Adrenal Glands and Kidneys
William E. Brant
Adrenal Glands
Imaging Methods
The current major challenge of adrenal imaging is to provide noninvasive characterization of the many adrenal nodules found incidentally on CT or MR performed for other purposes (1,2,3,4). Up to 5% of patients who undergo MDCT of the abdomen will have an incidental adrenal lesion, an “incidentaloma.” The predominant consideration is to determine if the lesion is a benign nonfunctioning adrenal adenoma or is it a metastasis. Within the differential diagnosis are subclinical pheochromocytomas, or functioning cortical adenomas, causing unrecognized hyperaldosteronism or Cushing syndrome. Additional considerations include myelolipoma, adrenal carcinoma, hemorrhage, cyst, neuroblastoma, and ganglioneuroma. The adrenal glands are routinely imaged in patients with known malignancy, especially lung cancer, in order to detect metastatic disease. In patient with adrenal endocrine syndromes diagnosed clinically, imaging is used to find and characterize the causative lesion. MDCT remains the imaging modality of choice, whereas MR, PET, PET-CT, US, scintigraphy, adrenal vein sampling, and image-guided adrenal biopsy all have significant roles (5).
Anatomy
The adrenal glands are composed of an outer cortex and an inner medulla that are functionally independent and distinct. The cortex secretes steroid hormones including cortisol, aldosterone, androgens, and estrogens. The medulla produces catecholamines.
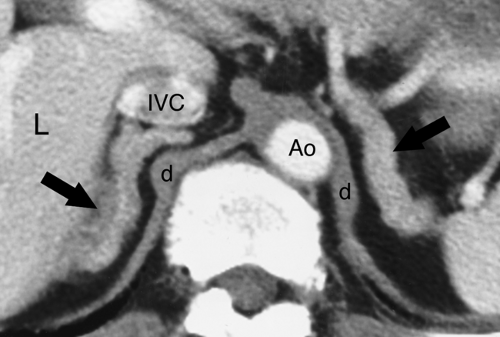
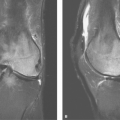
The adrenal glands lie within the perirenal space surrounded by fat. The right adrenal gland is located posterior to the inferior vena cava (IVC) at the level where the IVC enters the liver. The right adrenal gland is between the right lobe of the liver and the right crus of the diaphragm just above the upper pole of the right kidney. The left adrenal gland lies just medial and anterior to the upper pole of the left kidney, posterior to the pancreas and splenic vessels, and lateral to the left crus of the diaphragm. On cross-sectional imaging, the adrenal glands appear triangular, linear, or inverted V- or Y-shaped (Fig. 32.1). Each limb is smooth in outline and uniform in thickness with straight or concave borders. The limbs are 4 to 5 cm in length and 5 to 7 mm in thickness. The adrenal glands are of uniform soft-tissue density on CT and US. On MR, the normal adrenal is hypointense, about equal to striated muscle, on T1WI. On T2WI, the adrenals are isointense or slightly hypointense compared with the liver and hypointense compared with the spleen (Fig. 32.1B). Chemical shift MR imaging is used to demonstrate intracellular fat in benign adrenal adenomas by utilizing in-phase (IP) and out-of-phase (OP) gradient-recalled sequences. Intracellular fat demonstrates a loss of signal on OP images compared to IP images because of signal cancellation effect resulting from fat and water occupying the same voxel. Fat saturation MR technique is used to demonstrate macroscopic fat seen in adrenal myelolipomas. Macroscopic fat shows a loss of signal intensity on fat saturation images compared to pulse sequences of the same technique without fat saturation.
Adjacent structures may cause problems in adrenal imaging by mimicking adrenal masses. Tortuous splenic vessels, splenic lobulations, pancreatic projections, exophytic upper pole renal masses, portosystemic venous collaterals, retroperitoneal adenopathy, gastric diverticulum, and portions of the stomach may all cause adrenal pseudotumors. Judicious use of oral and intravenous contrast on CT, or supplemental US or MR studies, will reveal the true nature of these conditions.
