Mammography
Mammography Techniques
Mammography is primarily a screening and not a diagnostic tool. The mediolateral oblique (MLO) and craniocaudal (CC) views are standard screening views, whereas additional views described below are used mainly for diagnostic evaluation of lesions.
Mammographic Views ( Fig. 9.1 )
MLO View
This standard view is a projection parallel to the pectoralis major muscle (C-arm of mammographic unit is 40–60 degrees). The pectoralis should be seen to the level of or below the axis of the nipple and appear convex (never concave toward the nipple).

CC View
Projection with slight rotation toward the sternum to detect posteromedial tumors that may be missed on the MLO view. In general, better breast compression is achieved with the CC view than with the MLO view.
Exaggerated Craniocaudal (Xccl) View
This view is done to evaluate the lateral tissue (axillary tail of Spence). The patient is asked to rotate so the radiograph holder can be placed at the midaxillary line.
Lateral Views: Mediolateral (ML and Lateromedial (Lm)
These views are a true lateral projection (x-ray beam parallel to floor). Used commonly to evaluate lesions for triangulation and needle localization. The direction of the x-ray beam is defined by the name of the view.
Axillary Tail View (Cleopatra View)
This view allows imaging of the axillary tail of the breast. It resembles the ML view but allows evaluation of breast tissue more laterally oriented.
Cleavage Valley View
Modified CC view that improves visualization of area between breasts. Both breasts are positioned on the detector.
Spot Compression Views
With or without microfocus magnification. For evaluation of margins and morphology of lesions. Spreads structures; useful to determine if densities are real or not.
Magnification Views
Provides additional information on margins, satellite lesions, and microcalcifications. Can also be useful for asymmetric tissue or architectural distortion.
Tangential View
Performed to demonstrate dermal location of lesions.
Rolled Views ( Fig. 9.2 )
- •
Roll breast laterally: superior lesion moves laterally
- •
Roll breast medially: superior lesion moves medially

Proper Positioning
Proper positioning is crucial for lesion detection. A cancer not imaged will not be detected. For both the CC and MLO views, the mobile border of the breast (CC: inferior border, MLO: lateral border) should be moved as far as possible toward the fixed border before placing the breast on the bucky. Check for correct positioning on CC and MLO views:
- •
Pectoralis muscle: On the MLO view, the pectoralis major should be convex anteriorly (never concave) and be seen to or below the level of the axis of the nipple. On the CC view, the muscle is seen approximately 35% of the time. The perpendicular distance from the nipple to the pectoralis on the MLO is used as a reference for adequacy of the CC view. The measurement on the CC view (taken as the distance from the nipple to the pectoralis or the back of the image) should be within 1 cm of the MLO measurement.
- •
The nipple should be in profile on at least one view. This may require an extra view in addition to the screening CC and MLO views.
- •
Retroglandular fat should usually be seen behind all fibroglandular tissue.
- •
Improper positioning on the MLO results in sagging, which is manifested by low nipple position and skin folds near the inframammary fold. The breast should be pulled up and out.
- •
Skin folds are usually not problematic in the axilla but can obscure lesions elsewhere. Repeat such views.
- •
Although the CC view is taken to include all of the medial breast tissue, exaggerated positioning is not desired. To check for this, make sure the nipple is near midline and not off to one side.
- •
On the MLO view, check for “cutoff” of inferior breast or axillary tissue resulting from placing the breast too low or too high on the bucky.
- •
Problems with compression or cutoff may be related to the image receptor size. Both 18- × 24-cm and 24- × 30-cm sizes are available. Too small a size results in cutoff. Too large a size can impair compression by impinging on other body parts.
- •
Motion is best detected by checking the septations located inferiorly and/or posteriorly or calcifications, which will be blurred by motion.
To better evaluate difficult areas:
- •
Outer breast → exaggerated CC lateral and Cleopatra (axillary tail) view
- •
Inner breast → exaggerated CC medial and cleavage (“valley”) view
- •
Retroareolar area → nipple in profile view
- •
Skin lesions: tangential views and skin localization procedure
Place calcifications in center of alphanumeric paddle
Place marker
Release compression and go tangential; calcifications should be right below marker
Compression
Compression should always be symmetric. Breast compression is used to reduce patient dose and improve image quality:
- •
Reduction of motion artifacts by immobilization of breast
- •
Reduction of geometric blur
- •
Reduction in change of radiographic density (achieve uniform breast thickness)
- •
Reduction of scattered radiation by decreasing breast thickness
Lesion Localization
Start with the view in which the lesion is best seen, and modify it.
- •
If a finding is seen only on CC view, ask for rolled CC views (top-rolled medially and laterally)
Lesion in superior breast will now project in the direction to which the top half of the breast was rolled (e.g., a superior lesion will move laterally if top half of breast is rolled laterally).
Lesion in inferior breast will move opposite the direction of the top half of the breast roll (e.g., an inferior lesion will move medially compared with its starting point in a top-rolled lateral CC).
- •
If a finding is seen only on MLO view, ask for straight lateral. This is a quick version of nipple triangulation.
Lesion in medial breast will move up on straight lateral (ML)
Lesion in lateral breast will move down on straight lateral (ML)
Mnemonic: M uffins (medial) rise, L ead (lateral) sinks
Triangulation ( Fig. 9.3 )
This technique allows localization of an unseen lesion on the CC view if it is visible only on the straight lateral and MLO views.
- 1.
Align straight lateral, oblique, and CC views from left to right.
- 2.
Nipple should be on a horizontal line.
- 3.
Connect the lesion on any two views by a straight line.
- 4.
Lesion should be located along path of the line on the third view
- 5.
When describing the location of a lesion, the breast is seen as the face of a clock, and the location in this plane is given as clock position ( Fig. 9.4 ). Depth is then indicated as anterior, middle, or posterior.

FIG. 9.4
- 6.
Additional descriptions are subareolar, central, and axillary tail areas.

Mammography Technical Standards
Viewing Conditions
Viewing condition requirements were originally developed for screen-film mammography, which has been largely replaced by full-field digital mammography (FFDM); however, requirements still apply if screen-film or hard copy digital images are used for comparison. Minimal requirements include:
- •
Adequate view box luminescence
- •
Low ambient light
- •
Masking of mammograms to exclude peripheral view box light
- •
Magnifying glass: each radiograph should be reviewed with a magnifying glass after initial inspection
Image Labeling
American College of Radiology (ACR) requirements:
- •
Markers identifying the view and laterality are required and are to be placed on the image near the axilla.
- •
An identification label must include the patient’s name (first and last), ID number, facility name and location, examination date, and technologist’s initials if not included elsewhere on the radiograph.
- •
Cassette number for screen-film and computed radiography images
- •
Mammography unit identification
Quality Control
ACR requirements:
- •
Daily: processor, darkroom cleanliness
- •
Weekly: screen cleanliness, view box
- •
Monthly: replenishment rates, phantom, visual checklist. Some mammographers advocate more frequent evaluation of phantom images because such images evaluate the entire imaging system.
- •
Quarterly: fixer retention, repeat/reject, light x-ray field alignment analysis
- •
Semiannually: darkroom fog, screen-film contact, compression, view box luminance
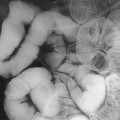
Mammography Interpretation ( Fig. 9.5 )
Each mammogram should be systematically evaluated for:
- •
Adequate quality of study; additional views required?
- •
Adequate penetration of fibroglandular breast tissue
- •
Breast composition/density pattern
- •
Skin, nipple, trabecular changes
- •
Presence of masses
- •
Calcifications
- •
Axillary nodes
- •
Asymmetry (usually a variant of normal)
- •
Architectural distortion

Comparison with previous examinations is essential. Breast cancers can grow slowly, and minimal progressive changes need to be documented. All masses and calcifications need to be further characterized. If the initial views are not adequate, additional views have to be obtained.
Pertinent Patient History
- •
Family history
- •
Risk factors for breast cancer
- •
Complaints
Mass, thickening
Pain
Nipple discharge
Mammography Reporting ( Fig. 9.6 )
The ACR Breast Imaging Reporting and Data System (BI-RADS) Atlas provides a lexicon to standardize breast imaging interpretation, reporting, and management recommendations. The following lexicon is excerpted from the BI-RADS Atlas 2013.

Breast Composition
BI-RADS 2013 promotes assignment of breast composition based on the overall volume of attenuating tissue and the likelihood that small lesions could be obscured. Composition is classified into four categories:
a = The breasts are almost entirely fatty.
b = There are scattered areas of fibroglandular density.
c = The breasts are heterogeneously dense, which may obscure small masses.
d = The breasts are extremely dense, which lowers the sensitivity of mammography.
Mass
A mass is a three-dimensional (3D) structure demonstrating convex outward borders seen on two orthogonal views. (Note that a potential mass seen on a single view only should be called an “asymmetry” until the structure is confirmed by additional imaging.) Masses should be described according to their margins , shape , and density .
Margins
- •
Circumscribed: well-defined borders (>75% circumference), uncommon sign of malignancy; only 2% of solitary masses with smooth margins are malignant.
- •
Obscured: margin cannot be seen or evaluated because of overlying normal tissue.
- •
Microlobulated: small lobulations, more worrisome for malignancy than larger lobulation
- •
Indistinct: a suspicious finding often seen in rapidly growing tumors that do not elicit significant fibrous tissue reaction. However, some benign lesions may also have indistinct margins:
Fat necrosis
Elastosis (refers to radial scar, indurative mastopathy, or sclerosing duct hyperplasia; elastosis is probably a form of sclerosing adenosis).
Infection/abscess
Hematomas
- •
Spiculated: a spiculated tumor margin is the only specific sign of malignancy; however, not all spiculated masses are cancers. Spiculations are also seen in:
Scar tissue
Desmoid tumors
Fat necrosis
Shape
Mass shape on mammography is categorized as round, oval, or irregular. (Note that lobular is no longer a shape descriptor in the BI-RADS 2013 Atlas.)
- •
Round: spherical
- •
Oval: elliptical/egg-shaped (may contain up to 2–3 undulations)
- •
Irregular: neither round nor oval, implies greater likelihood of malignancy
Density
The density of a mass is defined relative to the density of fibroglandular tissue, and is categorized as high density, equal density, low density, and fat-containing. Malignant lesions are usually very dense for their size. Fat-containing lesions are considered benign (posttraumatic oil cyst, lipoma, galactocele).
Other Features
- •
Size: the size of a mass does not correlate with likelihood of malignancy, however the larger the tumor the worse the prognosis. Malignant tumors >1 cm are twice as likely to have spread to axillary nodes.
- •
Location: identify quadrant, distinguish parenchymal mass from skin lesion
- •
Multiplicity: multiple, well-circumscribed masses in younger patients are commonly benign fibroadenomas or cysts. In older patients, metastases from other primaries should be excluded.
Calcifications
Fifty percent of all malignant tumors are discovered by mammography because of the presence of suspicious calcifications. In asymptomatic women, 75% of sampled clustered calcifications are benign and 25% are associated with cancer. Once detected, calcifications should be assessed on the basis of both morphology and distribution.
Morphology
Suspicious morphology ( Fig. 9.7 )
- •
Amorphous: hazy, too small to otherwise characterize shape
- •
Coarse heterogeneous: irregular calcifications between 0.5–1 mm in size, often coalescing. May be seen with both benign causes (involuting fibroadenoma, evolving dystrophic calcifications) or with malignancy.
- •
Fine pleomorphic: irregular, discrete shapes
- •
Fine linear/fine-linear branching: thin, irregular linear calcifications with or without branching configurations. Suggests ductal involvement of breast cancer.

Typically Benign Morphology ( Fig. 9.8 )
- •
Skin calcifications: pathognomonic lucent-centered shape, usually not requiring further workup. Typical locations include the inframammary fold, along the sternum, and around the nipple. If there is question about dermal location, a “skin localization” with tangential views can be useful.
- •
Vascular calcifications: parallel-tracks of calcifications clearly along a vessel. If seen in those younger than age 50, high correlation with coronary artery disease (CAD).
- •
Coarse “popcorn-like”: large (>2–3 mm) calcifications because of involuting fibroadenoma
- •
Large rod-like: also known as “secretory calcifications,” often seen in women >60 years. Continuous rods following a ductal distribution because of benign secretory disease (plasma cell mastitis) or duct ectasia.
- •
Round/punctate: considered benign when diffuse and small (<1 mm). If <0.5 mm, classified as punctuate. An isolated group of round/punctuate calcifications can be considered probably benign if no previous mammograms are available for comparison. If new or increasing, suspicious distribution, or associated with known cancer, biopsy must be performed.
- •
Rim: smooth, thin calcification in a spherical shape with lucent center because of fat necrosis and calcification in the walls of oil cysts.
- •
Dystrophic: irregular, large (>1 mm) calcifications often with lucent centers. Most commonly because of trauma, surgery, radiation.
- •
Milk of calcium ( Fig. 9.9 ): layering calcifications within microcysts or macrocysts. Classically demonstrate fuzzy, amorphous appearance on the CC view and semilunar or crescent-shaped on the lateral or MLO view.

FIG. 9.9
- •
Suture: calcifications deposited on surgical sutures. Typically linear or tubular, knot-shaped

Distribution
Distribution describes the arrangement of calcifications in the breast. The BI-RADS 2013 Atlas distribution categories are listed below in order of increasing likelihood of malignancy:
- •
Diffuse: randomly scattered throughout the breast (historically called “scattered”). Diffuse amorphous or punctuate calcifications are usually benign.
- •
Regional: used to describe calcifications that involve a large volume of breast tissue (>2 cm) not conforming to a ductal distribution. Malignancy unlikely.
- •
Grouped: used to describe few calcifications occupying a small volume of breast tissue, from five calcifications within 1 cm to a larger number of calcifications within 2 cm (historically called “cluster”). Raised suspicion for malignancy.
- •
Linear: arranged in a line, suggesting a ductal distribution and suspicious for malignancy. (Exceptions are vascular and large rod-like calcifications which have a linear distribution but benign morphology.)
- •
Segmental: ductal distribution involving multiple ducts and their branches, suspicious for extensive or multifocal breast cancer which may require bracketed localization.
Architectural Distortion
Architectural distortion refers to distorted breast parenchyma and appears as thin, straight lines radiating from a single focal point without an associated discrete mass. Architectural distortion may be due to trauma/surgery, high-risk lesions (e.g., radial scar), or malignancy. In the absence of history of previous trauma or surgery, architectural distortion should always be biopsied.
Asymmetries
Asymmetries are deposits of fibroglandular tissue without the convex borders of a mass. There are four types of asymmetry:
- •
Asymmetry: seen on only one view, usually because of summation artifact
- •
Focal asymmetry: small amount of tissue within a smaller area of the breast (less than one quadrant), seen in two views
- •
Global asymmetry: asymmetric tissue relative to the contralateral breast spanning at least one quadrant; may be normal variant
- •
Developing asymmetry: focal asymmetry that is new or enlarging, or more conspicuous compared with previous examinations. Suspicious for malignancy.
Skin, Nipple, and Trabecular Changes
- •
Skin retraction is due to fibrosis and shortening of Cooper ligaments (skin becomes flat or concave); the tumor itself is almost always palpable if skin retraction is present on the mammogram.
- •
Skin thickening (>3 mm) may be focal or diffuse; may be due to benign process (radiation, heart failure, mastitis) or malignancy (inflammatory and locally invasive breast cancer).
- •
Nipple retraction is worrisome when acute and unilateral.
- •
Fine-linear nipple calcification obliges one to rule out Paget disease (other causes of nipple calcification are benign).
Abnormal Ducts
Cancer may cause shortening, dilatation, or distortion of ducts.
- •
Ducts >2 mm in diameter extending >2 cm into the breast are usually due to benign ductal ectasia.
- •
Symmetric ductal ectasia is a benign condition.
- •
Asymmetric-appearing ducts are usually a normal variation, although this can be a very rare indication of malignancy.
- •
Papillomas and papillary cancers arise in main segmental ducts.
- •
Peripheral papillomas, epithelial hyperplasia, ductal carcinoma in situ (DCIS), and invasive ductal carcinoma arise in terminal ducts.
Lymph Node Abnormalities
Normal intramammary lymph nodes are usually visible only in the upper outer quadrant. Nodes may occasionally be seen below the medial plane. There have been rare reports of lymph nodes in the medial breast. An increase in size, number, or density of axillary lymph nodes is abnormal: axillary nodes >2 cm or intramammary nodes >1 cm without lucency or hilar notch are suspicious (if lucent fat center is present, even larger nodes may be benign). Nodes that contain tumor lose the radiolucent hilum and appear dense, although benign hyperplasia may appear similar. Nodal calcification implies:
- •
Metastasis (most common)
- •
Lymphoma
- •
Rheumatoid arthritis and previous gold injections
Asymmetry of Breast Tissue
Asymmetric, dense tissue is seen in 3% of breasts, usually in the upper outer quadrant and is considered a normal variant (caused by fibrosis). The mammographic finding of asymmetric breast tissue is suspicious only if it is palpable or if there are associated abnormalities (mass, calcifications, architectural distortion, or asymmetry that has developed over time). The following are the criteria that an opacity must fulfill to be called asymmetric tissue:
- •
Not a mass (i.e., changes morphology on different views)
- •
Contains fat
- •
No calcifications
- •
No architectural distortion
- •
If asymmetric tissue is palpable, ultrasound (US) may be useful for further workup.
Structured Reporting
Reports are organized by a short description of breast composition, description, and location of significant findings, as well as any interval changes and an overall impression. The ACR categorizes reports into seven categories according to BI-RADS:
0 = Needs additional mammographic evaluation and/or previous mammogram for comparison. This is almost always used in a screening situation. Category 0 should be used only for old examination comparison when such comparison is required to make a final assessment.
1 = Negative: return to annual screening
2 = Benign finding: return to annual screening. Includes characteristically benign lesions such as typical lymph nodes, calcified fibroadenomas, fat-containing lesions, and scattered benign calcifications.
3 = Probably benign (≤2% risk of malignancy): initial short-interval (6-month) follow-up suggested. Noncalcified circumscribed solid mass, focal asymmetry, and cluster of round/punctate calcifications are considered in this category.
4 = Suspicious abnormality (likelihood of malignancy >2% to <95%): Biopsy should be recommended. Can be further subdivided:
4a = Low suspicion for malignancy (>2% to ≤10%)
4b = Intermediate suspicion for malignancy (>10% to ≤50%)
4c = High suspicion but not highly suggestive for malignancy (>50% to ≤95%)
5 = Highly suggestive of malignancy (likelihood of cancer ≥95%): biopsy should be recommended.
6 = Known biopsy-proven malignancy: malignancy has been confirmed by biopsy but the patient has not undergone definitive therapy.
Ultrasound (US)
Indications
- •
Characterization of findings seen on mammography (e.g., differentiation of cyst vs. solid mass)
- •
Evaluation of palpable abnormalities
- •
Screening (currently limited use in the United States)
Interpretation
BI-RADS Lexicon
- •
Shape: oval, round, or irregular
- •
Margin: circumscribed, or not circumscribed (indistinct, angular, microlobulated, spiculated)
- •
Orientation of long-axis of lesion to chest wall: parallel or not parallel
- •
Echo pattern (relative to fat): anechoic, hypoechoic, hyperechoic, isoechoic, heterogeneous, or complex cystic and solid
- •
Posterior features: posterior acoustic enhancement, shadowing, or combined
- •
Calcifications: can be seen as echogenic foci with posterior shadowing within a mass, outside a mass, or intraductal (US usually cannot detect small microcalcifications)
- •
Associated features: architectural distortion, duct changes, skin changes, edema, vascularity, elasticity
- •
Ultrasound Appearance of Common Lesions
- •
Simple cysts are anechoic structures with thin, imperceptible walls and posterior acoustic enhancement.
- •
Complicated cysts are cysts containing internal echoes (debris).
- •
Cluster of microcysts are cluster of cysts <2–3 mm separated by thin septations.
- •
Fibroadenomas are usually oval, hypoechoic, and well circumscribed.
- •
Lipomas can be difficult to differentiate from surrounding tissue.
- •
Oil cysts are hypoechoic and have poor through-transmission. Correlate with mammography for confirmation.
- •
Skin lesions can be identified within the dermis; common skin lesions include sebaceous cyst/epidermal inclusion cyst, keloids, and neurofibromas.
- •
Benign lymph nodes have a characteristic reniform shape with echogenic center because of fat in the lymph node hilum. Loss of fatty hilum and/or cortical bulging suggests metastatic involvement.
- •
Malignant lesions tend to be taller than they are wide and have posterior acoustic shadowing.
Galactography
Indications
- •
Historically used for workup of solitary and spontaneous duct discharge (now largely replaced by magnetic resonance imaging)
- •
Identifies deep lesions that might be missed by surgery
- •
May be used to identify proximal lesions because papilloma and cancer have a similar appearance; take a biopsy to distinguish
- •
Multiple filling defects may be due to papillomatosis
Technique
- 1.
Patient sitting or lying down
- 2.
Express secretions to identify duct origins
- 3.
Prepare breast
- 4.
Blunt pediatric sialogram needle
- 5.
Inject 0.1–2 mL of water-soluble contrast agent
- 6.
Avoid air bubbles
- 7.
Obtain mammogram. Look for filling defects, distorted ducts, and/or extravasation.
Magnetic Resonance Imaging (MRI)
Indications
- •
High-risk screening
- •
Preoperative assessment to determine extent of disease
- •
Evaluation for recurrence or residual tumor after surgery
- •
Response to therapy
- •
Evaluation of postoperative scar versus recurrence
- •
Assessment of implant integrity
Advantages
- •
Can detect and characterize small lesions
- •
Can detect greater extent of disease than depicted by mammography/US
- •
Used effectively in dense breasts
- •
No radiation exposure
- •
Evaluation of axillary lymph nodes
Limitations
- •
Requires the use of a contrast agent.
- •
Higher cost
- •
Lower specificity
Technique
- •
Prone position
- •
Dedicated bilateral surface coils are usually receive-only coils but can be transmit/receive coils.
BI-RADS Lexicon
Fibroglandular Tissue (FGT)
FGT is best assessed on nonfat saturated sequence or fat-saturated T1-weighted sequence, categorized into four levels:
- a.
Almost entirely fat
- b.
Scattered fibroglandular tissue
- c.
Heterogeneous fibroglandular tissue
- d.
Extreme fibroglandular tissue
Background Parenchymal Enhancement (BPE)
BPE is enhancement of normal breast parenchyma and is assessed relative to the amount of fibroglandular tissue, categorized into four levels: minimal, mild, moderate, and marked.
Lesions
- •
Focus/foci: enhancement <5 mm that is too small to fully characterize
- •
Mass: 3D structure with convex borders. The following features should be assessed for masses:
Shape: round, oval, irregular
Margins: circumscribed or not circumscribed (irregular, spiculated)
Internal enhancement characteristics: homogeneous, heterogeneous, dark internal septations, and rim enhancement
- •
Nonmass enhancement (NME): enhancement pattern distinct from background parenchyma that is larger than a focus but lacks contour of a mass. NME is further characterized by its distribution and internal enhancement pattern.
Distribution
- •
Focal area: small area within one quadrant
- •
Linear: suggests involvement of single duct
- •
Segmental: cone-shaped with apex extending toward nipple, suggests involvement of large ductal system
- •
Regional: area broader than a ductal system spanning at least one quadrant
- •
Multiple regions: two or more regions separated by normal tissue
- •
Diffuse: scattered, symmetric
- •
Internal enhancement pattern: homogeneous, heterogeneous, clumped, or clustered ring
Enhancement Kinetics
Kinetic analysis depicts the uptake and washout of contrast following injection. Enhancement kinetics are described for two time points after injection:
- •
Initial upslope (first 2 min after injection): classified as slow (<50% increase in signal after injection), medium (50%–100% increase) or rapid (>100% increase).
- •
Delayed phase (after 2 min): classified as persistent (continued >10% increase in signal), plateau (signal intensity change is ≤10%), washout (intensity decreases >10%, most suspicious)
- •
Malignant tumors tend to have permeable tissue and thus demonstrate rapid upslope with plateau or washout on delayed phase (however, there is overlap between benign and malignant lesions).
Associated Features
Additional findings may be seen on MRI that raise suspicion for malignancy or may influence management and should be described:
- •
Nipple retraction
- •
Nipple invasion
- •
Skin thickening
- •
Skin invasion
- •
Chest wall invasion
MRI Features of Common Benign Lesions
- •
Intramammary lymph nodes are reniform shape and contain a fatty hilum.
- •
Duct ectasia is a common benign finding of intrinsic T1 hyperintensity within the ducts.
- •
Simple cysts are circumscribed T2 hyperintense structures with thin walls.
- •
Hematoma/seroma can contain intrinsic T1 hyperintensity due to blood products.
- •
Biopsy/surgical clips are seen as signal void from susceptibility artifact.
- •
Fat necrosis is often rim-enhancing and contains central fat, which can be confirmed on nonfat saturated sequences.
Evaluation of Implants
Technique
- •
MRI is used to assess integrity of silicone implants (saline implant rupture is a clinical diagnosis)
- •
Silicone has long T1 weight (T1W) and long T2 weight (T2W).
- •
Proton signal is from methyl groups in the dimethyl polysiloxane polymer.
- •
The silicone shell is of lower signal intensity than the silicone within the implant because of greater cross-linking of methyl groups.
- •
Fast spin-echo T2W, as well as orthogonal silicone-sensitive (fat-suppressed) inversion recovery sequences, are obtained. Chemical H 2 O suppression will yield a silicone-only image.
- •
MRI of implant rupture: 94% sensitivity, 97% specificity (compared with US: 70% sensitivity, 92% specificity)
Signs of Rupture (See Later Section on Implants in This Chapter)
- •
Intracapsular rupture: keyhole sign, linguine sign
- •
Extracapsular rupture: silicone seen outside implant capsule or within axillary lymph nodes
Breast Imaging Guided Procedures
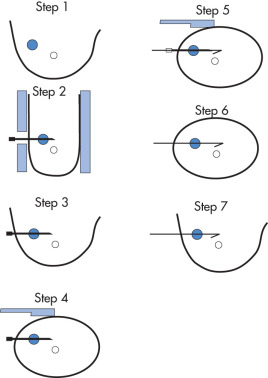
Mammographically Guided Wire Localization for Surgical Biopsy/Excision ( Fig. 9.10 )
- 1.
Identify lesion (use 90-degree radiographs to direct needle parallel to chest wall); choose shortest distance to lesion.
- 2.
Obtain view with breast in compression device; clean skin with iodine three times and then once with alcohol; pass needle tip in direction of x-ray beam past lesion.
- 3.
Obtain a second radiograph; if the needle is in good position, take a 90-degree opposed radiograph.
- 4.
If needle is in appropriate location, pass hook wire through needle. Pull needle back to engage hook; the wire may back out somewhat when patient stands up.
- 5.
Take a third mammogram perpendicular to wire with wire in place.
- 6.
A mammogram of the postbiopsy specimen should be obtained to ensure that the lesion is included in the specimen.