Chapter Outline
Esophageal carcinoma constitutes only about 1% of all cancers and 7% of cancers in the gastrointestinal (GI) tract. Nevertheless, it is a deadly disease, with an overall 5-year survival rate of only about 15%. At one time, most malignant tumors of the esophagus were thought to be squamous cell carcinomas, but adenocarcinomas arising in Barrett’s esophagus have increased dramatically in incidence since the 1970s. Because of important differences between these tumors, the chapter is divided into separate sections on squamous cell carcinoma and adenocarcinoma of the esophagus.
Squamous Cell Carcinoma
Epidemiology
Esophageal carcinoma is predominantly a disease of older men, with a male-to-female ratio of almost 4 : 1 and a peak incidence between 65 and 74 years of age. The development of squamous cell carcinoma of the esophagus is a multifactorial process associated with a variety of risk factors, including tobacco and alcohol consumption, obesity, nutritional deficiencies, exposure to various environmental carcinogens, and geographic location.
Two of the major risk factors for the development of esophageal cancer in the United States are tobacco and alcohol consumption. Tobacco and alcohol appear to have a synergistic effect, so people who smoke and drink have even higher rates of esophageal cancer. Although tobacco smoke is known to contain a variety of carcinogens, the development of esophageal carcinoma in alcoholics may be related to other factors such as poor health and nutritional deficiencies. Obesity also has been recognized as an important risk factor for the development of esophageal cancer.
Squamous cell carcinoma of the esophagus has striking geographic variations, with the highest incidences reported in an Asian esophageal cancer belt stretching from eastern Turkey and northern Iran to India and northern China. A high incidence has also been reported in South Africa and France. These regional variations in the frequency of esophageal cancer have been attributed primarily to environmental rather than hereditary factors. Dietary habits in particular have been implicated because people living in areas with a high incidence of esophageal cancer often have diets high in starch and low in fresh fruits and vegetables. Other environmental factors may also have a role in cancer pathogenesis. For example, nitrosamines and other nitroso compounds are potent carcinogens that occur in high concentrations in the food and water supply of parts of northern China. Epidemiologic studies in China and South Africa have shown that these areas also have unusually low levels of molybdenum in the soil. Because molybdenum is required for the metabolism of nitrite to ammonia, low levels of molybdenum in the soil could lead to the accumulation of nitrites and potentially carcinogenic nitrosamines in plants consumed by humans. The high prevalence of esophageal cancer in parts of Saudi Arabia has been attributed to contamination of drinking water by impurities such as petroleum oils. Other substances such as tannin, betel leaves, and asbestos fibers have also been implicated in the development of esophageal cancer.
Attention has also been focused on the potential role of human papillomavirus (HPV) in the pathogenesis of esophageal cancer, particularly in high-risk areas such as China and South Africa. In studies from China, HPV has been isolated by in situ hybridization techniques in 25% to 50% of esophageal cancer specimens. These data suggest that HPV may be an important contributing factor in the development of esophageal cancer.
Predisposing Conditions
Conditions that are thought to predispose patients to the development of squamous cell carcinoma of the esophagus include achalasia, lye strictures, head and neck tumors, celiac disease, Plummer-Vinson syndrome, radiation, and tylosis. Because of the increased risk of developing esophageal cancer, periodic surveillance has often been advocated for patients with these conditions.
Achalasia
Achalasia is thought to be a premalignant condition associated with an increased risk of developing esophageal carcinoma. In various studies, the prevalence of esophageal cancer in patients with long-standing achalasia has ranged from 2% to 8%. Malignant degeneration presumably occurs as a result of chronic stasis esophagitis caused by retained food and debris in a dilated, obstructed esophagus. Most patients have had achalasia for at least 20 years before the development of cancer. Unfortunately, a neoplastic lesion growing inside a massively dilated esophagus may not cause symptoms until it is an advanced, unresectable tumor. As a result, some authors believe that patients with long-standing achalasia should undergo annual surveillance with barium studies or endoscopy to detect developing cancers at the earliest possible stage. However, one study failed to show an increased cancer risk in these patients, so not all investigators accept the need for surveillance.
Lye Strictures