The Incidental Adrenal Mass
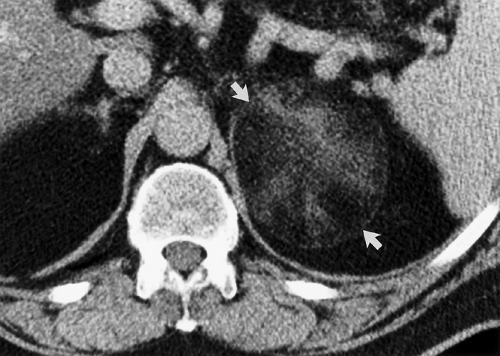
In patients without a known malignancy, most small (<4 cm) adrenal nodules are benign nonhyperfunctioning adrenal cortical adenomas (Fig. 32.2). Less than 3% of lesions are malignant (1). In patients with a known malignancy, the incidence of metastasis to the adrenal rises to 50%. These lesions are just as important to characterize in order to provide accurate
tumor staging. Steady advances in imaging allow noninvasive imaging characterization of the majority of these nodules. Table 32.1 provides a summary of the currently accepted criteria for characterization of adrenal lesions on CT, MR, and PET-CT.
tumor staging. Steady advances in imaging allow noninvasive imaging characterization of the majority of these nodules. Table 32.1 provides a summary of the currently accepted criteria for characterization of adrenal lesions on CT, MR, and PET-CT.
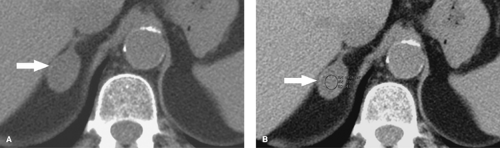
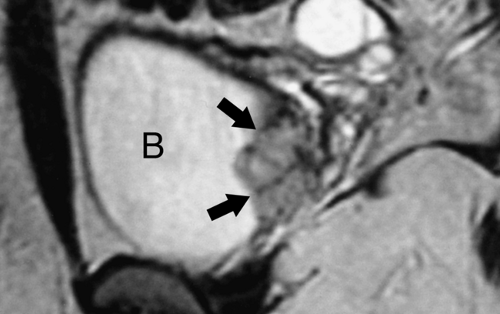
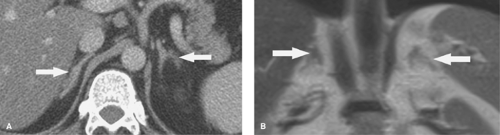 Figure 32.1. Normal Adrenal Glands. Contrast-enhanced CT image (A) and coronal T2-weighted MR image (B) show the normal appearance of the adrenal glands (arrows). |
Adrenal cortical adenomas are the most common adrenal mass found in 4% to 6% of the population and increasing in incidence with age. Most (94%) are nonhyperfunctioning, truly incidental, findings. Approximately 6% of adenomas secrete excess hormone and causes clinical or subclinical manifestations of one of the adrenal endocrine syndromes. Function of an adenoma cannot be determined by its imaging appearance but is assessed clinically. Cortical adenomas accumulate cholesterol, fatty acids, and other fatty substances, which serve as precursors of cortical hormones. Fat accumulation in 70% of adenomas is sufficient for them to be classified by imaging as lipid-rich adenomas. The remaining 30% are termed lipid-poor adenomas. Attenuation of adenomas on unenhanced CT has a range of -20 to 30 HU. Enhancement on postcontrast CT and MR is unpredictable and often heterogeneous. However, benign adenomas are characterized on MDCT by rapid washout of the contrast agent.
