Management of renal and genitourinary trauma has evolved during the last decades. There has been a transition towards conservative nonoperative management. This is due to the safety and outcome of the nonoperative approach in patients with renal trauma as well as in those with injury to other organs such as the liver and spleen. Improvement in imaging technique and minimally invasive treatment have also led to this. Techniques such as angioembolization in case of active bleeding and stenting in cases of urine extravasation have helped. FAST (focused assessment with sonography in trauma) plays a limited, though critical role as it evaluates presence of fluid presumed to be hemoperitoneum, on an abdominal ultrasound. This is important as ultrasound is easily available and inexpensive. In the presence of fluid, a stable patient undergoes CT abdomen. It is important to clamp the urinary bladder catheter if introduced prior to the scan, to partially distend the bladder so that it may be evaluated on US/CT. Contrast-Enhanced Multi-Detector Computed Tomography (MDCT) is the gold standard for imaging patients with blunt or penetrating abdominal injury who are hemodynamically stable. It rapidly completely evaluates both the upper and lower urinary tract. It also permits diagnosis of injury to other viscera, hence determining further patient management. Urethrography and cystography are useful for specific evaluation of urethral and bladder injuries. Protocol for the CT scan would include a non-contrast scan. In an acute setting no oral or rectal contrast can be administered. The protocol to be followed is a three-phase study – obtained in arterial, portal and venous phases followed by a delayed phase. Intravenous Pyelogram: In the era of CT, intravenous pyelogram has limited role in detection of renal injuries. Since its sensitivity is less, it should be done only when CT is not available. One-shot intravenous urography should be considered in hemodynamically unstable patients taken to the operating room for renal exploration or nephrectomy without prior imaging. In one-shot intravenous urography, 2 mL/kg contrast is injected and a delayed film of KUB region is obtained after 10–15 minutes to check for functioning contralateral kidney. Magnetic Resonance Imaging: Although CT and MRI are equally effective in staging the renal injuries, use of MRI is limited because of longer scan time, increased cost, and uncooperative patients in acute trauma setting. MRI can be an alternative to CT in patients allergic to iodinated contrast and pregnant patients. Retrograde Pyelography is very sensitive technique for detection of renal pelvis and ureter injuries. It is often used to outline the extent of renal pelvis and ureteric injury detected on CT or intravenous urography if additional clinical information is required. Its main advantage is it permits simultaneous placement of a ureteral stent if required. Radionuclide Renal Scintigraphy plays limited role in renal trauma. It is indicated to document function of kidney in patients with contraindication to iodine-based contrast or following up of repair of renovascular trauma. Commonly used radiopharmaceuticals in the renal scintigraphy are technetium 99m glucoheptonate, technetium 99m mercaptoacetyltriglycine, or technetium 99m diethylenetriamine pentacetic acid. Kidneys are third most commonly injured organ after blunt trauma. It is most commonly injured organ of genitourinary system and accounts for approximately 8%–10% of the abdominal injuries. The incidence of kidney injury in genitourinary trauma is 65%–90%. Males are three time more prone to injuries than female. In a large number of cases, renal trauma is isolated, however, 80%–95% of patients have other associated injuries. The kidneys are relatively well protected in the retroperitoneum, covered by fat and the Gerota’s fascia; however, despite this they are the most commonly injured organ of the urinary system. In a patient with history of trauma imaging is advised whenever there is abdominal tenderness, guarding, or rigidity. In institutes with dedicated trauma care centres, usually, all the patients with history of abdominal trauma undergo initial ultrasound evaluation (FAST). Hemodynamically stable patients found to have abdominal free fluid or hemoperitoneum on FAST further undergo dedicated cross-sectional imaging. All the trauma patients admitted to the hospital should be first evaluated for life-threatening injury with the advanced trauma life support approach. Then a survey should be done to look for flank tenderness, ecchymosis, and laceration, which may indicate renal injury. There are no consistent clinical features of renal injury and hence its clinical diagnosis is difficult to make. The best indicator of significant urinary system injury is macroscopic and microscopic haematuria but degree of haematuria does not match with the grade of injury. Minor injuries can produce macroscopic haematuria. Major injuries may not produce haematuria. Haematuria is absent in 50% of grade II injury, 7% of grade IV injury, 36% of renal vascular injury and 50% of ureteropelvic junction injury. Hence clinicians should not depend only on presence or absence of haematuria in the assessment of a patient with suspected renal trauma. Significant blow to the flank, especially