Patients with chronic lye strictures have an increased risk of developing esophageal carcinoma. In various studies, the prevalence of cancer has ranged from 2% to 16%. Although the pathogenesis is uncertain, it has been postulated that chronic inflammation and scarring from caustic esophagitis predispose these patients to the development of esophageal carcinoma. The average latent period between the ingestion of lye and development of cancer is 40 to 45 years. These patients usually seek medical attention for recurrent or suddenly worsening dysphagia many years after lye ingestion. Carcinomas arising in lye strictures have a better prognosis than most esophageal cancers, with 5-year survival rates of 8% to 33%. This more favorable prognosis may be related to the presence of dense scar tissue surrounding the tumor, which prevents early invasion of adjacent mediastinal structures. Some investigators advocate periodic surveillance of patients with long-standing lye strictures, but these individuals are often in a socioeconomic group least likely to be compliant with surveillance programs.
Head and Neck Tumors
Patients with primary squamous cell carcinomas of the oral cavity, pharynx, and larynx have a substantially increased risk of developing separate primary esophageal carcinomas. In various studies, 2% to 8% of patients with head and neck tumors who underwent endoscopic surveillance were found to have synchronous esophageal cancers. This association has been attributed to common predisposing factors, primarily smoking and drinking because exposure to tobacco and alcohol considerably increases the risk of squamous cell carcinoma in both areas. Radiologic or endoscopic evaluation of the esophagus has therefore been advocated in the initial work-up of all patients with head and neck tumors. Many of these synchronous esophageal cancers are small, asymptomatic lesions, so screening tests may detect such tumors at an early stage, when they are potentially curable. Discovery of an advanced lesion in the esophagus is also important because radical head and neck surgery may no longer be appropriate in these patients. The risk of developing subsequent metachronous esophageal carcinomas is also considerably increased in patients with head and neck tumors. Some form of ongoing surveillance is therefore required to detect metachronous esophageal lesions.
Celiac Disease
Celiac disease (nontropical sprue) is thought to be associated with an increased risk of developing esophageal carcinoma. The pathogenesis of cancer is uncertain, but it has been postulated that carcinogens are absorbed through an atrophic jejunal mucosa in patients with advanced celiac disease. Most patients have long-standing disease; malabsorption is present for an average of 35 years before the development of cancer. Some investigators therefore advocate radiologic or endoscopic surveillance of the esophagus in these patients.
Plummer-Vinson Syndrome
Plummer-Vinson, or Paterson-Kelly, syndrome is characterized by iron deficiency anemia, glossitis, postcricoid webs, and dysphagia. This syndrome has been described primarily in women of Scandinavian origin. The prevalence of hypopharyngeal or esophageal carcinoma in Plummer-Vinson syndrome has ranged from 4% to 16%. Almost all such cancers are associated with postcricoid webs. Radiologic or endoscopic examinations are therefore required to differentiate webs from superimposed hypopharyngeal or esophageal carcinomas in these patients.
Radiation
Esophageal cancer is a rare complication of chronic radiation injury to the esophagus. Most cases have occurred in the cervical or upper thoracic esophagus after radiation doses of 20 to 50 Gy to the mediastinum or neck. In one study, women who received radiation therapy for carcinoma of the breast were found to have an increased risk of esophageal carcinoma. In general, however, the average latent period between radiation therapy and the development of cancer is about 30 years. It is therefore difficult to prove that these lesions are not coincidental cancers arising in a previously irradiated area.
Tylosis
Tylosis (Howel-Evans syndrome) is an extremely rare, hereditary, autosomal dominant disorder characterized by hyperkeratosis of the palms and soles, with thickening and fissuring of the skin. This disorder is associated with an extraordinarily high risk of developing esophageal cancer. In one study, 95% of patients with tylosis had esophageal cancer by 65 years of age. Most of these patients are found to have advanced, unresectable tumors at the time of clinical presentation. However, asymptomatic individuals with tylosis may have hyperkeratotic esophageal plaques containing foci of dysplasia, intramucosal carcinoma, or invasive carcinoma. Thus, periodic surveillance of asymptomatic family members has been advocated to detect premalignant lesions before the development of overt carcinoma. Because of the high likelihood of developing esophageal cancer, a prophylactic esophagectomy is sometimes justified for these patients.
Pathology
Gross Features