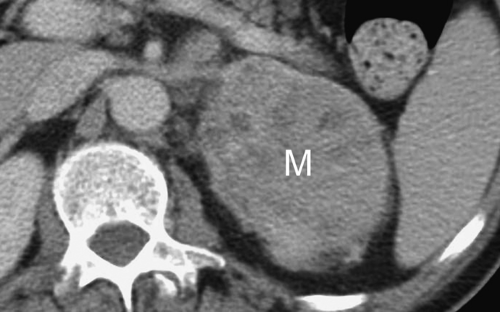
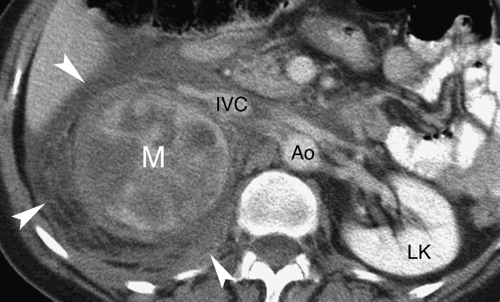
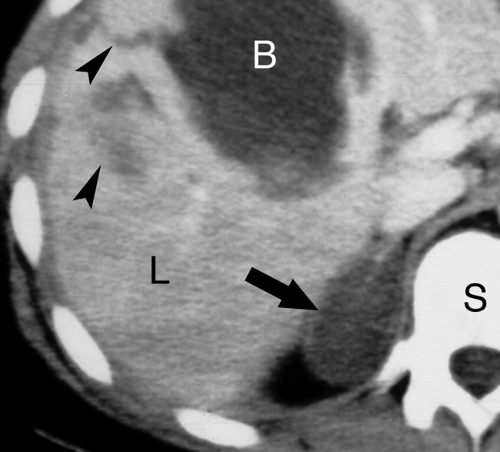
Adrenal metastases are exceedingly common, found in 27% of patients with malignant disease on autopsy series. The most common primary tumors are lung, breast, melanoma, gastrointestinal, thyroid, and renal. Small lesions (<4 cm) tend to be homogeneous, well defined, and difficult to distinguish from benign, nonfunctioning adenomas. To complicate the issue, even in patients with known primary malignancy, up to 50% of small adrenal masses are benign adenomas and not metastases. On CT and MR, larger lesions (>4 cm) generally show features characteristic of malignancy, including inhomogeneous density, irregular shape, thick irregular margination, internal hemorrhage or necrosis, and invasion of adjacent structures (Fig. 32.3). Small lesions show the malignant features listed in Table 32.1.
Stability over time is a well-recognized feature of benign adenoma. Detection of a small adrenal nodule should instigate a search for previous imaging studies. Absence of change in size and appearance of a small adrenal lesion for 6 months is generally accepted as evidence of benignancy (1). Stability for 1 to 2 years increases the confidence of a benign diagnosis. An increase in size of the lesion over 6 months is strong evidence of malignancy, though hemorrhage into a benign lesion causes an abrupt increase in size.
Clinical evaluation is recommended with increasing frequency as subclinical adrenal endocrine syndromes are recognized. Patients with hypertension should be evaluated for Cushing and Conn syndromes. Cushing syndrome is excluded with the absence of hypertension and obesity.
MDCT is the imaging modality of choice. CT without contrast successfully and accurately characterizes as benign the
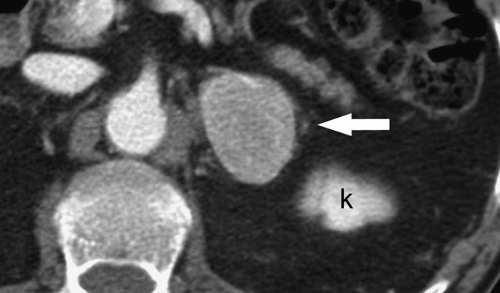
70% of patients with lipid-rich adenomas by demonstrating a CT attenuation of less than 10 H (6). Careful adherence to proper technique is essential when measuring CT densities (Fig. 32.2). Measurement is made on a thin section through the center of the lesion. The range-of-interest (ROI) cursor should cover at least half of the lesion surface area, avoiding areas of necrosis or hemorrhage. Measurements of attenuation below 10 H effectively exclude malignancy. Attenuation measurements greater than 10 H indicate indeterminate lesions that may be lipid-poor adenomas or malignancies. Unfortunately many CT examinations, on which incidental adrenal nodules are detected, are performed only after intravenous contrast administration. Perfusion differences between benign adenomas and metastasis provide a second reliable set of criteria for diagnosis (7,8). Adenomas are characterized by rapid washout of contrast agent, whereas metastasis show slow contrast washout (Figs. 32.4, 32.5). Percentage washout measurements are made on images taken at 60 to 75 seconds following the onset of intravenous contrast injection (enhanced attenuation). Delayed attenuation measurements are made on images obtained at 10 or 15 minutes following contrast injection. Absolute and relative percentage washout (RPW) calculations are made (Table 32.2). Benign lesions show greater than 60%
absolute percentage washout (APW) and greater than 40% relative percentage washout at 15 minutes. A number of studies have looked at calculating washout values from 10-minute and from 15-minute delay images. It seems to make little practical difference. The 60% APW and 40% RPW criteria are easier to remember and seem to work effectively using either 10- or 15-minute delayed images (1,9,10).
