and neck swelling was present, her neck exhibited normal range of motion, and there was no respiratory distress suggesting airway compromise. Mallampati score was 2. Positive S1/S2 heart sounds were auscultated with normal cardiac rate and rhythm. Lungs were clear bilaterally.
9.2.3 Noninvasive Testing Imaging
At the initial presentation at the time of acute symptoms, the patient had bilateral upper extremity duplex ultrasound interrogation revealing thrombus in her bilateral axillary and subclavian veins. A chest CT performed at the same time demonstrated thrombus throughout the SVC as well.
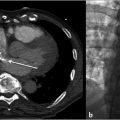
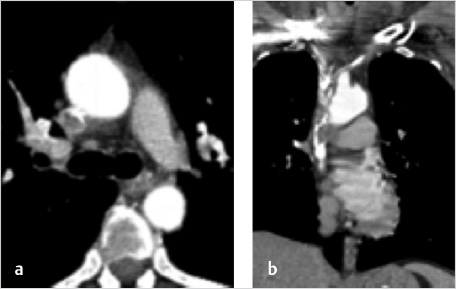
For evaluation of the etiology of port dysfunction, a venogram was performed via port access. This revealed restriction of contrast flow from the tip of the catheter, indicative of a fibrin sheath (Fig. 9.2), with palpable resistance to injection of contrast through both lumens.
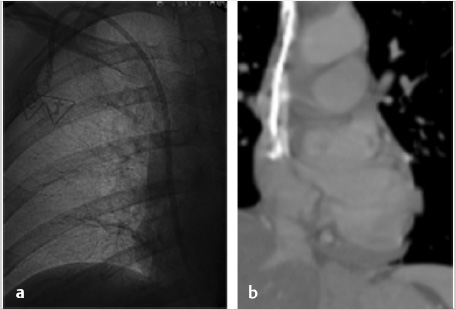
A follow-up chest CT was obtained prior to intervention for procedural planning. This demonstrated narrowing of the SVC with resolution of the previously visualized thrombus. In addition, asymmetric right chest wall venous collateral channels had formed (Fig. 9.3).
9.2.4 Specifics of Consent
The spectrum of options were discussed, including attempted snaring/stripping of the original access versus complete removal of the left subclavian port, with new port placement through right internal jugular vein access, and techniques for managing SVC stenosis. Standard procedural risks were discussed, including those relative to moderate sedation, risk of bleeding, pneumothorax, and infection. In addition, the likely need for SVC venoplasty and attendant possibility of stent placement were reviewed, including risk of SVC perforation and hemopericardium from SVC balloon dilatation, leading to surgical intervention or death. The patient elected for attempted stripping of her preexisting port with subsequent new port placement if this was unsuccessful, and consented to balloon dilation with possible stent placement in the SVC.
9.2.5 Details of Procedure
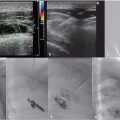
After obtaining informed consent, the patient was placed on the fluoroscopy table in supine position. Patency of the right common femoral vein was confirmed with a brief ultrasound exam. The right groin was prepped and draped in the usual sterile fashion. The skin and subcutaneous soft tissues were anesthetized using 1% lidocaine. A small incision was made with a #11 scalpel. Access to the right common femoral vein was obtained with ultrasound guidance using micropuncture technique and a 25-cm 6-French vascular sheath was positioned with its tip in the inferior vena cava (IVC). The sidearm of the sheath was flushed and connected to an infusion of heparinized saline solution.
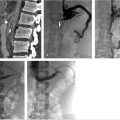
Over a 0.035-inch, 180-cm-length Glidewire Advantage guidewire (Terumo Medical Corporation, Somerset, NJ), a 4-French 100-cm hydrophilic angled catheter was advanced across the SVC stenosis into the right brachiocephalic vein and venography was performed (Fig. 9.4a). There was retrograde flow of contrast from the right brachiocephalic vein into the azygous vein without opacification of the SVC or right atrium.