acceleration and deceleration injury, ecchymosis of the flank, presence of entry and exit wounds in the flank, fracture of the lower ribs and dorso-lumbar vertebrae, hypotension with microscopic haematuria, macroscopic haematuria and presence of associated major injuries should make the clinician alert of renal injury and warrants immediate imaging assessment. Kidneys are well secured intraabdominal organ. Its high location in the retroperitoneum, surrounding fat and fascia, and neighbouring muscles, vertebrae and overlying ribs provide protection to the kidney. In children and adolescent, kidneys are more vulnerable to damage in abdominal injury because it is more inferiorly present in the abdomen, its larger size relative to the abdominal cavity, weaker connective tissue and fascia, less developed abdominal and paraspinal muscles, less ossification of lower ribs, and more mobility on its vascular pedicle. Understanding the mechanism of injury is important, because if a history of high acceleration and deceleration forces is elicited, the likelihood of renal injury is high and the patient should undergo imaging. The major elements causing renal trauma are the acceleration-deceleration forces. These cause injury to the renal pedicle and the ureteropelvic junction with resultant rupture or thrombosis. Acceleration forces may cause collision of the kidney with the surrounding ribs and spine. This results in parenchymal and vascular injury. Penetrating trauma causes greater damage due to the large amount of energy which is transmitted to the tissues. This causes cavitation, creating shear forces when the cavity collapses resulting in destruction in a larger area than just the urinary tract. There is tissue disruption, rupture of blood vessels and nerves and associated fractures. Most common mechanism of injury is blunt trauma and accounts for 80%–90% of cases. It is caused by motor vehicle accident, fall, sport, assault and pedestrian accident. Major factors that contributes to renal injury in blunt trauma are acceleration and deceleration forces. These forces result in shear injury. Kidneys are freely floating organ in the surrounding fat and fixed only at renal hilum and ureteropelvic junction. Hence it is more vulnerable to acceleration and deceleration injuries that rapidly and forcibly dislocate the kidneys. Rapid deceleration causes relative foreword movement of the kidney against a fixed hilum. It produces tension on the renal pedicle that lead to vascular laceration, thrombosis and ureteropelvic junction disruption. Rapid acceleration causes collision of kidney against the surrounding ribs and spine. It produces renal parenchymal and vascular injury. If the patient has past history of existing renal disease further imaging studies are indicated. In case of a solitary kidney or solitary functioning kidney, it is important to preserve the kidney even if it is damaged. Penetrating injury accounts for 10%–20% of cases. It is caused by gunshot and stab wounds. Most injuries are more severe and less predictable than blunt trauma. They are classified as high velocity, medium velocity, and low-velocity injuries. In high-velocity injuries, the damage usually extends beyond the path of projectile. The bullet conducts large amounts of energy to the tissues that creates a transient expansive cavitation in the tissue around the tract. This cavitation immediately collapses and built shear forces that causes tissue damage, injury to the blood vessels, nerves and bones in a much larger area than the projectile tract itself. In low-velocity injuries, the damage is usually limited to the path of the projectile. Entry site in the flank, upper abdomen and lower chest should alert the clinician and the radiologist to possible renal injury. Trauma through the anterior axillary line is commonly associated with injury to renal pelvis and vascular pedicle while trauma through the posterior axillary line causes damage to the renal parenchyma. Rarely kidneys can be exposed to iatrogenic injuries. It takes place during abdominal surgeries, percutaneous needle biopsy, percutaneous nephrostomy and extracorporeal shock wave lithotripsy. Kidneys with congenital and acquired conditions like ectopic location, PUJ obstruction, hydronephrosis, cysts and tumours are more vulnerable to blunt low velocity impact. Pathological kidney is less anatomically protected from the nearby structures. Furthermore, urine containing hydronephrotic kidney increases the force applied, worsening the severity of injury. Purpose of imaging are to detect renal injury, to assess the extent of organ damage and classify it, to demonstrate preexisting renal abnormalities, and to detect injuries of other organs. Criteria for imaging are: Children often do not show signs of hypotension despite significant blood loss but can still have significant renal injury. After trauma, they have high catecholamine output which maintains blood pressure until approximately 50% of blood volume has been lost. Hence liberal use of imaging is warranted in children. Organ Injury Scale (OIS) by American Association for the surgery of trauma (AAST) is most widely used trauma injury grading system. It was originally established in 1989. The original grading system did not fully consider vascular injury in its classification. Hence, it was revised in 2015 and published in 2018 (Table 10.12.5.2.1 and Graphic 10.12.5.2.1). It provides five grades of injury. Grade I–III represents low-grade injuries while grade IV–V represents high-grade injuries. Grade I injury accounts for 22%–28% of cases. It is characterized by renal parenchymal contusions and subcapsular haematoma. Contusions may be hypoechoic, hyperechoic or heterogeneous on sonography. It is seen as an ill-defined hypoechoic area with reduced perfusion on contrast-enhanced sonography. It may appear iso-dense or sometime hyperdense to normal renal parenchyma depending on the presence of clotted blood on plain CT images. Its detection is limited on the corticomedullary phase due to suboptimal enhancement of renal pyramids. It is characterized by focal ill-defined area of decreased enhancement on nephrographic phase. Excretory phase shows delayed or striated nephrogram of increased enhancement due to retained parenchymal contrast. A contusion must be differentiated from renal infarct, which would upgrade the renal injury. Contusion is ill-defined with enhancement on delayed images while infarct is typically wedge-shaped with no enhancement on delayed images. Subcapsular haematoma is well defined haemorrhagic collection along the renal surface between the renal capsule and parenchyma. It is seen as a non-enhancing lenticular area around renal parenchyma on contrast-enhanced ultrasound. Its density depends on the stage of blood. Acute haematoma typically appears hyperdense relative to renal parenchyma on plain CT images and does not show enhancement on post contrast images. As chronicity increases, its density decreases. Small haematoma is crescentic and large haematoma is biconvex. It causes mass effect on the underlying renal parenchyma, deforms the surface but does no displace the kidney. Occasionally, it causes enough pressure to decrease the renal perfusion and results in reactive hypertension, the so-called page kidney. Sometime, pseudosubcapsular haematoma may appear as hypodense collection along the renal surface. It is caused by patient movement during data acquisition. Presence of similar artifact in the other region on the same image helps to differentiate between true subcapsular haematoma and pseudosubcapsular haematoma. Pseudofracture appears as a sharp indentation of the renal contour near the renal hilum. It is seen on axial CT images at the level of hilar lobulation. Its characteristic location and absence of the perirenal fluid are clues to the correct diagnosis. Grade II injury accounts for 28%–30% of cases. It is characterized by renal parenchymal laceration ≤1 cm in depth without involvement of collecting system and perirenal haematoma limited by Gerota fascia. On contrast-enhanced ultrasound, renal lacerations are seen as linear or branching hypoechoic bands perpendicular to the capsule and may be associated with capsular discontinuity. They appear as irregular, linear or branching hypodense clefts extending from the renal capsule in the parenchyma on CT. Lacerations generally contains clotted blood and hence do not enhance on post contrast images. They are visualized on portal venous phase. Perinephric haematoma is collection present between the renal capsule and Gerota fascia. Because it is contained in the Gerota fascia it produces tamponade effect on renal bleeding. Acute haematoma typically appears hyperdense relative to renal parenchyma on plain CT images and does not show enhancement on post contrast images. It extends over a wider area and generally displaces the kidney rather than causing focal indentation on the renal surface. Rarely, localized perinephric haematoma limited to the renal capsule and bridging septum can produce focal indentation of the renal surface and mimics like subcapsular haematoma. Perinephric haematoma can occur as an isolated injury, but it is often associated with renal and/or vascular injury. Hence presence of perinephric haematoma should make the radiologist alert to look for associated injuries. Occasionally, it crosses the midline and spreads in the pelvis while still being limited by Gerota fascia. Presence of fluid in this region may be medial extension of perinephric haematoma in grade II injury, haemorrhage from renal vascular damage in grade III injury, or extravasated urine in grade IV injury. Hence, medially present collections should be more carefully evaluated. Grade III injury (Fig. 10.12.5.2.1) accounts for 22%–26% of cases. It is characterized by renal parenchymal laceration >1 cm in depth without involvement of collecting system and any low-grade trauma with associated vascular injury or active bleeding limited by Gerota fascia. Laceration involves the renal cortex and medulla. When deep lacerations are present, it is important to check the status of pelvicalyceal system. Presence of homogeneously enhancing renal tissue around the calyx on nephrographic phase and absence of extravasation of contrast on excretory phase helps to rule out pelvicalyceal system injury. Criteria for vascular injury is newly added in the 2018 OIS revision and includes pseudoaneurysm and arteriovenous fistula. Collectively, it is referred as contained vascular lesions. Pseudoaneurysm is well-defined, oval or round collection limited to the renal parenchyma or lacerated segment showing post contrast enhancement. Arteriovenous fistula is diagnosed when there is distension and early enhancement of the renal vein during the arterial phase. These contained vascular lesions follow enhancement pattern of adjacent renal artery and aorta and on delayed images remain similar in size and morphology. In contrast, active bleeding tends to track into surrounding tissue and limited to Gerota fascia. It has linear or flamelike appearance. It has a tendency to increase in size and retained a higher density than the renal artery and aorta on delayed images. Grade IV injury accounts for 15%–19% of cases. It is characterized by renal parenchymal laceration involving collecting system, renal pelvis laceration or complete pelviureteric laceration, intimal injury/thrombus of segmental renal artery or vein, segmental or complete renal infarction due to renal vessel thrombosis in the absence of active bleeding, and active bleeding outside Gerota fascia into the retroperitoneum or peritoneum. Injury to collecting system is considered when the renal lacerations are deep and extends through the renal calyces, renal pelvis and ureteropelvic junction. Its definitive diagnosis is made when there is presence of extravasation of excreted contrast beyond the collecting system during the excretory phase. Hence delayed phase must be acquired in case of suspected renal injury Ureteropelvic injuries are often associated with renal injury. Occasionally, it can occur in isolation. Isolated renal pelvic injuries can present as medial fluid collection around the pelvis. There are two types of ureteropelvic injury. In complete ureteropelvic avulsion, majority of the lumen is disrupted. There is absence of excreted contrast in the distal ureter and presence of excreted contrast around the injured renal pelvis. In partial ureteropelvic avulsion, lumen is partially disrupted. There is presence of excreted contrast in the distal ureter and around the injured renal pelvis. Segmental or complete renal infarction occurs due to segmental or complete thrombosis of the renal artery. Thrombosis is the result of intimal tearing or dissection secondary to shearing forces. Segmental infarcts manifest as a well-defined wedge-shaped area of non-enhancing hypodensity in the renal parenchyma during the corticomedullary and pyelographic phases. It has wider base at the renal capsule and apex towards the renal hilum. Complete renal infarct manifest as non-visualization of the entire kidney with abrupt cut-off of the renal artery on contrast-enhanced scan. Active bleeding is absent when the devascularization is secondary to complete renal artery thrombosis. Active bleeding in grade IV injury extends beyond Gerota fascia and spread into the anterior perirenal space, posterior perirenal space or peritoneum. On contrast-enhanced ultrasound, it is visualized in early stage as microbubble extravasation in perinephric area. It increases in size and density on delayed CT images. Grade V injury (Fig. 10.12.5.2.2A and B) accounts for 6%–7% of cases. It is characterized by main renal artery or vein laceration or avulsion from renal hilum, complete devascularization of kidney with active bleeding, and shattered kidney. Renal pedicle injuries are not common. Early detection and treatment of traumatic thrombosis or avulsion of renal artery is must because permanent, progressive loss of renal function starts after 2 hours of warm-ischemia time. In renal artery thrombosis, kidney perfusion is typically absent on contrast-enhanced ultrasound. Contrast-enhanced CT reveals abrupt truncation of renal artery just beyond its origin, infarction of the entire kidney with or without cortical rim sign and retrograde opacification of the renal vein from the inferior vena cava. Perinephric haematoma is typically absent in main renal artery occlusion. Cortical rim sign may be absent in acute injury and is reported to occur as early as 8 hours after injury. In cortical rim sign, there is a thin capsular and subcapsular enhancement due to intact blood supply from the capsular, peri-pelvic and periureteric vessels. Renal venous thrombosis is suggested by enlarged kidney, absent opacification of the renal vein, presence of filling defect in the renal vein, a persistent nephrogram, and delayed excretion of contrast into the collecting system. Avulsion of renal artery causes complete devascularization of the kidney. It is uncommon and life-threatening injury. It is caused by tearing of tunica muscularis and adventitia. Contrast enhancement CT reveals global infarction of the kidney, large medial perinephric haematoma around the aorta and renal hilum, and active arterial extravasation of contrast from the disrupted stump. This is in contradiction to grade IV injury in which devascularization is caused by vascular thrombosis without active bleeding. Shattered kidney is severe form of renal laceration. Several lacerations result in multiple fragments and loss of detectable renal parenchyma. The differentiation between the shattered kidney and multiple lacerations of lower-grade injury is subjective. The term shattered (Fig. 10.12.5.2.3) denotes extreme grade of tissue damage that prevents any meaningful healing and is usually associated with urine leak and active bleed. Imagine finding that raise suspicion of major renal injuries are medial haematoma indicating vascular injury, medial urinary extravasation of excreted contrast indicating renal pelvis or ureteropelvic junction injury, global lack of contrast enhancement of the renal parenchyma indicating renal artery occlusion, and combination of two or more of the following: haematoma more than 3.5 cm, medial renal laceration, and vascular contrast extravasation indicating brisk active bleeding. Box 10.12.5.2.1 includes the essential elements of renal injury to be mentioned in the report. ESSENTIAL ELEMENTS TO BE INCLUDED WHILE REPORTING RENAL INJURIES ON CECT Radiological assessment should give clear idea about the AAST grade of injury that helps to decide the treatment options. Important parameters that the clinician want to know are presence or absence of renal contusions and subcapsular haematoma, depth and severity of lacerations, extent of perinephric haematoma, active bleeding and its extent, status of renal vessels, pelvicalyceal system and pelviureteric junction and associated injuries (Table 10.12.5.2.2). Nonoperative management is the standard of care in hemodynamically stable patient regardless of AAST grade. Aims of conservative management are to reduce negative explorations and unnecessary repairs, avoid needless nephrectomy, increase the rate of renal salvage, and avoid long term complications like dialysis. Almost all grade I, II and few grade III renal injuries are treated conservatively with bed rest, analgesics, and hydration. More than 80% of collecting system injuries not involving the renal pelvis and ureter resolves spontaneously. Drainage procedures are indicated in patients with an enlarging urinoma, haematoma, abscess, fever, and increasing pain. Drainage should be achieved either with ureteral stent, percutaneous nephrostomy, or percutaneous catheter placement. First, ureteral stent placement or percutaneous nephrostomy is considered in case of persistent urinary extravasation. When perinephric collection persists in spite of stenting or nephrostomy, percutaneous catheter drainage should be considered. Nowadays, angioembolization is increasingly used in renal trauma. It is crucial adjunct for the successful conservative management of renal injuries. It increases the chances of renal salvage and preservation of renal function. Inappropriate settings, super-selective embolization is used to stop significant renal bleeding without the need for laparotomy. In selective cases, endovascular stents have been used in patients with renal artery thrombosis occurring secondary to intimal flaps. Indications of angioembolization are contained vascular injuries in hemodynamically stable patients, intravascular contrast extravasation, perinephric haematoma of more than 3.5–4 cm and medial site of injury. Operative management is reserved for hemodynamically unstable patients. Indications for laparotomy are life-threatening haemorrhage, renal pedicle injury, ureteropelvic junction avulsion, shattered kidney, rapidly expanding retroperitoneal haematoma, failed conservative treatment, and associated pancreatic or bowel injuries or significantly devascularized tissue in grade III or IV lacerations. These injuries have more risk of delayed complications like secondary haemorrhage from an arteriovenous fistula or pseudoaneurysm, urinoma or perirenal abscess, and renal hypertension. Depending on the severity of injury, repair of the main renal artery or vein or partial or total nephrectomy are surgical options to achieve early control of bleeding. Most surgeons concur that vascular repair should be done within 4 hours of injury if optimal renal function is to be expected. Most surgeons avoid surgery and allow the devascularized kidney to atrophy if the renal ischemia exceeded 4 hours and the opposite kidney is normal. If devascularization injury involves bilateral or a solitary kidney, reconstruction surgery is attempted even if the ischemia time has exceeded 4 hours. Follow-up imaging can be safely omitted in low-grade renal trauma and grade IV renal trauma without urinary leak. Purpose of reimaging is to diagnose possible complications and to assess cause of clinical deterioration Reimaging is recommended after 2–4 days in patients who had grade IV renal injuries with urinary extravasation on baseline scan, contained vascular injury, grade V renal injuries, and patients with signs of complications like fever, increasing flank pain, and abdominal distension. This timeframe allows contained vascular injuries to develop or urinoma to clinically progress. Fluid collections present on successive imaging in renal injury are either haematoma, urinoma, or abscess. Urinoma density ranges from 0 to 20 Hounsfield units and it shows contrast pooling during excretory phase. Haematoma density is generally more than 30 Hounsfield units and it does not show enhancement on postcontrast images. Abscess density is around 20 Hounsfield units and it shows peripheral enhancement on postcontrast images. Contrast pooling during excretory phase is absent in haematoma and abscess. Complications occur in 3%–10% of patients. Early complications are bleeding, infection, abscess, sepsis, urinary fistula, urinary extravasation with urinoma formation and hypertension. Most common complication is urinoma formation. Late complications are