70% of patients with lipid-rich adenomas by demonstrating a CT attenuation of less than 10 H (6). Careful adherence to proper technique is essential when measuring CT densities (Fig. 32.2). Measurement is made on a thin section through the center of the lesion. The range-of-interest (ROI) cursor should cover at least half of the lesion surface area, avoiding areas of necrosis or hemorrhage. Measurements of attenuation below 10 H effectively exclude malignancy. Attenuation measurements greater than 10 H indicate indeterminate lesions that may be lipid-poor adenomas or malignancies. Unfortunately many CT examinations, on which incidental adrenal nodules are detected, are performed only after intravenous contrast administration. Perfusion differences between benign adenomas and metastasis provide a second reliable set of criteria for diagnosis (7,8). Adenomas are characterized by rapid washout of contrast agent, whereas metastasis show slow contrast washout (Figs. 32.4, 32.5). Percentage washout measurements are made on images taken at 60 to 75 seconds following the onset of intravenous contrast injection (enhanced attenuation). Delayed attenuation measurements are made on images obtained at 10 or 15 minutes following contrast injection. Absolute and relative percentage washout (RPW) calculations are made (Table 32.2). Benign lesions show greater than 60%
absolute percentage washout (APW) and greater than 40% relative percentage washout at 15 minutes. A number of studies have looked at calculating washout values from 10-minute and from 15-minute delay images. It seems to make little practical difference. The 60% APW and 40% RPW criteria are easier to remember and seem to work effectively using either 10- or 15-minute delayed images (1,9,10).
Table 32.1 Imaging Findings of Malignant and Benign Adrenal Lesions | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||
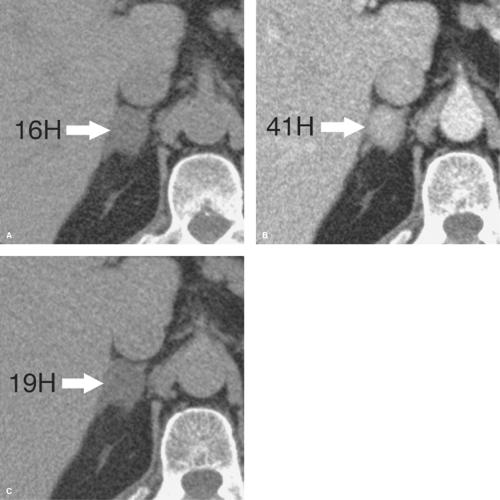 Figure 32.4. Benign Lipid-Poor Adrenal Adenoma—CT. A. Pre-contrast scan shows a small right adrenal mass (arrow) with an attenuation of 16 H, too high to characterize the lesion as a lipid-rich adrenal adenoma. B. Image at 1-minute postintravenous contrast administration shows enhancement attenuation of the lesion (arrow) at 41 H. C. Delayed image obtained at 15-minute postcontrast administration shows a delayed attenuation of the lesion (arrow) at 19 H. Absolute percentage washout (APW) calculates to 88% (see Table 32.2). Relative percentage washout calculates to 53%. These findings characterize this lesion as a lipid-poor adrenal adenoma (see Table 32.1). |
MR characterization depends on chemical shift techniques that detect intracellular lipid (11). Chemical shift MR relies on the different precession frequencies of fat protons versus water protons. When fat and water molecules occupy the same voxel, the MR signal from fat and water tend to cancel out each other reducing the signal intensity. Chemical shift MR consists of IP sequences when fat and water signals are additive and OP sequences when fat and water signals are subtractive. A reduction in signal intensity on OP images as compared to IP images is indicative of intracellular fat. When evaluating adrenal nodules, the chemical shift MR finding of signal drop indicates a benign lipid-rich adenoma (Fig. 32.6). Though some studies indicate a slight increase in the sensitivity of MR as compared to noncontrast CT, both modalities characterize essentially the same subset of lipid-rich adenomas. MR offers no capability to characterize lipid-poor adenomas, which are categorized along with metastases as indeterminate lesions when they fail to show signal loss on OP images. Contrast washout techniques following gadolinium enhancement on MR have so far not been successful in characterizing incidentalomas. Patients with lesions not characterized by chemical shift MR should be considered for repeat study with contrast-enhanced CT or PET-CT.