Squamous cell carcinomas of the esophagus may appear grossly as infiltrating, polypoid, ulcerative, or superficial spreading lesions. Infiltrating lesions, the most common type, cause irregular narrowing and constriction of the lumen. Polypoid lesions are lobulated or fungating masses that protrude into the lumen. Primary ulcerative lesions are relatively flat masses in which the bulk of the tumor is necrotic and ulcerated. Less frequently, superficial spreading lesions may extend longitudinally in the wall, without invading beyond the mucosa or submucosa. Patients with superficial spreading carcinomas tend to have a better prognosis than those with more invasive forms of esophageal cancer.
Histologic Features
About 50% of esophageal cancers are squamous cell carcinomas, and the remaining 50% are adenocarcinomas arising in Barrett’s mucosa. Other less common malignant tumors of the esophagus are discussed in Chapter 24 .
At the time of clinical presentation, most squamous cell carcinomas of the esophagus are advanced lesions that have already invaded regional lymph nodes or other local or distant structures. As a result, affected individuals have a dismal prognosis, with overall 5-year survival rates of only about 15%. In contrast, early esophageal cancers are relatively curable lesions, with 5-year survival rates of more than 90%. According to the Japanese Society for Esophageal Diseases, early esophageal cancer is defined histologically as cancer limited to the mucosa or submucosa without lymph node involvement. Many of these cases have been reported in the Chinese literature as a result of mass screening of the adult population because of the high incidence of esophageal cancer in that country.
Considerable confusion exists in the literature regarding the terminology for “early” cancer. The terms early esophageal cancer, superficial esophageal cancer, and small esophageal cancer have been used interchangeably to describe malignant esophageal tumors diagnosed at an early stage. However, these lesions should not be considered synonymous because they have different histopathologic features that alter the prognosis of this disease. According to the Japanese Society for Esophageal Diseases, superficial esophageal cancer is confined to the mucosa or submucosa but, unlike patients with early esophageal cancer, patients with superficial cancers may have lymph node metastases. Small esophageal cancer is another term used to describe tumors smaller than 3.5 cm, regardless of the depth of invasion or the presence or absence of lymph node metastases. Previous studies have shown that the 5-year survival for esophageal cancer decreases markedly when regional lymph nodes are involved by tumor. Thus, some superficial or small esophageal cancers may be early lesions histologically, whereas others may have already invaded regional lymph nodes, with a prognosis comparable to that of advanced esophageal cancer.
Distribution
Squamous cell carcinomas of the esophagus tend to be located in the upper, middle or, less commonly, distal third of the esophagus. Unlike adenocarcinomas arising in Barrett’s mucosa, squamous cell carcinomas of the distal esophagus almost never invade the stomach, and there is usually a discrete segment of normal esophagus between the tumor and gastric cardia.
Routes of Spread
Esophageal carcinoma may invade local, regional, or distant structures by various pathways, including direct extension, lymphatic spread, and hematogenous metastases.
Direct Extension
Because the esophagus lacks a serosa and is attached to neighboring structures by only a loose adventitia, there is no anatomic barrier to prevent rapid spread of tumor into the adjacent mediastinum. As a result, esophageal cancer has a marked tendency to invade contiguous structures in the neck or chest, such as the thyroid, larynx, trachea, bronchi (usually the left main bronchus), aorta, thoracic duct, lung, pericardium, and diaphragm. The tracheobronchial tree is a particularly common site of involvement; tracheoesophageal or esophagobronchial fistulas develop in 5% to 10% of all patients with esophageal cancer. Rarely, aortoesophageal fistulas or even esophagopericardial fistulas may occur as a terminal complication of esophageal cancer caused by aortic or pericardial invasion by tumor.
Lymphatic Spread
Lymphatic metastases are found in up to 75% of patients with esophageal cancer. Because the esophagus contains a rich network of interconnecting lymphatic channels, lymphatic spread from esophageal cancer is unpredictable, with jump metastases to lymph nodes in the neck or mediastinum often occurring in the absence of segmental lymph node involvement. Submucosal esophageal lymphatics also communicate subdiaphragmatically with paracardiac, lesser curvature, and celiac nodes in the upper abdomen; these nodal groups are involved by tumor in 25% to 50% of patients with esophageal cancer. Although tumors in the distal esophagus are more likely to metastasize to the abdomen, lymphatic spread of cancers in the upper or midesophagus can also result in metastases to celiac or other abdominal lymph nodes.
Discrete lymphatic metastases or satellite nodules are found in the esophagus at autopsy in about 50% of patients with esophageal cancer. These lesions should be distinguished pathologically from rare double primary carcinomas of the esophagus. However, it may be impossible to determine whether two discrete lesions represent synchronous primary tumors or a single cancer with lymphatic dissemination.
Between 2% and 15% of patients dying of esophageal cancer have gastric metastases at autopsy. These lesions probably result from tumor emboli that seed the gastric fundus via submucosal esophageal lymphatics extending subdiaphragmatically to the stomach. In such cases, the primary esophageal cancer may be located a considerable distance from the gastroesophageal junction, with a normal esophageal segment below the lesion.
Hematogenous Metastases
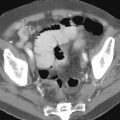
Hematogenous (bloodborne) metastases are often found in patients with advanced esophageal carcinoma. The most common sites of metastases are the lungs, liver, adrenals, kidneys, pancreas, peritoneum, and bones.
Clinical Aspects
Most patients with esophageal cancer develop dysphagia only when the lumen of the esophagus has been reduced by 50% to 75% of its normal size. By that time, malignant invasion of periesophageal lymph nodes or surrounding mediastinal structures has usually occurred. As a result, most patients have advanced, unresectable tumors at the time of diagnosis. Occasionally, however, patients experience dysphagia while the tumor is at an early stage. It is therefore possible to detect early esophageal cancer in some patients who are symptomatic.
Dysphagia is the most common complaint in patients with advanced esophageal cancer. Dysphagia is usually present for 2 to 4 months before these patients seek medical attention. Some patients can accurately localize the level of obstruction, but others may have a sensation of blockage referred to the thoracic inlet or even the pharynx by a cancer arising in the middle or lower thoracic esophagus. The esophagus should therefore be carefully evaluated in all patients with unexplained pharyngeal dysphagia to rule out an esophageal cancer below the subjective site of obstruction.
Patients with esophageal cancer may also present with odynophagia if the tumor is ulcerated or with substernal chest pain unrelated to swallowing if the tumor has invaded the mediastinum, so unremitting chest pain is an extremely poor prognostic sign. Other common symptoms include anorexia and weight loss, which are present in up to 75% of cases. Some patients may present with guaiac-positive stool or iron deficiency anemia as a result of occult bleeding from the friable surface of the tumor. However, frank hematemesis is uncommon. Rarely, fatal hemorrhage may be caused by an aortoesophageal fistula. The latter patients may have minimal hematemesis before the sudden development of massive hemorrhage, shock, and death.
Other patients with esophageal cancer may develop hoarseness because of direct extension of tumor into the larynx or involvement of the recurrent laryngeal nerve. Recurrent aspiration may lead to a chronic cough. However, the presence of a paroxysmal cough on swallowing should suggest the development of a malignant tracheoesophageal or esophagobronchial fistula. Rarely, patients with esophageal cancer may have anorexia, weight loss, or other signs of widespread malignancy without experiencing dysphagia, so localizing esophageal symptoms are not always present.
Endoscopic Findings
When multiple biopsy specimens are obtained, endoscopy has an overall sensitivity of almost 100% in the diagnosis of esophageal carcinoma. Brush cytology is also helpful when the esophageal lumen is so compromised by tumor that adequate biopsy specimens cannot be obtained. Detection of suspicious lesions on barium studies should therefore lead to early endoscopy and biopsy for a definitive diagnosis.
Radiographic Findings
Early Esophageal Cancer
Double-contrast esophagography has been widely advocated as the best radiologic technique for diagnosing early esophageal cancer. Unfortunately, the increased sensitivity of this technique has resulted in a lower specificity because more subtle abnormalities are suspected of representing cancer. Nevertheless, it is probably best to accept a certain percentage of false-positive findings to avoid missing early tumors. When a lesion is detected on barium studies, endoscopy and biopsy should therefore be performed to confirm the presence of carcinoma.
Early esophageal cancers classically appear on double-contrast esophagograms as small protruded lesions less than 3.5 cm in diameter. They may be plaquelike lesions, often with central ulceration ( Fig. 23-1 ), or small sessile polyps with a smooth or slightly lobulated contour ( Fig. 23-2 ). Other early cancers may be superficial or depressed lesions, causing focal irregularity, nodularity, or ulceration of the mucosa ( Fig. 23-3 ). When a lesion is detected on double-contrast studies, multiple projections should be obtained to determine its appearance en face and in profile (see Fig. 23-1 ).



Although most early esophageal cancers appear as focal lesions, superficial spreading carcinomas may be manifested by poorly defined nodules or plaques, producing a confluent area of mucosal nodularity or granularity ( Fig. 23-4 ). Some superficial spreading carcinomas may be localized lesions, whereas others may involve a considerable segment of the esophagus.

Early esophageal cancers are generally thought to be small lesions, but some early cancers may appear on barium studies as relatively large intraluminal mass larger than 3.5 cm in diameter ( Fig. 23-5 ). Such lesions may be indistinguishable from advanced carcinomas. Thus, early esophageal cancers are not necessarily small cancers because they may undergo considerable intraluminal or intramural growth and still be classified histologically as early lesions.

Advanced Carcinoma
Chest Radiography.
Almost 50% of patients with advanced esophageal cancers have abnormal chest radiographs. The most common findings include mediastinal widening, a hilar, retrohilar, or retrocardiac mass, anterior tracheal bowing, a widened retrotracheal stripe, and an air-fluid level in the esophagus ( Fig. 23-6 ). Anterior bowing of the trachea or thickening of the retrotracheal stripe beyond 3 mm in width may result from lymphatic infiltration or direct invasion of the retrotracheal area by tumor. Obstructing cancers may be recognized by an air-fluid level in the esophagus. However, esophageal obstruction from achalasia or other causes may also be manifested by anterior tracheal bowing or an esophageal air-fluid level.

Barium Studies.
Double-contrast esophagography is often performed on patients with dysphagia to rule out carcinoma or other abnormalities in the esophagus. Some gastroenterologists believe that endoscopy is required for all patients with negative esophagograms to find tumors that are missed on barium studies. In a large series of patients with esophageal cancer, however, the lesion was detected on double-contrast esophagograms in 98% of cases, and malignant tumor was diagnosed or suspected on the basis of the radiographic findings in 96%. An argument could be made that a high sensitivity is achieved in the radiographic diagnosis of esophageal cancer only by exposing an inordinate number of patients to unnecessary endoscopy. In the previous study, however, endoscopy was recommended to rule out malignant tumor in only about 1% of all patients who underwent double-contrast examinations. Similarly, in other series, endoscopy has failed to reveal any cases of esophageal carcinoma that were missed on double-contrast esophagograms. Thus, endoscopy is not routinely warranted for patients who have normal findings on barium studies.
Advanced esophageal carcinomas may appear on barium studies as infiltrating, polypoid, ulcerative, or varicoid lesions ( Fig. 23-7 ). However, many esophageal cancers have mixed morphologic features, so there is considerable overlap in the classification of these tumors.

Infiltrating esophageal carcinomas are characterized by irregular narrowing and constriction of the lumen associated with a nodular or ulcerated mucosa and abrupt, well-defined proximal and distal margins (see Fig. 23-7A ). Occasionally, these cancers have shelflike, overhanging margins, producing true annular lesions ( Fig. 23-8A ). However, other infiltrating lesions have more gradual, tapered margins, occasionally mimicking the appearance of benign strictures ( Fig. 23-8B ). Infiltrating lesions may eventually cause partial or even complete esophageal obstruction, with proximal dilation and minimal or no emptying of barium into the stomach.

Polypoid carcinomas appear as lobulated or fungating intraluminal masses, usually larger than 3.5 cm (see Fig. 23-7B ). They often contain areas of ulceration caused by tumor necrosis. Bulky lesions may eventually cause luminal encroachment and obstruction. However, squamous cell carcinomas are not generally polypoid, so other malignant tumors such as spindle cell carcinoma should be considered in these patients (see Chapter 24 ).
Primary ulcerative carcinomas are those in which the bulk of the tumor mass is replaced by ulceration. When viewed in profile, these lesions appear as well-defined meniscoid ulcers, with a thick, irregular, radiolucent rim of tumor surrounding the ulcer (see Fig. 23-7C ).
Varicoid carcinomas are those in which submucosal spread of tumor results in thickened, tortuous, or serpiginous longitudinal folds, mimicking the appearance of esophageal varices (see Fig. 23-7D ). However, these entities can usually be differentiated at fluoroscopy (see later, “ Differential Diagnosis ”). Although varicoid carcinomas are uncommon, submucosal extension of tumor not infrequently produces a focal varicoid pattern adjacent to an obvious squamous cell carcinoma. In one study, a varicoid pattern was seen on esophagography in 40% of patients with esophageal cancer.
Mediastinal involvement by advanced esophageal carcinoma can also be recognized on barium studies. Lymphatic spread of tumor to paratracheal, subcarinal, or paraesophageal lymph nodes may lead to extrinsic compression or displacement of the esophagus, often at a considerable distance from the primary lesion ( Fig. 23-9 ). Mediastinal lymphadenopathy is usually characterized by a smooth, extrinsic esophageal impression with gently sloping, obtuse borders. This finding almost always indicates an advanced, unresectable lesion.

Lymphatic metastases from esophageal cancer may be manifested by discrete implants adjacent to or remote from the primary lesion. These metastases may appear on barium studies as small plaquelike, polypoid, submucosal, or ulcerated lesions separated from the main tumor by normal intervening mucosa ( Fig. 23-10 ). Although most of these satellite lesions represent lymphatic metastases from the original cancer, the possibility of two primary carcinomas (double primaries) should be considered when the lesions are separated by an unusually long segment of normal mucosa.

Squamous cell metastases to the stomach usually appear on barium studies as solitary, large submucosal masses in the gastric fundus. These lesions often contain areas of ulceration, so they may resemble ulcerated gastrointestinal stromal tumors (GISTs; Fig. 23-11 ). Less frequently, they can be mistaken for primary gastric carcinomas. Because the appropriate treatment for esophageal cancer depends on the stage of the tumor, the gastric cardia and fundus should be carefully examined radiographically in all patients with esophageal cancer to rule out unsuspected metastases to the stomach.