hydronephrosis, arteriovenous fistula, pseudoaneurysm, delayed hypertension, calculi and chronic pyelonephritis. Ureteral injuries are uncommon because they are present deep in the retroperitoneum and well-protected by fat and surrounding organs. They comprise less than 1% of genitourinary trauma. Only 20% injuries occur secondary to external trauma, with most due to penetrating trauma. Stab injury causes direct damage to short segment of ureter by penetrating object. Remaining 80% injuries are iatrogenic during intraabdominal surgeries. High-energy force in blunt injury results in rapid deceleration. It injures the ureters at fixed points, commonly at ureteropelvic and vesicoureteral junctions. It is often associated with injury to other organs mainly small and large bowel, kidney and urinary bladder. High energy also causes uncommon injuries like fracture of lumbar process and dorso-lumbar spine dislocation. Hence, the presence of high-energy forces like fall from height or high-speed motor vehicle crash should always raise the suspicion of ureteral injury. Patient often presents with gross or microscopic haematuria and flank pain. Ecchymosis may be present in flanks. As with renal injuries, haematuria is an unreliable indicator of ureter injury. It may be absent in one-quarter of patient. Variable clinical symptoms, rarity of ureteral injuries, and difficulty in clinical judgment often result in delay in diagnosis of ureter injury. Hence radiologists play a crucial role in identifying ureteral injuries. AAST grading system is used to guide the management (Table 10.12.5.2.2). The grading system is surgical and not well correlated with the imaging. Detection of ureteral contusions and degree of laceration are the major limitations of imaging. Distinction between grade II and III injury is almost impossible with any imaging technique. Role of radiologist is to detect partial laceration, complete transection, and its location. This distinction is important because partial lacerations are treated with stenting and complete transection require surgical repair. Acquisition of delayed images is very important to detect ureteral injury. Detection of contrast-opacified urine outside the confines of the ureter on excretory phase support the diagnosis of ureteric injury (Fig. 10.12.5.2.5). Presence or absence of contrast in the ureter distal the site of injury allows distinction between ureteral laceration and transection. Contrast will be present in the distal ureter in laceration and absent in transection. Opacification of distal ureter may be absent in transection, clearance of excreted contrast from the distal ureter by peristalsis, absence of distal passage of contrast due to poor peristalsis, or compression by nearby collection. Patients with injuries to the lower urinary tract (LUT) are being encountered more frequently in the radiology department as the incidence of severe trauma appears to be on the rise, mostly due to RTAs. The most common association of bladder and urethral injuries is with pelvic fractures. The LUT trauma occurs with pelvic fracture in high-speed collisions, run over of pedestrians or falls from heights. There is a high incidence of associated vascular damage, abdominal and thoracic visceral, ribcage, spinal and head injuries. Steering wheel impact may cause a distended bladder to burst from the steep rise of internal pressure. Imaging includes assessment of pelvic ring, vascular supply, urinary bladder, posterior male urethra, female urethra, anterior male urethra and the scrotum. These may each require a particular imaging modality or technique. The urinary bladder is a collapsible, muscular organ. When empty, it is protected from injury as it lies deep inside the pelvis and rests on the pelvic floor. When distended, it may reach up to the umbilicus. The bladder can be divided into a broad fundus, a body, an apex and a neck. The fundus or dome is the most mobile and weakest part of the bladder. The apex of the bladder is directed anteriorly towards the superior aspect of the pubic symphysis. The median umbilical ligament is seen extending from the apex of the bladder to the umbilicus along the anterior abdominal wall. It carries the peritoneum along with it forming the middle umbilical fold. The perivesical space lies under this peritoneal fold. The peritoneal reflection continues to form the recto-vesical (or uterovesical) pouch. The superior surface is related to intraperitoneal viscera like the intestine and the body of the uterus. The inferolateral surfaces of the bladder are related to the retro-pubic space of Retzius. Anterior to the urinary bladder and posterior to the pubic symphysis lies the retropubic space of Retzius which contains veins and a pad of fat. The transversalis fascia separates the space from anterior abdominal wall musculature. This potential space extends up to the umbilicus. The detrusor muscle is the thick muscular layer of the urinary bladder, made of smooth muscle fibres arranged in spiral, longitudinal and circular bundles. The trigone is a triangular part of its wall, between ureteral and internal urethral orifices, that forms the floor of the bladder above the urethra. It presents a smooth area of the bladder mucosa, in contrast to the rest of the irregular inner surface marked by rugae, a series of ridges, thick