PET-CT shows high sensitivity in the detection of malignant lesions, which because of high metabolic activity accumulate
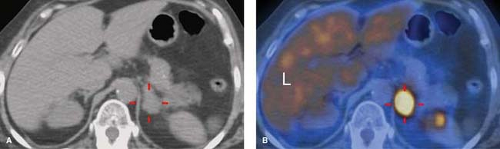
FDG (12,13,14). Hemorrhage or necrosis within a metastasis may cause falsely negative FDG uptake. Some metastases are false negative on PET including those from neuroendocrine tumors and bronchoalveolar lung carcinoma (5). Some benign lesions include occasional adenomas, infectious and inflammatory lesions may show slightly increased activity. Of PET-positive adrenal lesions, about 5% are false positive (5). Lesions smaller than 1 cm are not accurately evaluated by PET. Lesions that show more FDG uptake than the liver parenchyma are considered to be malignant (Fig. 32.7). SUVmax greater than 3.1 correlates with malignancy (5).
FDG (12,13,14). Hemorrhage or necrosis within a metastasis may cause falsely negative FDG uptake. Some metastases are false negative on PET including those from neuroendocrine tumors and bronchoalveolar lung carcinoma (5). Some benign lesions include occasional adenomas, infectious and inflammatory lesions may show slightly increased activity. Of PET-positive adrenal lesions, about 5% are false positive (5). Lesions smaller than 1 cm are not accurately evaluated by PET. Lesions that show more FDG uptake than the liver parenchyma are considered to be malignant (Fig. 32.7). SUVmax greater than 3.1 correlates with malignancy (5).
Lesions not categorized by these methods are considered for follow-up imaging in 4 to 6 months or for image-guided biopsy. CT-guided adrenal biopsy is safe, with hemorrhage and pneumothorax as unusual complications. Biopsy may be performed using a transhepatic approach or with decubitus positioning with the adrenal lesion side down to diminish the risk of pneumothorax. Caution should be used and biopsy generally avoided if the lesion is likely to be a pheochromocytoma. Percutaneous biopsy of a pheochromocytoma may precipitate a hypertensive crisis.
Table 32.2 Adrenal Nodule Percentage Washout Formulas | ||||||||
|---|---|---|---|---|---|---|---|---|
| ||||||||
Adrenal Endocrine Syndromes
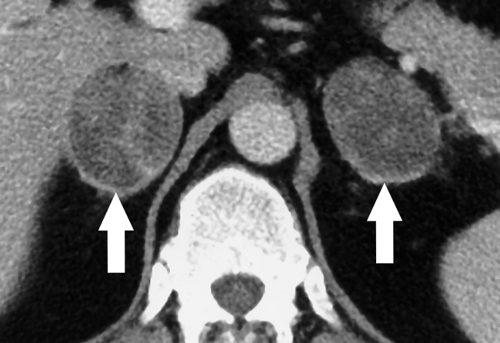
Cushing syndrome is caused by excessive amounts of hydrocortisone and corticosterone released by the adrenal cortex. Clinical signs include hypertension, truncal obesity, easy bruisability, generalized weakness, diabetes mellitus, and oligomenorrhea. Adrenal hyperplasia causes 70% of cases of noniatrogenic Cushing syndrome. The hyperplasia is stimulated in 90% of cases by a pituitary microadenoma that produces adrenocorticotropic hormone (ACTH). MR of the sella turcica is recommended for suspected pituitary adenomas. In 10% of cases, the source of ACTH is ectopic, usually from lung malignancies. Benign adrenal adenomas cause 20% of cases of Cushing syndrome, and adrenal carcinoma causes the remaining 10%. A subclinical form of Cushing syndrome has been associated with the presence of small adrenal adenomas found incidentally. It may be more common than classic Cushing syndrome. Clinical evaluation for hypertension, Type 2 diabetes, and obesity is often recommended. Atrophy of the contralateral adrenal gland may be present due to ACTH inhibition.
Conn syndrome, produced by elevated levels of aldosterone, causes 1% to 2% of systemic hypertension (15). The clinical diagnosis is made by the findings of persistent hypokalemia,
increased serum and urine aldosterone, and decreased renin activity in the plasma. A solitary, benign, hyperfunctioning adrenal cortical adenoma is the cause of 80% of cases, and adrenal hyperplasia is the cause of the remaining 20%. Adenomas are treated with surgical resection, whereas hyperplasia is treated medically. Adenomas that produce Conn syndrome tend to be small (<2 cm); therefore, strict attention to excellent MDCT technique using thin slices is necessary for accurate localization. Adrenal venous sampling is used to confirm the site of excess aldosterone secretion and to differentiate adenoma from hyperplasia in problem cases.
increased serum and urine aldosterone, and decreased renin activity in the plasma. A solitary, benign, hyperfunctioning adrenal cortical adenoma is the cause of 80% of cases, and adrenal hyperplasia is the cause of the remaining 20%. Adenomas are treated with surgical resection, whereas hyperplasia is treated medically. Adenomas that produce Conn syndrome tend to be small (<2 cm); therefore, strict attention to excellent MDCT technique using thin slices is necessary for accurate localization. Adrenal venous sampling is used to confirm the site of excess aldosterone secretion and to differentiate adenoma from hyperplasia in problem cases.
Adrenogenital syndrome usually occurs in newborns and infants who have an enzyme deficiency (11β- or 22-hydroxylase), leading to the deficient production of cortisol and aldosterone and an excess of precursors, especially androgens. These infants have adrenal hyperplasia, which is usually well demonstrated by US. Both adrenal adenomas and carcinomas may be a cause of masculinizing or feminizing syndromes in older patients.
Addison disease refers to primary adrenal insufficiency, which occurs only after 90% of the adrenal cortex is destroyed. The most common cause (60% to 70%) in the United States is idiopathic atrophy, which is probably an autoimmune disorder. The adrenal glands shrink and may not be detectable with imaging methods. Additional causes involve destruction of the glands by tuberculosis, histoplasmosis, infarction, disseminated fungal infection, lymphoma, or metastatic tumor. Adrenal calcification suggests prior tuberculosis or histoplasmosis. Bilateral enlargement is seen with active infection. Lymphoma and metastases replace the glands with tumor.
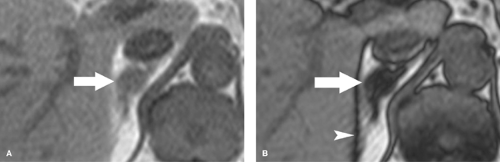
Pheochromocytoma is a rare catecholamine-secreting tumor that causes hypertension, headaches, and tremors. Paroxysmal attacks are characteristic but not always present. Pheochromocytoma is said to follow the “rule of tens”: 10% are bilateral, 10% are extra-adrenal, 10% are malignant, 10% are familial, and 10% are detected as “incidental” findings (Fig. 32.8). Pheochromocytoma is associated with multiple endocrine neoplasia (MEN II), von Hippel–Lindau syndrome (16), and neurofibromatosis. Pheochromocytoma is the most common adrenal tumor to hemorrhage spontaneously (Fig. 32.9). CT is the usual imaging method of choice for detecting the tumor when clinical manifestations are present (3). The literature has traditionally advised against the use of intravenous contrast media in patients with pheochromocytoma because of a presumed risk of precipitating adrenergic crisis. More recent experience indicates no significant risk with nonionic contrast media (17). Most tumors are larger than 2 cm in diameter. Tumors vary from purely solid to complex to predominantly cystic. Calcification is rare, but usually “eggshell”
in configuration when present. Most tumors enhance avidly and washout slowly similar to malignant lesions. Findings are variable, however, as some lesions show poor contrast enhancement or rapid washout seen with benign lesions. On MR, high signal (a “lightbulb” lesion) on T2WI is characteristic but seen in only 70% (18). Chemical shift MR shows no change in the signal intensity between IP and OP images. If no lesion is found in the adrenal gland, scanning is extended to include the chest and remainder of the abdomen and pelvis. Extra-adrenal sites for pheochromocytoma include the organ of Zuckerkandl near the bifurcation of the aorta, the bladder (Fig. 32.10), and the para-aortic sympathetic chain. Radionuclide scans using 131I- or 123I-metaiodobenzylguanidine (MIBG) are also effective in localizing pheochromocytoma. PET-CT shows increased FDG uptake in most tumors including some that are MIBG negative. Atypical appearance of the tumor is relatively common on all imaging modalities.
in configuration when present. Most tumors enhance avidly and washout slowly similar to malignant lesions. Findings are variable, however, as some lesions show poor contrast enhancement or rapid washout seen with benign lesions. On MR, high signal (a “lightbulb” lesion) on T2WI is characteristic but seen in only 70% (18). Chemical shift MR shows no change in the signal intensity between IP and OP images. If no lesion is found in the adrenal gland, scanning is extended to include the chest and remainder of the abdomen and pelvis. Extra-adrenal sites for pheochromocytoma include the organ of Zuckerkandl near the bifurcation of the aorta, the bladder (Fig. 32.10), and the para-aortic sympathetic chain. Radionuclide scans using 131I- or 123I-metaiodobenzylguanidine (MIBG) are also effective in localizing pheochromocytoma. PET-CT shows increased FDG uptake in most tumors including some that are MIBG negative. Atypical appearance of the tumor is relatively common on all imaging modalities.
Benign Adrenal Lesions
Adrenal hyperplasia is the cause of 70% of the cases of Cushing syndrome and 20% of the cases of Conn syndrome (15). Adrenal hyperplasia is important to differentiate from adrenal adenoma as a cause of endocrine syndromes. The syndrome is usually treated medically when hyperplasia is causative, whereas surgical removal of hyperfunctioning adrenal adenomas is usually curative. Half of the cases of biochemically hyperplastic glands will appear anatomically normal on CT and MR. In the remainder of cases, both glands will be diffusely enlarged but maintain their normal adrenal shape (Fig. 32.11). Uncommonly, hyperplasia may appear nodular and mimic solitary or multiple adenomas. In diffuse hyperplasia, the limbs of the adrenal glands are longer than 5 cm and exceed 10-mm thickness. Chemical shift MR occasionally shows a loss of signal on opposed-phase images. Metastatic disease, tuberculosis, and histoplasmosis may also cause diffuse adrenal enlargement and mimic the appearance of adrenal hyperplasia.
Adrenal myelolipomas are rare, nonfunctioning benign tumors arising from bone marrow elements in the adrenal gland. The tumors have no malignant potential. They range in size up to 30 cm and are frequently inhomogeneous because of their mixed components of marrow fat and hemopoietic tissue. Large lesions (>5 cm) have a tendency to hemorrhage. Calcifications are present in 20%. Identification of regions of macroscopic fat (-30 to -100 HU) within the tumor by CT or MR is definitive in making the diagnosis (Fig. 32.12). CT attenuation less than -30 H is definitive (19). MR shows high-signal fat on T1WI and T2WI. Fat saturation pulse sequences showing decreased signal confirm the diagnosis. Chemical shift MR is usually not useful as the macroscopic fat cells have little intracellular water. PET typically shows no avid FDG uptake. On US, they may be extremely echogenic and blend in with retroperitoneal fat.
Adrenal hemorrhage is most common in newborn infants, usually induced by episodes of hypoxia, birth trauma, or septicemia. Most cases are bilateral. In children, adrenal hemorrhage may be associated with child abuse. In adults, blunt trauma (80%) and infection are the most common causes of adrenal hemorrhage. Unilateral hemorrhage is most common in adults, with the right adrenal most frequently affected. Bilateral hemorrhage may cause adrenal insufficiency. On unenhanced CT, hemorrhage shows high attenuation (50 to 90 HU). Hemorrhage on CT is hypodense compared with the
liver and spleen on contrast-enhanced studies (Fig. 32.13). Stranding in the periadrenal fat and thickening of the adjacent fascia are additional findings. MR is highly sensitive and specific for adrenal hemorrhage with imaging features dependent on the age of the hemorrhage. Acute hemorrhage is isointense on T1WI and low intensity on T2WI. Subacute hemorrhage is bright on T1WI and either dark or bright on T2WI. Old hemorrhage with hemosiderin content is low signal on both T1WI and T2WI. US demonstrates a hypoechoic mass that shrinks and becomes less echogenic over time.
liver and spleen on contrast-enhanced studies (Fig. 32.13). Stranding in the periadrenal fat and thickening of the adjacent fascia are additional findings. MR is highly sensitive and specific for adrenal hemorrhage with imaging features dependent on the age of the hemorrhage. Acute hemorrhage is isointense on T1WI and low intensity on T2WI. Subacute hemorrhage is bright on T1WI and either dark or bright on T2WI. Old hemorrhage with hemosiderin content is low signal on both T1WI and T2WI. US demonstrates a hypoechoic mass that shrinks and becomes less echogenic over time.
Adrenal calcifications, in both children and adults, most commonly result from adrenal hemorrhage (Fig. 32.14). Tuberculosis and histoplasmosis may cause diffuse adrenal calcification associated with Addison disease. Adrenal tumors that calcify include neuroblastoma and ganglioneuroma in children and adrenal carcinoma, pheochromocytoma, and ganglioneuroma in adults. Adrenal pseudocysts attributable to previous hemorrhage are the most common calcified adrenal masses in adults. Wolman disease is a rare, autosomal recessive lipid disorder associated with enlarged calcified adrenal glands, hepatomegaly, and splenomegaly.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree