Vision is one of our most vital senses, deriving from the eyes as well as structures deep within the intracranial compartment. MR imaging, through its wide selection of sequences, offers an array of structural and functional imaging tools to interrogate this intricate system. This review describes several advanced MR imaging sequences and explores their potential clinical applications as well as areas for further development.
Key points
- •
MR imaging allows for high-resolution imaging of the retina but remains inferior to optical coherence tomography (OCT) in this aspect. Its primary advantage lies in its ability to characterize anatomic and functional parameters without depth limitation.
- •
Conventional single-shot echo planar-based diffusion-weighted imaging (DWI) is prone to susceptibility artifacts at tissue interfaces, which can be prominent in the periorbital region.
- •
Magnetization transfer ratios (MTRs) may play a role in monitoring therapy in demyelinating pathologies, such as multiple sclerosis.
- •
Diffusion tensor tractography (DTT) may aid in surgical planning of lesions within the vicinity of the optic radiations (ORs).
- •
Retinotopic mapping using functional MR (fMR) imaging can elucidate the visual cortex and potentially help in surgical planning.
Introduction
Millions of individuals are afflicted by blindness, and millions more suffer from varying degrees of visual impairment. There are several tools at the clinician’s disposal for evaluating this population, including OCT, fluorescein angiography, visual evoked potentials, and high-resolution ultrasonography. However, these techniques have inherent limitations, especially in evaluating the intracranial components of the visual pathway. MR imaging, through its wide selection of sequences, offers an array of structural and functional imaging tools to characterize these otherwise hidden regions. This review describes several advanced MR imaging sequences using an anatomically based approach from the retina to the visual cortex. It also explores their potential clinical applications as well as areas for further development.
Introduction
Millions of individuals are afflicted by blindness, and millions more suffer from varying degrees of visual impairment. There are several tools at the clinician’s disposal for evaluating this population, including OCT, fluorescein angiography, visual evoked potentials, and high-resolution ultrasonography. However, these techniques have inherent limitations, especially in evaluating the intracranial components of the visual pathway. MR imaging, through its wide selection of sequences, offers an array of structural and functional imaging tools to characterize these otherwise hidden regions. This review describes several advanced MR imaging sequences using an anatomically based approach from the retina to the visual cortex. It also explores their potential clinical applications as well as areas for further development.
Retinal MR imaging
The retina forms the innermost lining of our eyes, measuring between 200 and 250 μm in thickness. It is partitioned into 9 major anatomic layers: the inner limiting membrane, nerve fiber layer, ganglion layer, inner plexiform layer, inner nuclear layer, outer plexiform layer, outer nuclear layer, outer limiting membrane, and the retinal pigment epithelium. Light traverses the cornea, lens, and vitreous before interacting with the retina, where it is then converted into biologic signals in the form of action potentials. These action potentials are then transmitted along the optic nerves to the rest of the visual pathway.
Historically, in vivo imaging of the human retina has been confined to the domain of near-infrared light techniques, such as OCT. Recently, high-resolution anatomic MR imaging of the human retina has been performed on 3-T clinical scanners using balanced steady state fast precession (bSSFP) ( Fig. 1 ). To minimize motion artifacts, eye-fixation was used with cued blinking every 4 to 8 seconds. Using a conventional head coil together with a custom-designed eye-shaped receiver coil (to improve signal to noise ratio [SNR]), resolutions of up to 100 × 200 × 2000 μm were achieved. Despite such advances, OCT continues to hold the advantage in terms of spatiotemporal resolution.
MR imaging does offer several advantages including the ability to measure various physiologic as well as functional parameters, such as changes in local blood flow, in a noninvasive manner. The different parameters can be evaluated within a single session without a significant depth limitation ( Box 1 ). For instance, bSSFP-based pseudocontinuous arterial spin labeling (pCASL) has been used to achieve high-resolution blood flow mapping of the human retina. In particular, this sequence is considerably more resistant to susceptibility artifact than many conventional fMR imaging techniques. Similar results have also been achieved using turbo spin echo (TSE)-based pCASL. Potential clinical applications include the preclinical and longitudinal evaluation of diabetic retinopathy, glaucoma, and retinal degeneration. Areas for future work include improving the spatiotemporal resolution as well as additional contrasts to better visualize the retinal layers; this can be facilitated through technological advances such as increased field gradients, novel fast acquisition sequences, as well as intravascular contrast agents.
- •
High-resolution (100 × 200 μm in plane) anatomic images can be obtained using bSSFP as well as turbo spin-echo sequences.
Advantages
- •
Not depth limited
- •
Able to characterize different physiologic and functional parameters in a single session
Disadvantages
- •
Limited spatial resolution compared with optical imaging techniques such as OCT
- •
May require specialized equipment such as eye-shaped surface receiver coils
Optic nerve MR imaging
The optic nerve (cranial nerve II) transmits visual information from the retina to the brain and comprises retinal ganglion cell axons. It derives from the diencephalon and is considered a part of the central nervous system. Consequently, it is myelinated by oligodendrocytes rather than by Schwann cells. A portion of the optic nerve fibers decussate at the level of the optic chiasm, where most synapse at the lateral geniculate nucleus of the thalamus (Fig. 2 of Imaging of Ocular Motor Pathway). A smaller proportion synapses at other nuclei, including the pretectal nuclei.