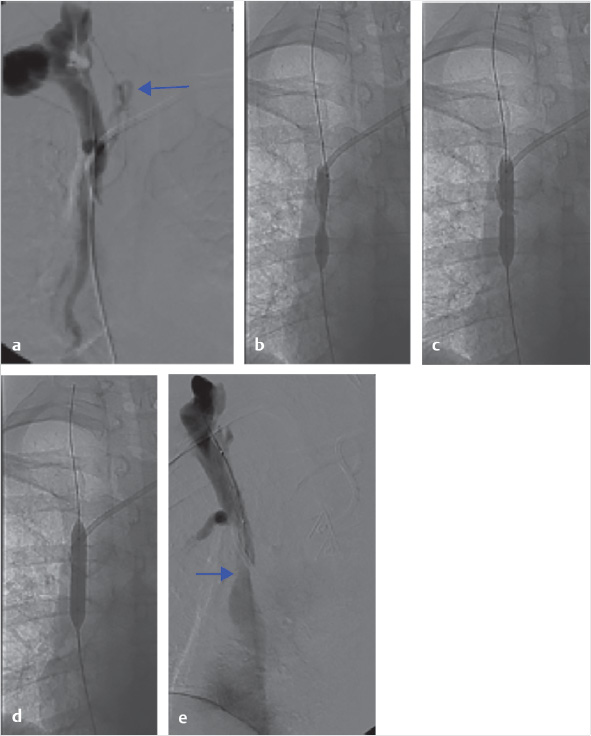
The diagnostic catheter was exchanged for an 8 mm by 4 cm Ultraverse balloon (BARD Peripheral Vascular, Tempe, AZ). Balloon dilation of the SVC was performed, with a significant waist identified at the level of the catheter tip (Fig. 9.4b; Fig. 9.4c; Fig. 9.4d). Subsequently, a 10 mm by 4 cm balloon was easily inflated at the same level without residual waist or patient discomfort. Final central venography confirmed restored antegrade flow of contrast from the right brachiocephalic vein through the SVC (Fig. 9.4e). Due to presentation with port dysfunction without symptoms of SVC syndrome, stenting of the SVC was not pursued.
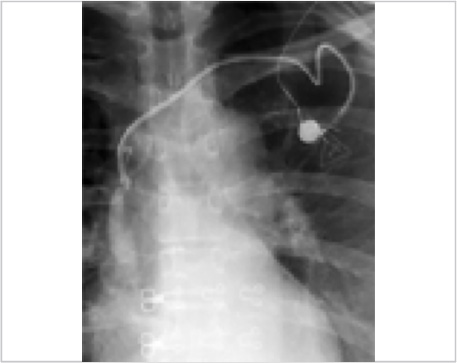
Due to persistent adherence of the catheter to the vein wall/fibrin sheath, with inability to snare the catheter to strip the sheath, decision was made to place a new catheter in the reestablished lumen. A new 9.5-French dual-lumen port was placed without difficulty via right internal jugular vein access (Fig. 9.5), with removal of the left subclavian port. The catheter tip was positioned significantly deeper than the original port, with the tip terminating at the junction of the upper and mid-thirds of the right atrium. The patient’s cardiac rhythm was observed for several minutes to ensure no evidence of ectopy incited by the catheter. The patient was discharged home the same day.
9.2.6 Follow-up
The new port served the patient well up to 6 months after the intervention. Unfortunately, the patient’s metastatic disease burden continued to progress despite therapy. She declined further therapy and pursued hospice care.
9.3 Epidemiology and Scope of the Problem
The spectrum of disease processes treated for which long-term central venous catheter access is required is vast, including but not limited to end-stage renal disease, cancer, infection, malnutrition, and pulmonary hypertension. Any patient requiring chronic, recurrent peripheral infusion therapy may also require central venous catheter access as peripheral veins thrombose.
Over 5 million central venous catheters are placed in the United States each year. 1 , 2 From 1992 to 2011, placement of long-term central venous access devices in the Medicare patient population increased by an astounding 303% nationally, from 76,444 to 307,838. 3 Per the U.S. Renal Data System, 80.2% of patients with end-stage renal disease were using dialysis catheters for initiation of hemodialysis by the end of 2013, translating to approximately 376,900 patients. 4
As the clinical indications for and number of patients requiring central venous access increase, the demand for expertise in those managing venous access also increases. The interventional radiologist is in a unique position as both procedural and clinical content expert in the entire range of central venous access challenges and considerations. While many medical providers are able to place central venous catheters into a patent subclavian, internal jugular, or basilic vein, it is incumbent upon the interventional radiologist to uphold National Kidney Foundation Kidney Disease Outcomes Quality Initiative (NKF KDOQI) guidelines (www.kidney.org) for correct vein choice and preserve arm veins for possible future dialysis fistula/graft creation. Any clinician can remove a catheter in a bacteremic patient; the interventional radiologist should remind the clinical team of current Centers for Disease Control (CDC) guidelines and provide strategies that potentially preserve access for these patients. Any clinician can manage a patient with a stenosed or occluded SVC by placement of a femoral catheter; the interventional radiologist can reconstruct, preserve, or obtain central venous access through a wide variety of techniques.
As the number and duration of long-term central venous catheters increases, managing complex vascular access will become a greater need. The interventional radiologist, as a steward of central venous access, should work to ensure excellent outcomes for patients from initial catheter placement and beyond.
9.3.1 Patient Presentation and Evaluation
There are numerous factors to be considered in the evaluation of a patient for central venous access, and the evaluation should allow selection of the most appropriate catheter for the patient’s needs. In simplest terms, the interventional radiologist needs to determine what the catheter will be used for, how many lumens are necessary, how long the catheter will dwell within the patient, and how frequently it will be accessed. It is unreasonable to expect the referring provider to understand the nuances associated with the current breadth of central venous catheter choices, and the interventional radiologist should be prepared to make recommendations regarding appropriate access choice, rather than simply provide a technical service. A cursory understanding of how the catheter will be used will rapidly narrow down the choices for the correct catheter. Table 9.1 provides a basic framework for catheter selection and procedural planning.
9.3.2 Duration of Access Requirement
Short-term access can generally be achieved with a peripheral IV (PIV). PIVs are appropriate for nonsclerosant and nonvesicant agents that need to be administered intravenously for short durations. The most common indication for a PIV would be for intravenous crystalloid infusions during hospitalization or for rapid resuscitation. Saline is isotonic to the blood and can be rapidly infused in a peripheral vein with few adverse consequences. Many hospitals require that PIVs be changed every few days to avoid thrombophlebitis or local infection. With ever shortening lengths of stay, most patients will likely only need one PIV during a hospitalization. For this reason, the CDC recommends peripherally inserted central catheters (PICCs) and midline catheters only be placed when the duration of IV access will be more than 6 days. 5
When access is needed for a longer duration, for example, 6 weeks of IV antibiotics for treatment of osteomyelitis, a PICC is a suitable peripherally inserted line that can be maintained for a long duration with limited risk of infection. When treatment is needed for several months or longer, then a tunneled catheter may be better than a PICC because the use of a tunneled cuffed catheter can further reduce the risk of infection. Finally, ports represent long-term access devices that are appropriate when accessed only intermittently.
9.3.3 Sclerosants and Vesicants
Sclerosants and vesicants are drugs that have a direct caustic effect on the endothelium, related to the pH and the osmolality of the medication. Vesicants are defined as drugs that can cause tissue destruction or skin blistering, while irritants are any drug with a pH of less than 5 or greater than 9. Examples of drugs and infusions that are vesicants are 10% dextrose, dopamine, midazolam, lorazepam, nitroprusside, potassium chloride in > 40 mEq, total parenteral nutrition (TPN), and vancomycin. Irritants include drugs such as ciprofloxacin, ceftriaxone, fluconazole, morphine, octreotide, and propofol.
It is very important to restrict the use of vesicants and irritants to central veins where adequate mixing can occur. This is important for the prevention of thrombophlebitis and venous occlusion. It has been demonstrated that extremes of pH and osmolarity increase venous thrombosis rate. 6 , 7
9.3.4 Lumens Required
The more lumens a catheter has, the larger the device and the more manipulation it will undergo. This leads to an increase in the risk of infection and venous thrombosis. Ideally, when requested to provide venous access, systems should be in place to ensure that each lumen of the access device is justified on the basis of either continuous simultaneous infusion or incompatibility of medications. When there are more lumens, each will be frequently manipulated during changes of medications and the risk of infection goes up. For this reason, CDC guidelines recommend limiting the number of lumens to the minimum needed for infusions. 8
Another consideration with respect to lumens is that the size of the catheter increases with increasing number of lumens. As a consequence, in a study of triple-lumen PICCs, the trial was terminated early due to an unacceptable thrombosis rate associated with three-lumen 7-French PICC lines. 8
9.3.5 Frequency of Use: Intermittent or Continuous Access
The frequency of administration is a factor when considering placing a port. In general, the advantage of a port over an externalized tunneled catheter is a lower rate of infection because the port does not need to be continuously accessed. For example, typical chemotherapy regimens involve several days of chemotherapy every 3 to 4 weeks. In this situation, the port makes sense because between treatments the port is completely subcutaneous and at very low risk of infection. However, if a port were used for TPN, daily access would raise the infection rate to one approaching that of a tunneled catheter. The consequences of an infected port are greater, in that an infected port would require an incision and dissection to remove the device. For this reason, daily infusions or continuous infusions should be given through tunneled catheters rather than ports.
9.3.6 Flow Rates
Flow rates should be considered for specific situations such as dialysis and plasmapheresis. Flow rates are directly proportionate to catheter inner diameter (ID), and inversely proportionate to catheter length. Therefore, for volume resuscitation, short large catheters are best, whether peripheral or central, and resuscitation through a PICC should be avoided. In general, the highest flow rates are required for dialysis treatments (generally between 400 and 500 mL/min), and for this purpose a large ID is desired. It is reasonable to attempt to place the smallest outer diameter (OD) catheter that will accomplish necessary flow rates, due to the higher risk of vascular thrombosis or stenosis with larger catheters. Minimizing catheter length may allow for placement of a smaller diameter catheter for improved long-term outcomes. Plasmapheresis is generally run at flow rates of 250 mL/min and some of the older dialysis catheters designs can achieve this with a slightly smaller profile than current dialysis catheters (12.5 vs. 14.5 French). The 9.6-French Hickman catheter (Bard Peripheral Vascular, Tempe, AZ), which is 90 cm in length and has a 1.6-mm ID, can sustain a flow rate of 80 mL/min. This is typically adequate for most infusion therapy.
9.4 Considerations for Specific Indications
9.4.1 Hemodialysis
Understanding the pathogenesis of the patient’s renal insufficiency and close communication with the nephrology service is vital. In cases of acute renal insufficiency, determine whether a temporary, nontunneled catheter will suffice until there has been appropriate treatment and recovery of the patient’s native renal function. If more long-term or permanent dialysis needs are anticipated, determine where along the arc of decision making the patient may be for upper extremity fistula/graft creation allowing avoidance of ipsilateral catheter placement if possible.
9.4.2 Infection
For most patients, a PICC will suffice for long-term intravenous antibiotic therapy. A key consideration is the patient’s underlying renal function. The AV Fistula First Breakthrough Initiative National Coalition recommends against using PICCs in patients at risk for or with known midstage 3 chronic kidney disease or higher renal disease. In this instance, it is prudent to use a low-profile tunneled central venous catheter placed via an internal jugular vein approach, rather than peripherally inserting a catheter, due to the long-term consequences of endothelial damage in this patient population. While many larger health systems have protocols in place to prevent PICC placement in patients with renal insufficiency, this practice is by no means universal. The interventional radiologist can be a key advocate in assuring hospital compliance with this initiative.
An additional consideration in assessing for PICC versus tunneled catheter placement is a patient’s mentation. If the patient has an altered mental status potentially placing the PICC at risk of accidental removal, tunneled access may be more suitable and secure. For a patient who may be aggressive enough to remove tunneled catheters, these may be tunneled across the supraclavicular fossa, superficial to the trapezius musculature, to make the catheter harder to reach by the patient.
9.4.3 Total Parenteral Nutrition
TPN is a broad title given to any intravenous supplementation used to bypass the gastrointestinal tract. The initial decision regarding catheter type again comes down to duration, determining whether a PICC is adequate or if a tunneled catheter will be needed. There are a variety of tunneled catheters that differ in their OD, number of lumens, and ID. The type of TPN solution the patient will receive will dictate the caliber of lumen needed. The TPN lumen should only be used for TPN. If there is anticipation for intravenous medication or frequent blood draws, this will require at least one additional lumen.
A complete review of the various catheters and their properties would exhaust the space in this text. Review of the vendor catalogues which provide details of the different catheter characteristics is a useful endeavor to establish a basic knowledge of the pros and cons of different devices for different uses. For example, there are significant differences between a 10-French dual-lumen Leonard catheter (Bard Peripheral Vascular, Tempe, AZ) and a 9-French dual-lumen Hickman catheter. The Leonard catheter lumens are equal in ID (1.3 mm) as compared to the Hickman catheter, with a 1.3-mm red lumen ID and a 0.7-mm white lumen ID. Careful consideration as to the viscosity of the solution for which each lumen is utilized will help optimize infusion rates, minimize catheter size, and decrease possibility of luminal occlusion. An active dialogue with the TPN service can help determine which catheters meet the needs of the majority of the patient population. Maintaining and regularly reviewing practice level quality data, including catheter-related infections and frequency of need for catheter repair/exchange, can help guide quality improvement efforts to optimize patient care in this arena.
9.4.4 Chemotherapy
The most common catheter choice for administration of chemotherapy is the subcutaneous port. Choice between single- and dual-lumen port catheter placement should be predicated upon the need for simultaneous infusion. Single-lumen catheters will suffice for the majority of cancer patients and are anecdotally better tolerated by patients due to smaller profile of the reservoir. Reservoirs come in a variety of compositions from various manufactures and will not be comprehensively reviewed here.
Determining a patient preference for side of placement may help improve the patient’s functionality and quality of life. One patient disclosed that he preferred a left-sided chest port because he was an avid rifle shooter and was concerned how the butt of the rifle would sit against his chest with the subcutaneous reservoir in place. Furthermore, he raised the question about the kick of the gun resulting in disconnection between the catheter and reservoir. Therefore, a left-sided port was placed. Though a regionally common example, this example underlines the importance of the consideration of patient needs, to facilitate improved quality of life.
9.4.5 Previous Vascular Access
When replacement or repeat vascular access is needed, knowledge regarding the circumstances of the prior access is necessary: what, where, when, and why? The answers to these questions should be known before the patient arrives in the IR suite.
What type of catheter did the patient previously have? Something as simple as prior central line placement may be pertinent if there was a documented complication or difficulty at that time. Prior tunneled dialysis catheter placement or prior malpositioned port introduces the possibility of access vein occlusion and central venous stenosis.
Location of the prior catheter is an important consideration. Multiple prior right internal jugular vein tunneled dialysis catheter placements will decrease the probability of right internal jugular vein patency and should prompt a thorough ultrasound assessment prior to the patient being prepped and draped.
When and why the prior access was removed are important questions to answer. If the patient’s most recent port or tunneled dialysis catheter was removed for infection, has the patient been appropriately treated? Do they meet criteria for new implanted/tunneled catheter placement or should a temporary catheter be placed until they do? Management of this scenario will be discussed in detail below.
9.5 Technical Tips and Tricks
9.5.1 Basics of Procedure
Most interventional radiologists have a routine, automatic approach to the placement of the uncomplicated central venous access. There is an advantage to establishing a standard approach, the fundamentals of which are outlined here. High variability exists in the approach used and the order of completion of the steps to catheter placement.
Ultrasound the anticipated target vein prior to scrubbing and ensure that the ultrasound settings are optimized to see the vein best.
Ensure that a complete barrier scrub is performed to minimize the risk of infection from placement. For physician performance measures, standard documentation of the procedure should include the adherence to “CLIP” (central line insertion practices) bundle criteria mandated by the CDC: chlorhexidine scrub, prep dry prior to insertion, five maximal sterile barriers used, including gloves, gown, cap, mask, and large sterile drape covering patient’s entire body.
Puncture the vein with direct visualization with ultrasound, directly observing the needle tip puncture the wall of the vein.
Advance a wire into the vein. Use of a micropuncture system may reduce the risk of air embolus.
Make the tunnel +/– port pocket.
Exchange the access catheter for the peel-away sheath.
Quickly exchange the wire for the catheter to avoid air embolus.
Perform fluoroscopy to ensure the tip is appropriately positioned before removing the peel-away sheath.
Remove the peel-away sheath and adjust the catheter to optimize placement of the tip near the junction of the SVC and the right atrium.
Obtain an image of the catheter to document appropriate location. Note that this will be supine and that the tip position is likely to change when the patient is moved form a supine to an upright position.
9.5.2 Access Dilation
Passage of dilators and the peel-away sheath reflects one of the most critical moments of central venous access. Kinking of the guidewire causes a sharp point at the bend of the wire, which can penetrate the SVC/right atrium when aggressively advanced as a unit. There are a few technical maneuvers that can significantly reduce the risk of wire kinking and thus reduce the risk of vascular trauma. One technique is to slightly withdraw the wire during advancement of the dilators and peel-away sheath ensuring a smooth coaxial interface between the wire and dilator/sheath. Immediately upon encountering resistance, advancement of the dilator/sheath should stop, and the trajectory of the dilator/sheath evaluated under fluoroscopy. Changing the plane of the dilator/sheath to one more parallel with the patient/vessel will usually allow advancement. Difficult anatomy may require a wire upgrade from that which comes with the catheter kit. In tough cases, a longer wire placed deep into the IVC will ensure enough purchase to perform the withdraw technique while not losing access.
In the event of persistent resistance to dilation, increased guidewire stiffness is helpful. For patients that have a history of multiple prior catheters, anticipate encountering scar tissue and select an appropriately stiff wire for dilation. Another useful approach is the addition of intermediate dilator sizes. Most tunneled dialysis catheter kits come with two dilators and the peel-away sheath. A few intermediary dilatations in addition to what is provided in the kit may facilitate peel-away sheath passage.
9.5.3 Left-Sided Access
Left-sided central venous access is inherently more challenging due to two natural curves the catheter must traverse before taking its final position: (1) curve at the junction of the left internal jugular vein (LIJV) with the left subclavian vein (LSCV); (2) curve at the junction of the left brachiocephalic vein junction with the right, forming the SVC. Therefore, catheter length from left-sided access must take into account both the increased distance of LIJV access compared to right internal jugular vein, and the length “eaten up” by the aforementioned curves as well. On initial placement, the catheter will tend to be stiffer, somewhat straightening the anatomic curves. As the catheter warms to body temperature and becomes more compliant, it will tend to conform more to the curvatures and foreshorten from its original position.
Foreshortening of left-sided access is particularly problematic for several reasons. From a simple malposition perspective, a tunneled dialysis catheter that was in the right atrium upon implantation may not function adequately if the tip is reduced back to the SVC. A unique malposition challenge from left-sided access is the tip terminating in the azygos vein, either acutely or upon delayed foreshortening/repositioning. In the acute setting, this is easily remedied by retracting and repositioning the catheter. If there is any doubt about stability of position, it is reasonable to exchange for a longer catheter in the acute setting to prevent the delayed grief.
9.5.4 The Kinked Peel-Away Sheath
This challenge tends to go hand in hand with left-sided internal jugular vein access as the inherent curves, described above, exert force upon the sheath as the internal dilator is removed. The kink can make it difficult or impossible to advance the catheter. Kinks at the skin level are easily remedied by pinching out the kink or peeling down to/beyond the kink. Internal kinks are more challenging. One initial approach to remedying a kink is to replace the inner dilator to relieve the kink and either advance or slightly retract the sheath/dilator system and reattempt catheter passage. Sometimes the kink will return immediately upon removing the internal dilator. Placing a stiff wire through the sheath, over which to advance the catheter, or placement of a stiff wire into the catheter to give the catheter more body for pushability may allow passage. If still unsuccessful, consider exchange for a new peel-away sheath.
9.6 Potential Complications and Pitfalls
9.6.1 Acute and Intraprocedural Complications
Pneumothorax: The overall pneumothorax rate associated with central venous access placement ranges from 1 to 6% in the literature, with well-established elements that increase the risk including catheters placed in emergent situations, subclavian vein access, increased number of passes, and larger bore catheters. 9 These data do not discriminate between attempts under ultrasound guidance versus anatomical landmarks. Ultrasound guidance has been proven to prevent the number of mechanical complications. 10 , 11 With impeccable ultrasound-guided technique where there is constant visualization of the needle approaching the jugular vein, the risk of pneumothorax likely approaches 0%.
There are some patients that are inherently more challenging in terms of optimizing visualization of the internal jugular vein. The standard trifecta is the obese tracheostomy patient with a short neck. It may only be possible to get a portion of the ultrasound footprint in a useful position for guidance. If the needle is not aligned perfectly with the probe, there is a chance of overreaching the target and creating a pneumothorax. Use help from all members of the team, such as requesting assistance from the respiratory therapist in turning the patient’s head and stabilizing the airway. The other consideration is to allow a slightly higher access above the clavicle than usual for better visualization. Both of these adjustments will decrease the likelihood of complication.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree