RENOVASCULAR HYPERTENSION Ankur Gupta, Vasantha Kumar Venugopal Hypertension (HT) is a common condition affecting approximately 20% of adults and 2%–10% of children of all age groups. The prevalence of HT is about 25% in the urban population and 10% in the rural population of India. The essential HT (idiopathic) is the most common HT in all the age groups, accounting for 90%–95% of the total cases, and remainder are contributed by secondary known aetiology. Renovascular hypertension (RVH) is the most important cause of secondary HT and is mostly due occlusive lesion of renal arteries (RA) or its branches. It is responsible for 75% cases of secondary HT. It accounts for 1%–5% of all hypertensives in general population, whereas renovascular disease (RVD) causes 10% of the cases in children. The prevalence of RVH in selected referral group of population where clinical suspicion is high, its prevalence is around 30%. The clinical spectrum of RVD includes-isolated anatomical RAS without HT, RAS with HT, renal insufficiency (ischemic nephropathy) or both. Detection of the RVH in early stage is essential because it is potentially curable and reversible cause, and failure to treat unequivocally results in chronic ischemic nephropathy/end-stage renal disease. Numerous noninvasive (Colour Doppler Ultrasonography [CDUS], CTA, MRA and Captopril radionuclide renography) and minimally invasive (Conventional catheter angiography-gold standard and Renal vein renin measurement) imaging modalities are available for identification of renal vascular lesions. The intravenous urography for RVH imaging is obsolete and is of historical note. The ideal imaging modality should detect and localize renal artery stenosis (RAS), quantify the haemodynamic significance of the stenosis, to predict the response to a revascularization procedure. Most investigators consider 50%–60% stenosis of the renal artery significant; however, haemodynamically significant reduction in perfusion pressure in RA is seen at 70%–75% occlusion when systolic pressure gradient across stenosis is >10–20 mm Hg or >20% of aortic systolic pressure. The antihypertensive drug therapy leads to satisfactory medical management of RVH in most cases of moderate RVD. Interventional procedures like angioplasty, stenting are suitable for high risk and progressive renovascular syndromes refractory to medical therapy and in young adults in whom long-term management is required. The two most important causes of RVH are Atherosclerotic renal artery stenosis (ARAS), which involves older population, and fibromuscular dysplasia (FMD) which more often affects children and young adults. ARAS accounts for more than two-thirds cases of RVH. It tends to involve males above 45 years. It more often involves ostium and proximal one-third of renal artery, and bilateral affliction is seen in at least one-third of the cases. It is often associated with diabetes mellitus, dyslipidaemias and coexisting atherosclerosis of other arteries. The abdominal bruit in ARAS is of less diagnostic value. FMD on the other hand tends involve female (F:M ratio of 8:1) <35 years of age. It is usually unilateral when diagnosed, but can involve both RA. It usually involves distal two-thirds and branches of RA. A high pitched, often intermittent bruit, best heard in mid epigastrium is audible in about 60% of patients. Three histological types of FMD exists – intimal, medial and adventitial. The medial FMD is most common (has classical string of beads appearance) and it shows progression in one-third of cases and complications like thrombosis and dissection are rare. The intimal and adventitial FMD are rarely encountered and commonly shows progression and aforementioned complications. Other less common aetiologies of RAS are autoimmune diseases (Takayasu arteritis and polyarteritis nodosa), dissection, embolism, compression by extrinsic lesions, neurofibromatosis and hypercoagulable states. It is well established that the principal mechanism underlying RVH is Renin angiotensin aldosterone system (RAAS). Haemodynamically significant stenosis leads to decreased renal perfusion (ischemia) and as a compensatory response to this, affected kidney initiates hypersecretion of renin which in turn accelerates conversion of angiotensinogen to Angiotensin I, and further to Angiotensin II by angiotensin-converting enzyme (ACE). Angiotensin II acts in multiple ways to restore glomerular filtration-vasoconstriction of efferent arteriole, increase aldosterone release from adrenal, increased sodium absorption from proximal tubule, sympathetic nerve system activation, intra-renal prostaglandin concentrations, and nitric oxide production; all contributing to HT. Another important factor to consider in pathophysiology RAAS mediated RVH is presence or absence of contralateral kidney. In unilateral renal artery disease pressure diuresis and renin suppression by nonstenotic kidney mitigates the angiotensin II effects of ischemic kidney to some extent. But in a clinical scenario of solitary ischemic kidney or bilateral disease this salvage mechanism is absent, hence playing an additive role in the HT. Long duration HT leads to reverse tachyphylaxis (reduction in renin level) thereby leading to increase in total fluid volume and cardiac output. It might be interesting to note that in prolonged RVH, the nephrosclerosis that happens in the opposite kidney (initially normal) becomes the culprit for HT and hence correction of stenosis in the affected kidney may not relieve the HT. However, nephrectomy of the contralateral kidney can lead to correction of HT. The clinical features of RVH include The evidence-based guidelines as listed in ACR Appropriateness Criteria are for specific clinical conditions that are reviewed annually by a multidisciplinary expert panel. Although previously mentioned clinical scenarios points towards possibility of RVH, testing for RAS is not typically warranted for patients whose hypertension is well managed with medical therapy. This fact is supported by a randomized control trial of 947 patients from 113 centres ‘Cardiovascular Outcomes in Renal Atherosclerotic Lesions’ which showed no difference in multiple endpoints between medical therapy and renal stenting in patients with atherosclerotic RAS and hypertension or chronic kidney disease. The testing for RAS is warranted in following clinical scenarios The ACR appropriateness of imaging investigations in whom testing of RAS is warranted can be broadly categorized into two groups Radiology imaging investigations may be categorized as (a) noninvasive and (b) minimally invasive, or it may be more logical group them as procedures defining (i) structural abnormalities and (ii) functional status of kidneys (Refer to Table 10.4.1.3). Colour Doppler ultrasound is an easily available, noninvasive and inexpensive investigation without any ionizing radiation, however, is operator dependent and time taking relative to other tests. It is appropriate initial screening investigation of choice in strongly suspected cases of RVH with or without renal dysfunction. It demonstrates the main RA, hilar, segmental and the intraparenchymal arteries (interlobar and arcuate arteries); and also enables velocity assessment and waveform analysis. CDUS requires great amount of skill, hence pre-scan patient preparation and knowledge of examination techniques are important from the perspective of performing radiologist/technologist. From practical point of view obstacles encountered during scanning are obese habitus of patient, inability to hold breath and interposition of bowel gas, which limit optimal visualization of main RA in 20%–30% of the cases. First two are nonmodifiable factors and depends on skill of performing radiologist to some extent; however, problem to bowel gas can be mitigated by performing examination in early morning after overnight 12 hours fasting. The procedure begins with 30 degrees reverse Trendelenburg supine position. Low-frequency probe (2.5–5 MHz) is required to visualize aorta and RA. Before starting dedicated CDUS examination, an overview of B mode appearance of bilateral kidneys is suggested to look for asymmetry in size and cortical echotexture, which may help localize the laterality, degree and duration of RAS and exclude other renal pathologies. The two main imaging techniques for examination are (a) Anterior approach (b) Flank, and other two are ‘banana peel view’ and rarely used posterior approach. Right RA originates from anterolateral aspect of aorta and courses behind IVC. The problem frequently encountered in this approach is that Doppler angle is perpendicular to renal artery flow and interposition of IVC. These technical difficulties are alleviated by flank approach in which angle of insonation becomes parallel to blood flow and renal artery is visualized separately from IVC. Left RA originates from posterolateral surface of aorta and imaging clue to visualize it is identification left renal vein which is large and easy to locate, behind which lies artery. A pitfall that should be avoided is mistaking of inferior mesenteric artery (IMA) as left RA; however, this can be dealt with knowledge that IMA shows high resistance waveform and RA low resistance and typical IMA arises much lower in aorta. A second approach to visualize left RA is modified flank approach in which patient is turned to right lateral decubitus position resulting in mild midline shift of kidney, which then acts as its own window. Flank Approach demonstrates intra-renal/parenchymal arteries, and bilateral main RA as described above. Banana peel view is particularly useful to identify the ostium of RA, where most stenosis occurs in elderly. In this approach also, the patient is turned to the opposite decubitus position from the vessel being examined. The transducer is placed longitudinally, aorta is identified, and then probe is moved in an AP direction until the RA is identified arising from it. Correct use and knowledge of controls of USG machine (Knobology) is essential, and important ones required in CDUS are Doppler gain, pulse repetition frequency/Velocity scale, Doppler gate and Doppler angle. Doppler gain should be optimized to detect flow by increasing the gain to a level just below colour artefact. The pulse repetition frequency is the frequency of sampling; under sampling may underestimate peak velocities. To get the largest spectral images possible, one should decrease the velocity scale until the waveform fills almost the entire field and increase sweep speed. The Doppler gate should be set to include the entire artery lumen and Doppler angle/angle of insonation should be ≤60 degrees. The spectral waveform of renal artery is a typically low resistance waveform (just like any other parenchymal organ) with rapid early upstroke. The normal peak systolic velocity in main renal artery without any stenosis is <100 cm/s and it decreases distally in intrarenal branches. The resistive index (RI) value of 0.7 is used to discriminate normal and pathologic resistance to flow. The normal acceleration time (AT) is <0.07 s and acceleration index (AI) is >3.5 m/s2. The acceleration indices should be measured along the initial portion of systolic upstroke, avoiding compliance peak. The strongest and reproducible signals come from segmental and interlobar arteries as they course directly towards transducer. The ideal situation is when CDUS demonstrates bilateral RA along its entire length from ostium to its divisions into segmental and interlobar arteries. The Doppler criteria for RAS can broadly grouped as (a) Proximal/Direct and (b) Distal/Indirect. ACE inhibitor renal scintigraphy is functional investigation which complements anatomical data provided by CDUS, CTA and MRA. It gives result/data in the form of poor resolution scintigraphy images and scintigraphy time–activity curve. The rationale behind this test is disruption of the glomerular filtration autoregulation of the low perfusion kidney with RAS, which has become dependent on RAAS, by inhibition of ACE. The ACE inhibitor like captopril and enalapril decreases GFR in affected kidney, whereas function of normal contralateral kidney remains unaffected. The time–activity curve of affected kidney is obtained by intravenous administration of radiopharmaceutical agents like technetium-99m mercaptoacetyltriglycine (MAG3), iodine-131 orthoiodohippurate (OIH), or Tc-99m diethylenetriaminepentaacetic acid (DTPA) before and after ACE inhibition. Tc-99m DTPA is excreted by glomerular filtration whereas Tc-99m MAG3 and I-131 OIH are excreted by tubular secretion. Remarkable changes in the curve are obtained in kidney with haemodynamically significant RAS, whereas in normal kidney baseline curve and post-ACE inhibition curve are same. Choice of Radionuclide: In patients with azotemia, tubular agents such as 99mTc-MAG3 or 123I are the agents of choice. In patients with normal function, 99mTc-MAG3 and 99mTc-DTPA appear to give comparable results. Table 10.4.1.5 explains the diagnostic criteria for MAG3 and DTPA scintigraphy suggestive of high probability of RAS. Patient preparation for test includes stopping the ACE inhibitor 2–5 days prior to test and adequate hydration. After obtaining baseline scintigraphy data (images and curve), 25 mg oral captopril or 0.04 mg/kg of intravenous enalapril maleate is administered. ACE inhibitor scintigraphy data is obtained for 30 min after 1 hour of oral dose of captopril or 15 min of intravenous injection of enalapril. Time–activity curves are generated from the renal cortex and pelvis. Renal uptake is measured at 1–2-min intervals after injection. Consensus panels have recommended that the test be interpreted as low, intermediate (indeterminate) and high probability for RVH. A low-probability test shows a normal ACE inhibition renogram or a grade II renogram that is unchanged or improves after ACE inhibition. An intermediate probably test shows a kidney that is small and poorly functioning or kidneys with abnormal baseline renograms (grades III–V) that are unchanged after ACE inhibition. A high probability test or findings suggestive of haemodynamically significant stenosis are worsening of the scintigraphic curve (exaggeration of baseline asymmetry), change in the scintigraphic grade, decrease in the relative uptake, parenchymal retention, increase in the 20-min/peak uptake ratio or renal cortical activity at 20 min (RCA20), and prolongation of Tp (time to peak). The ACE inhibitor scintigraphy can be very accurate in selected high-risk cases. Acosta GMJ et al. reported a specificity of 91%, but the sensitivity was 60%. The shortcomings of this test are low sensitivity in presence of bilateral RAS, impaired renal function (Sr Creatinine > 2 mg/dL), chronic intake of ACE inhibitors and obstructive uropathy. MRA is a robust, safer, reproducible but expensive alternative for imaging the RA. It lacks the risks associated with nephrotoxic iodinated contrast and ionizing radiation. The MRA techniques can be broadly categorized as (1) Contrast-Enhanced MRA (CE-MRA) and (2) Unenhanced MRA (UMRA). CEMRA: Time-resolved breath-hold CE-MRA using 3D GRE sequence reveals good agreement with DSA (gold standard) in grading RAS. It is based on the T1 shortening effect of intravenously injected paramagnetic agent which renders the blood bright and stationary tissue dark. The imaging technique involves intravenous injection of gadolinium contrast (0.2–0.4 mmol/kg) at the rate of 2–4 mL/s, bolus tracking method allowing automatic triggering of the acquisition, and K space readout algorithm in which centre of the K space with contrast related information is acquired first and periphery of the K space is interpreted last. Postprocessing techniques such as subtraction, multiplanar reformation (MPR), Volume rendering (VR) and maximum intensity projection (MIP) are used to gain maximum out of source images. However, using the reconstructed images alone for diagnosing and grading the stenosis can give erroneous results and correlation with source image is important. UMRA: UMRA are innovative techniques not requiring paramagnetic contrast agents. These techniques are now gaining importance and are considered valid alternative to CEMRA. This is particularly more relevant in patients with severe renal dysfunction, due to known association between nephrogenic systemic fibrosis (NSF) and gadolinium-based contrast agents. The most commonly used UMRA technique is ‘balanced’ steady state-free precision (SSFP). Other techniques are Time of Flight (TOF) and Phase Contrast (PC). ‘Balanced’ SSFP is the gradient echo sequence which has proved to be promising in the study of RAS for several reasons, such as high signal-to-noise ratio, relatively short acquisition time, the application of ASL preparatory pulses (to suppress background signals and selectively enhance arteries), use of cardiac and respiratory triggers (navigators) and as well as the radical reduction of motion artefacts. ASL in combination with SSFP is also known as NATIVE True FISP (SIEMENS), Time-SLIP (TOSHIBA), Inhance inflow inversion recovery (GE) or B-TRANCE (PHILIPS). The phase contrast-MRA (PC-MRA) can serve as an adjunct to CE-MRA and provide functional/physiological information like peak systolic velocity and renal aortic ratio, but unlike Doppler, no obvious guidelines exist for grading stenosis. As compared with CEMRI, UMRA tends to slightly overestimate the stenosis due to distal signal loss for reduced entry flow and/or for spin dephasing caused by blood flow turbulence and it’s another limitation is restricted volumetric coverage due to which accessory renal artery may go undetected. UMRA, however, performs better than CEMRA in the imaging of intrarenal segmental arterial branches due to high signal-to-noise ratio. Segmental renal artery imaging is of diagnostic value in patients with dysplasia or vasculitis involving peripheral renal arterial branches. Most series have shown excellent correlation between CEMRI and DSA. Bakker et al. studied 44 patients suspected of having RAS and 10 potential kidney donors, and found sensitivity of 97% and specificity of 92% for correct identification of stenosis >50%. Many studies have also been performed comparing UMRA and DSA. Wyttenback et al. studied 43 patients suspected having RVH and found that for balanced SSFP sensitivity, specificity, accuracy, and NPV of 100%, 93%, 94% and 100%, respectively, for observer 1 and 95%, 95%, 95% and 99%, respectively, for observer 2 (artery-by-artery analysis). The patients in the above comparison studies had atherosclerotic RAS which is generally located at ostium or in proximal segment of artery. FMD is far less common but important because angioplasty has shown good results in hypertension secondary to FMD. The main limitation of CEMRA for the diagnosis of renal FMD has been its lack of spatial resolution and its inability to demonstrate the distal portion and small intrarenal branches of the main renal artery. The study by Willoteaux et al. revealed that CEMRI has sensitivity of 97% in detection of renal artery FMD (string of beads appearance); however, this study was biased because it was done retrospectively after obtaining definitive diagnosis by conventional angiography. Pitfalls/limitations of MRA are presence of stent, limited evaluation of intra-renal arterial branches due to poor spatial resolution and detection of accessory RA. MR is contraindicated in patients with cardiac implantable electronic device (CIED) like pacemakers, cochlear implants, metallic intraocular foreign bodies, drug infusion pumps and in claustrophobic patients. CTA is the investigation of choice for evaluation of RVH. It can quantify the severity of the occlusion accurately. It is quick and not limited by breath-hold capacity of the patients. It can assess the presence of accessory RA, other possible nonrenal causes of secondary hypertension (adrenal lesions, pheochromocytoma etc) as well as renal parenchymal diseases. The risk of radiation is minimal compared to the benefits offered by the modality. Several studies had established that CTA is more sensitive than MRA or colour Doppler for identification of renal vascular lesions. URINARY TRACT CALCIFICATIONS Jagneet Singh, Vasantha Kumar Venugopal Urinary tract calcifications include calcifications occurring within the lumen of excretory system, that is, renal, ureteric and urinary bladder calculi. In nephrocalcinosis, the calcification is occurring in the renal parenchyma, that is, medulla and cortex. Calcifications can also occur in renal parenchyma in renal tumours and infections like tuberculosis. Intracystic calcifications, calcification due to sloughed papilla and vascular calcifications are also seen. Primary amyloidosis is known to cause calcification in renal pelvis. Urinary bladder tumours can also show calcification. Mural calcifications in urinary bladder can be seen in schistosomiasis and tuberculosis. It is difficult to identify each type of calcification; however, some of the calcifications have specific pattern and appearance which can help us to find the aetiology and cause of calcification. Renal calculus is by far the most common cause of renal as well as urinary tract calcifications. This chapter deals with imaging approach for urinary tract calcifications primarily renal stones and also the brief description of specific features of other types of calcification. Urolithiasis causing ureteric colic is an important and frequent emergency condition in medical practice. Between 5% and 12% of the world population will have a urinary stone during their lifetime and recurrence rate is 50%. In the Indian scenario, the prevalence rate of kidney stones is 12%–15% and recurrence rate is again 50%. The following are the four main chemical types of renal calculi which together are associated with more than 20 underlying aetiologies: Table 10.4.2.1 The most common presenting complaints of urinary calculi include pain, haematuria or urinary tract infection. Small nonobstructive stones are usually silent. The passage of stones into the ureter can cause obstructive symptoms of colicky pain and vomiting. Large staghorn calculi are often relatively asymptomatic. It refers to the presence of a branched kidney stone occupying the renal pelvis and at least one calyceal system. They manifest as either haematuria or UTI rather than acute pain. In patients with nephrolithiasis, noncontrast CT or CT-KUB radiography is most often used. The ACR estimates the specificity of CT to be 97% when a patient presents with acute flank pain suspicious of an obstructing stone. The sensitivity of CT for detecting kidney stones is the highest of all the available modalities and reasonable estimates suggest it is ∼95%. All types of renal stones are usually seen on CT, rare exceptions include stones that are caused by the precipitation of protease-inhibitor medications in the urine. CT generates a 3D image of the stone and the surrounding anatomy, which can be reconstructed into multiple viewing planes. Few large stones are missed using CT but small stones (<3 mm) might slip between the imaged tissue planes and not be detected. The accuracy of CT is significantly better than that of ultrasonography. Standard CT is the imaging modality of choice for patients with a BMI >30 according to the ACR, AUA and EAU. A standard CT scan exposes patient to an effective dose of ionizing radiation of ∼10 mSv. Low-dose CT is used to reduce radiation exposure by lowering the tube current to the radiation source. Overall, CT is a highly sensitive and specific technique for imaging stones in patients presenting with renal colic in emergency, which is useful in assisting surgical decision making, owing to the superior anatomical detail obtained. The ACR and AUA both recommend CT as the first-line investigation for adult patients with symptoms suggestive of obstructive urolithiasis. When considering low-dose protocols, one should keep in mind patient’s age, BMI and degree of clinical suspicion of stones in order to choose the optimal imaging option. It requires additional images after administration of IV contrast. First scan is done after 90–100 s and second scan after 5–15 min of contrast administration. Unenhanced CT KUB has many advantages over Ct urography (CTU) for detection of urinary calculi with sensitivity ranging from 98% to 100% and specificity of 92%–100%. Due to administration of IV contrast, there is always risk of nephrotoxicity associated with CTU which is eliminated by the use of unenhanced CT without any compromise in the detection rate of calculi. Dual-energy CT can be used to differentiate between uric acid and nonuric acid stones. Further characterization of nonuric acid stones into cystine, struvite, calcium oxalate and hydroxyapatite stones can be done by applying additional tin filtration to the high-energy spectrum. Various colour coding systems are provided by different vendors for different calculi (Fig. 10.4.2.1). Accuracy of stone characterization is the highest for stones larger than 3–5 mm in diameter. Small stones have tendency to pass spontaneously, so this limitation may not be clinically significant. Another advantage of dual-energy CT in CTU is virtual nonenhanced images allowing detection of urinary calculi that would otherwise be obscured. The ACR and American Urological Association recommend CT evaluation as a first-line investigation of patients with suspected kidney stones, whereas the European Association of Urology recommends ultrasonography. Ultrasound has a low sensitivity of 45% for detection of ureteric and renal calculi and a specificity of 94% and 88% for the same. Ultrasonography can also be used to detect surrogate markers of obstructing stones such as hydronephrosis, and a lack of a ureteral jet using Doppler ultrasonography. Ultrasonography is very limited in its ability to detect small stones (<5 mm), due to partial volume effect or the absence of posterior shadowing. Stones in the mid ureters are also not detectable, due to masking by overlying intestinal loops and gas, especially in obese patients. The accuracy of diagnosing renal obstruction and stones can be improved by combining Doppler US and Colour Doppler for identifying secondary signs. An absent, asymmetric and/or reduced ureteric jet from the ureteric orifices usually suggests presence of ureteric obstruction. However, the contrary may not be true always, that is, presence of a positive ureteral jet does not rule out the presence of ureteral stones 14 since ureteral stones quite often only cause partial obstruction. A renal resistivity index (RI) >0.70 and/or a 10% difference between the kidneys is considered as diagnostic of obstructive uropathy. To summarize ultrasonography can reliably detect hydronephrosis but is less sensitive and specific than CT imaging for detecting and sizing of stones. It is recommended as the first-line imaging modality for pregnant and paediatric patients. The sensitivity of MRI for detection of renal or ureteric calculi is variable, can be augmented by hydro nephrosis. Using standard MRI sequences, stones appear as a nonspecific signal void; The sensitivity of MRI is 82% more than that of ultrasonography and X-ray but less than that of CT. A major advantage of MRI is that it provides 3D imaging without radiation. Unfortunately, the drawbacks of MRI like high cost, lower accuracy and longer image acquisition times prevent it from widespread use in stone imaging. The ACR, AUA and EAU guidelines suggest that MRI be used as a second-line modality when ultrasonography is nondiagnostic in pregnant and paediatric patients. This technique uses the same fundamental concepts as CT but in a single plane. Generally, 75% of stones are calcium-based and should be visible on plain film, only about 60% are found to be visible on plain films. Relatively low ionizing radiation exposure compared with CT (0.15 mSv) and low cost. It can aid in the detection of a calcified stone, determine its location and size, and provide an assessment of bowel gas patterns and faecal debris, stone’s progression through the urinary tract. As this imaging modality only views stones at one angle, accuracy is decreased yielding reduced sensitivity and specificity and, therefore, limiting its utility. Cystine and struvite stones often are poorly visible and uric acid and matrix stones are not visible at all. Small stones over a bony structure or those shadowed by bowel gas can be concealed. X-ray KUB in isolation is insufficient in detecting kidney stones and should always be paired with another imaging modality such as ultrasound. It has a sensitivity of 37.0% for stones <5 mm, but this increases to 87.5% for stones >5 mm. The AUA suggests use of this combined imaging approach for the evaluation of ureteral stone disease during stone passage or after treatment. If a stone has previously been shown to be radio-opaque, some clinicians advocate the use of KUB in following the case. Overall, KUB radiography is cost-effective compared with other modalities for monitoring stone size in stone formers who are receiving medical therapy. IVP is less sensitive than noncontrast helical CT. CT scanning with delayed contrast series and thin slices has reduced the need for IVP in most cases. The advantage of IVP:
10.4: Approach based algorithms
Introduction
Aetiology
Pathophysiology
When to clinically suspect RVH?
Do all suspects need imaging evaluation? american college of radiology (ACR) appropriateness criteria
RADIOLOGIC INVESTIGATION
MRA abdomen without and with IV contrast
Appropriate
CTA abdomen with IV contrast
Appropriate
US kidney retroperitoneal with duplex Doppler
Appropriate
MRA abdomen without IV contrast
Maybe Appropriate
Tc-99m ACE-inhibitor renography
Maybe Appropriate
Arteriography kidney
Not Appropriate
Venography with renal vein Sampling
Not Appropriate
RADIOLOGIC INVESTIGATION
US kidney retroperitoneal with duplex Doppler
Appropriate
MRA abdomen without IV contrast
Appropriate
CTA abdomen with IV contrast
Maybe Appropriate
MRA abdomen without and with IV contrast
Not Appropriate
99mTc ACE-inhibitor renography
Not Appropriate
Arteriography kidney
Not Appropriate
Venography with renal vein sampling
Not Appropriate
Radiology investigations
Noninvasive
Minimally Invasive
Tests Defining Structural Abnormality
Tests Defining Functional Abnormality
Colour doppler ultrasonography
Examination technique
Anterior approach demonstrates bilateral main renal arteries
Knobology
Normal doppler findings
Doppler criteria for diagnosis of RAS
ACE inhibitor renal scintigraphy
DIAGNOSTIC CRITERIA
MAG3
DTPA
Diagnostic criteria
MR angiography
CT angiography (CTA)
Introduction
Urolithiasis
Types of renal stone
Stone Type
Frequency (%)
Calcium
70–80
Calcium phosphate (pure)
5–10
Calcium oxalate/phosphate
30–45
Calcium oxalate (pure)
20–30
Struvite
15–20
Cystine
1–3
Uric Acid
5–10
Clinical presentation
Imaging
Noncontrast CT or CT-KUB
CT urography
Dual-energy CT
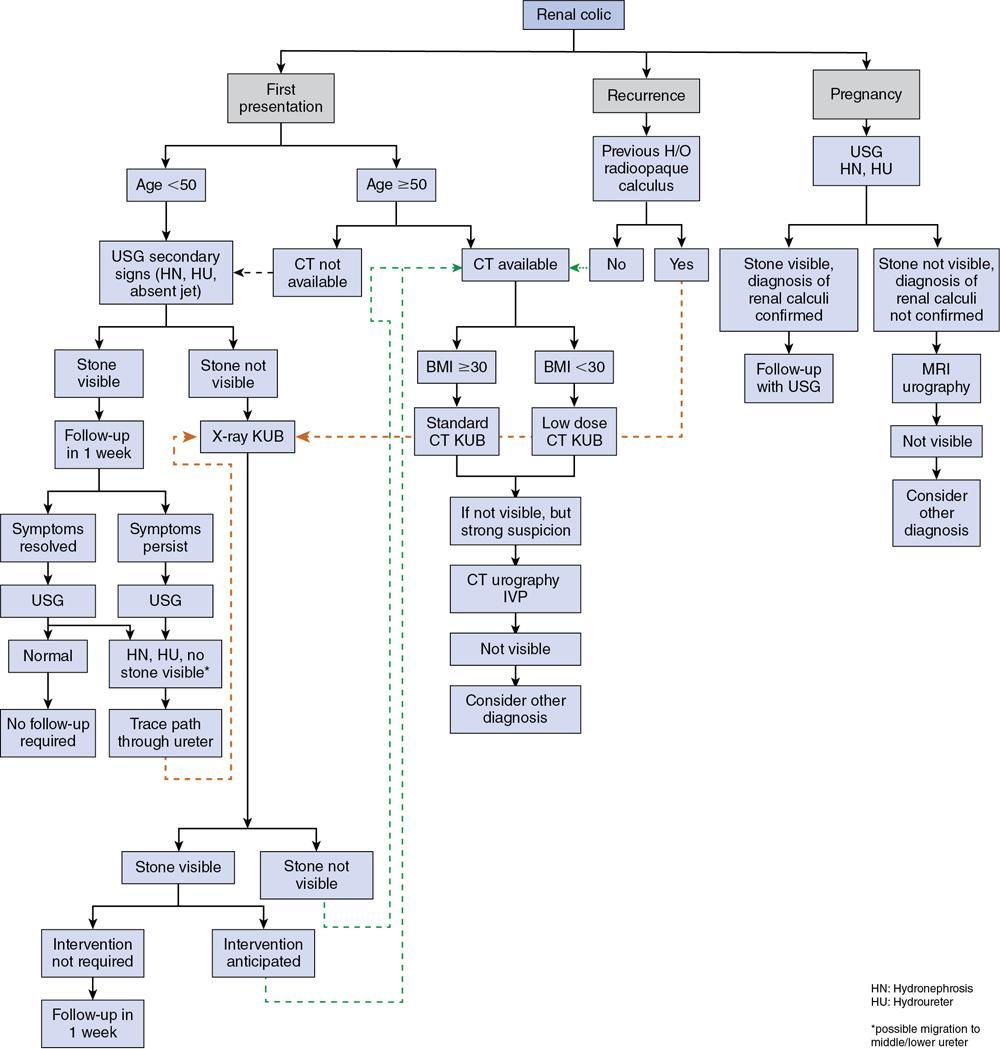
Ultrasonography
MRI
X-RAY: KUB
Advantages
Disadvantages
Intravenous pyelography
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