Diffusion-weighted imaging
DWI is based on the micromovements of extracellular water molecules, which are elucidated by the application of dephasing and rephasing diffusion gradients during the preparatory phase of a sequence (usually spin-echo echo planar imaging [EPI]) ( Box 2 ). Immobile hydrogen proton molecules (eg, abscesses, stroke) are rephased by the second gradient, whereas those associated with mobile water molecules (eg, cerebrospinal fluid [CSF]) experience a phase shift and generate a weaker signal. The diffusion magnitude is calculated from 3 diffusion images, obtained through the application of the above-mentioned gradients in 3 spatial directions. The degree of diffusion weighting of a sequence is expressed by its b-factor (seconds per square millimeter), which is proportional to the product of the gradient strength squared and the diffusion time interval ( b ∼ q 2 · Δ t ) . An apparent diffusion coefficient (ADC) map can then be generated from 2 diffusion sequences with different b-factors, which is independent of T2:
DWI is the diffusion-weighted signal intensity for a specific b-value, and b 0 is the reference image.
Diffusion-weighted imaging
- •
This method uses dephasing and rephrasing gradients during the preparatory phase of a sequence to determine the diffusivity of hydrogen protons.
- •
Diffusion restriction of the optic nerve may be seen in ischemic optic neuropathy, which may help in distinguishing from other causes of acute vision loss.
- •
Optic nerve imaging can be challenging because of its small size, as well as susceptibility artifacts from the adjacent periorbital structures.
Magnetization transfer contrast
- •
This method uses an off-resonance saturation pulse to saturate the macromolecular proton population, as seen in lipid-rich myelinated structures, resulting in apparent T1 shortening.
- •
A useful application is the magnetization ratio, whereby regions of demyelination, as seen in optic neuritis, demonstrate decrease in the ratio.
A variety of conditions may affect the optic nerve, including neoplastic, infectious, ischemic, and autoimmune processes. DWI has been used in the characterization of several of these diseases, including benign and malignant tumors, optic neuritis, and ischemic optic neuropathy (ION). ION is typically divided into anterior (AION) and posterior (PION) forms. AION typically presents with sudden painless loss of vision, a relative afferent pupillary defect, and a pale edematous optic disc. It may be further categorized into arteritic (associated with inflammatory causes) and nonarteritic (associated with vasculopathic risk factors) forms.
PION is less common and primarily involves the retrobulbar optic nerve. Its cause is similar to that of nonarteritic AION, with a predominance of systemic vascular diseases, including embolism and thrombosis. Generally, nonarteritic ION has better prognoses than arteritic cases. Like ischemic events in the cerebrum, the optic nerve may demonstrate diffusion restriction during an acute episode, with gradual pseudonormalization. Using ROIs to compare ADC values between the affected regions and normal cerebral white matter may serve as an internal validation. In this regard, MR imaging may be a useful adjunct in diagnosis, as PION is generally normal on fundoscopy. Unfortunately, some diseases that typically present with increased diffusivity, such as optic neuritis (ON), may occasionally demonstrate diffusion restriction. Therefore, other factors (eg, age, cardiovascular risk factors, and clinical presentation) should be taken into consideration when establishing the diagnosis.
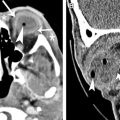
From a technical standpoint, several challenges exist to imaging the optic nerves with DWI. For instance, the optic nerves are small structures, and adjusting parameters such as slice thickness (eg, decreasing from 5 to 3 mm) may be necessary for adequate lesion characterization ( Fig. 2 ). In addition, the typical single-shot EPI-based sequences used for cerebral DWI are sensitive to susceptibility artifact and image distortions, which are prominent at the air-tissue and bone-tissue interfaces surrounding the orbits. Although these artifacts can be decreased to an extent using parallel imaging, they become more pronounced at higher field strengths (3 T and more). Alternative approaches include the implementation of modified multishot EPI sequences, as well as non-EPI-based sequences (which reduce artifacts at the expense of increased scan time). For instance, Porter and Heidemann implemented a readout-segmented EPI with parallel imaging, which combined reduced susceptibility artifact with superior motion correction while only marginally increasing the scan time.
Magnetization transfer contrast
Magnetization transfer contrast has shown promise in quantifying the extent of demyelination and axonal damage, offering advantages over T1 and T2 mapping in terms of image resolution, acquisition times, and postprocessing requirements. With this technique, an off-resonance saturation pulse is first used to saturate bound macromolecule protons, without affecting the free water proton population, which has a narrow frequency bandwidth (see Box 2 ). An exchange of longitudinal magnetization then occurs between the free water and macromolecular proton populations, whose longitudinal magnetization has been saturated. This exchange leads to shortening of the apparent longitudinal relaxation time T 1 sat , as only the free water protons are visible on clinical MR images. The T 1 shortening is most profound in tissues with a large macromolecule component, such as lipid-rich myelin sheaths ( Fig. 3 ).