mucosal folds that allow for the expansion of the bladder as it fills. The trigone faces backward and downward and is related to the seminal vesicles, ductus deferens in men, and the vagina and cervix in women. The area surrounding the internal urethral orifice at the base of the trigone is described as the bladder neck. trigone A mucosal flap, ‘The Uvula of urinary bladder’ overhangs the internal urethral meatus posteriorly in the neck of the bladder. It probably plays a role in maintaining continence. Hypertrophy of the median lobe of prostate accentuates its obstructive effect and likely results in incomplete voiding or postvoid dribbling. The extraperitoneal parts of urinary bladder and the posterior urethra are fixed to the pelvic walls and thus more vulnerable to the shearing forces generated by rapid deceleration. Disruption of binding ligaments occurs with pelvic fractures, due to severe crushing/shearing forces. Intraperitoneal bladder injury occurs on a full bladder, due to rupture of the dome along with its investing layer of peritoneum. It may occur due to a direct impact on a distended bladder, even in absence of a pelvic fracture. This type of injury may occur more commonly in childhood due to the greater intraabdominal location of the bladder. The male urethra is divided into posterior and anterior parts. Posterior male urethra includes prostatic and membranous segments. The posterior urethra begins at the neck of the bladder and traverses the pelvic and urogenital diaphragms. The prostatic urethra, the most distensible part of urethra, is approximately 3.5 cm long. The verumontanum is a median ridge over the posterior wall of the urethral crest. The verumontanum or the colliculus seminalis is the summit of the urethral crest which is a median ridge over the posterior urethral wall. The prostatic utricle opens as a diverticulum on the colliculus along with the ejaculatory duct. The proximal urethral sphincter has two components, the internal and the intrinsic. The internal sphincter continues from the detrusor muscle, surrounding the neck of the bladder and urethra till the lower end of the urethral crest. The intrinsic urethral sphincter is smooth muscle around the distal part of prostatic urethra. Both internal, as well as intrinsic sphincters, are innervated by sympathetic nervous system, primarily responsible for passive continence. Beyond the prostate, it continues as the membranous urethra. This segment is the narrowest part of the male urethra. It is approximately 1–1.5 cm long. Sphincter urethrae, a band of striated voluntary muscle, arises from the posterior surface of pubic rami, and along with the transverse perineal, it encircles the membranous urethra, forming the external sphincter and inserts into the perineal body. The external urethral sphincter is under voluntary control, responsible for active continence. The male urethra is fixed to the pelvis at two points, the junction with the neck of the urinary bladder by puboprostatic ligaments and the membranous urethra by the perineal membrane at the floor of the pelvis. Injuries of the posterior urethra often involve these points. The anterior urethra in males is about 15–20 cm long and traverses through the corpus spongiosum. It runs over the ventral surface of the anus. It comprises of two parts, the bulbous (bulbar) and pendulous (penile) urethra. The membranous urethra enters the bulb of the penis below the floor of pelvis to join the bulbar urethra. The bulbar urethra has two components, a funnel-shaped proximal part and a distal part called the sump, which constitutes the widest part of the urethra. Both are located within the proximal part of corpus spongiosum of the penis, the bulb of the penis. The pendulous part of the urethra continues within the penis beyond the penoscrotal junction to the external urethral meatus at the tip of the glans penis. Within the expansion of the glans lies the dilatation called the navicular fossa of the urethra. The female urethra is 4 cm long, akin to the posterior urethra in the male (females lack anterior urethra). The proximal part of the urethral wall is made up of smooth muscle continuing from detrusor muscle of the bladder, the internal sphincter, responsible for continence. The lower urethra in the females is in close proximity to the anterior vaginal wall and the two are surrounded by the urethra-vaginal sphincter. This extends up to the inferior pubic ramus above the urogenital diaphragm. Injuries to the female urethra thus involve vaginal injury too. It is loosely attached to the pelvis and thus less prone to injury in pelvic fractures. The pelvic diaphragm consists of Levator ani muscles and their superior and inferior layers of investing fascia. Levator ani group of muscles comprise the pubococcygeus, the iliococcygeus and the puborectalis which converge from pelvic sidewalls to the perineal body. The coccygeus muscle and anococcygeal raphe complete the diaphragm posteriorly. It supports the viscera in the pelvic cavity, and surrounds the various structures that pass through it, namely the urethra, the rectum and the vagina in women. Perineal Body is a wedge-shaped fibromuscular mass, containing both collagenous and elastic fibres as well as skeletal and smooth muscles. In females, it lies between anal canal and lower end (vestibule) of vagina, in males, between anal canal and root of penis. It is larger in the female than in the male and supports to the posterior wall of the vagina. It is the site of convergence of many perineal muscles, including the bulbospongiosus, superficial and deep transverse perineal muscle, sphincter urethrae, superficial and deep parts of external anal sphincter and the levator ani. Fascial membranes of the pelvic and urogenital diaphragms, as well as the Colle’s fascia of perineum, merge into it. The urogenital diaphragm consists of fat and muscles around the membranous urethra invested by superior and inferior fascial layers. The muscles of the urogenital diaphragm consist of flat sheet of striated muscle originating from the pubic bone and ischiopubic arch of the pelvis. These are placed obliquely, converging to insert in the perineal body. The anterior part of these muscles encircle the urethra and act as the voluntary sphincter. The posterior margin of the urogenital diaphragm is bounded by the superior and inferior membranes which also converge into the perineal body. The fascial layers fuse anteriorly below the pubic symphysis to form the transverse pubic ligament. The fat contained within the superior and inferior fascial layers is imaged on CT as a V-shaped lucency with one limb on either side of the apex of prostate. Superiorly, this fat plane communicates with fat contained within the ischiorectal fossa. Anteriorly, it is limited by the transverse pubic ligament. The space of Retzius between the urinary bladder and pubic bones communicates inferiorly with the subcutaneous perineal fat (superficial perineal space) anterior to the transverse pubic ligament. Superiorly, it communicates with fat in the anterior abdominal wall external to the transversalis fascia. It extends superiorly into the rectus sheath where the posterior fascial layer ends below the level of umbilicus. The superficial perineal space lies between urogenital diaphragm and Colle’s fascia, the membranous layer of superficial fascia which is continuous with the membranous layer of superficial fascia of anterior abdominal wall (Scarpa’s fascia). The deep perineal space lies between superior and inferior fascia of the urogenital diaphragm. Posteriorly both superficial and deep spaces are closed by the convergence and merging of all three fascial layers in the perineal body. Pudendal canal, also known as Alcock’s canal, is a sheath derived from the fascia of the obturator internus muscle and is found in the lower lateral wall of the ischiorectal fossa. The pudendal canal lies on the medial surface of the obturator internus muscle and the medial aspect of ischial tuberosity above the falciform ridge. It transmits the pudendal nerve with internal pudendal artery and vein from the lesser sciatic foramen posteriorly to the deep perineal pouch anteriorly. The nerve supply of the lower urinary tract is derived from three groups of nerves. The pelvic parasympathetic nerves cause urinary bladder excitation and urethral relaxation. The lumbar sympathetic nerves, on the other hand, cause excitation of the urinary bladder base and urethra while inhibiting the body of urinary bladder. The third group of nerves, that is the pudendal nerve contracts the external urethral sphincter. The mortality (20%) and morbidity related to bladder trauma are high as a consequence of associated vascular, visceral and bony injuries and complications thereof, rather than bladder perforation itself. Urinary bladder trauma occurs in vehicular collision, fall from a height and pelvic crush injury. Rapid deceleration involves a shearing force that stretches and damages its ligamentous attachments along with the bladder walls. The seatbelt or a direct blow on a distended bladder may sometimes result in rupture without a pelvic fracture. Other causes are, penetrating injuries and iatrogenic injuries. Iatrogenic intraoperative injuries occur during endoscopic biopsy, prostatectomy, hysterectomy and emergency caesarian etc. If the patient has spina bifida or a previous spinal cord injury, there is limited awareness of bladder fullness and pelvic pain. The abdominal pain may be vague in onset and nature. A high index of suspicion in painless abdominal distension is needed in such cases. Intraperitoneal blood or urine may lead to ascites and ileus. Radiologic classification of bladder injuries is mentioned in Table 10.12.5.2.3.
Renal and genitourinary trauma
Introduction
Imaging modalities
Renal injuries
Clinical features
Mechanism of injury
Penetrating injuries
Traumatic injuries to kidneys with preexisting abnormalities
Indications of imaging
Grading

Low grade renal injuries

High-grade renal injuries

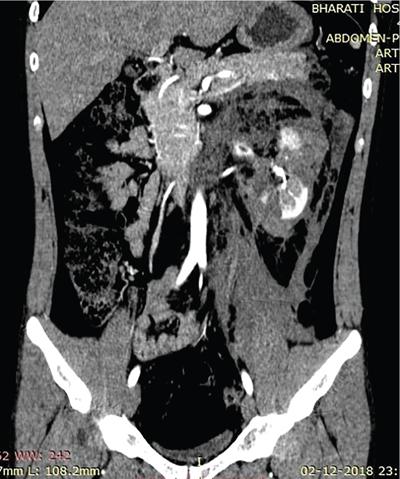
Management
Role of angiography and embolization (Fig. 10.12.5.2.4)
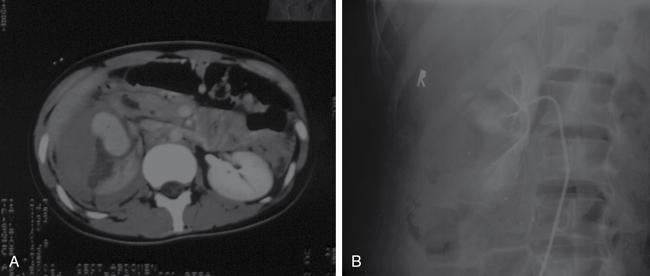
Complications
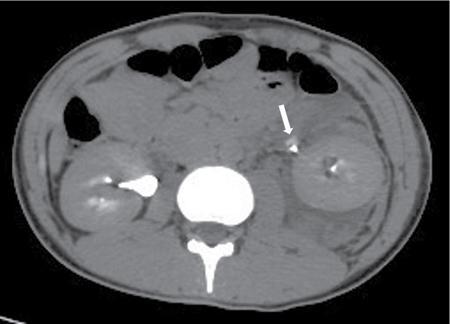
Ureter injuries
Introduction
Clinical features
Grade
Type
Injury
I
Haematoma
Contusion or haematoma without devascularization
II
Laceration
<50% transection
III
Laceration
>50% transection
IV
Laceration
Complete transection
V
Laceration
Complete transection with devascularization
Lower urinary tract trauma
Applied anatomy
Injuries of the urinary bladder
Radiology Key
Fastest Radiology Insight Engine