Five percent to 10% of patients with esophageal cancer develop an esophageal-airway fistula ( Fig. 23-12 ). This complication frequently occurs after radiation therapy, probably as a result of radiation-induced tumor necrosis. Most such fistulas involve the trachea or left main bronchus. Occasionally, however, a locally aggressive esophageal cancer may lead to the development of a necrotic, tumor-containing cavity in the mediastinum or lung that communicates directly with the esophagus ( Fig. 23-13 ). When an esophageal-airway fistula is suspected, the esophagogram should be performed with barium rather than water-soluble contrast agents because the latter agents are hyperosmolar and may draw fluid into the lungs if a fistula is present, causing severe pulmonary edema.


Esophageal-airway fistulas are usually recognized on esophagography by the presence of barium in the bronchi or distal trachea. In many cases, the origin of the fistulous track is identified within an obvious infiltrating carcinoma (see Fig. 23-12B ). Once barium has entered the trachea or left main bronchus, however, it can be coughed up into the proximal trachea or larynx, so delayed overhead radiographs may erroneously suggest tracheobronchial aspiration. When an esophageal-airway fistula is suspected, the initial swallow should therefore be performed in a lateral projection with a video recording of the hypopharynx to differentiate a fistula from aspiration.
Rarely, patients with advanced esophageal cancer may develop esophagopericardial fistulas, manifested by pneumopericardium on chest radiographs or computed tomography (CT) scans. In such cases, esophagography with water-soluble contrast agents may demonstrate esophageal perforation at the site of the tumor, with contrast material entering the pericardial sac.
Associated Conditions
Achalasia.
Esophageal carcinomas arising in patients with achalasia usually appear on barium studies as polypoid masses in the middle or, less commonly, distal third of the esophagus ( Fig. 23-14 ). Because these lesions often develop in a massively dilated esophagus, they can reach an enormous size, producing bulky intraluminal masses that have a fungating or cauliflower appearance. Nevertheless, the lesions may be obscured by retained fluid and debris in a dilated, partially obstructed esophagus. Careful esophageal lavage with a soft rubber catheter may therefore be required to cleanse the esophagus before performing the radiologic examination.

Lye Strictures.
The development of a lye-induced cancer may be manifested on barium studies by increasing stenosis, mass effect, nodularity, and/or ulceration within a preexisting lye stricture ( Fig. 23-15 ). The underlying strictures are often located in the region of the tracheal bifurcation, so lye cancers may be complicated by the development of tracheoesophageal or esophagobronchial fistulas (see Fig. 23-15B ). Any change in the appearance of a chronic lye stricture should therefore be evaluated by endoscopy with multiple biopsy specimens and brushings to rule out a superimposed carcinoma.

Tylosis.
When patients with tylosis develop esophageal symptoms, they usually have advanced esophageal carcinomas, manifested on barium studies by annular, infiltrating, or plaquelike lesions. Occasionally, however, asymptomatic patients with tylosis may have discrete hyperkeratotic plaques containing one or more foci of dysplasia or intramucosal carcinoma. These lesions may appear on esophagograms as large, well-defined plaques, with normal intervening mucosa ( Fig. 23-16 ).

Adenocarcinoma
Primary esophageal adenocarcinomas are almost always found to arise in the setting of Barrett’s esophagus. In the past, such tumors were thought to be rare lesions, accounting for less than 5% of all esophageal cancers. However, many of these adenocarcinomas involving the gastroesophageal junction or gastric fundus were incorrectly classified as primary gastric carcinomas secondarily invading the lower end of the esophagus. Apart from difficulties in tumor classification, the incidence of esophageal adenocarcinoma in white men has increased by 300% to 500% in the past 30 years, and this tumor is the fastest growing cause of cancer mortality. Currently, adenocarcinomas are thought to constitute at least 50% of all esophageal cancers. Nevertheless, many questions remain about the risk of malignant degeneration in Barrett’s esophagus and the long-term management of patients with this condition.
Epidemiology and Pathogenesis
Considerable attention has been focused on the relationship between esophageal adenocarcinoma and the columnar epithelium–lined esophagus, or Barrett’s esophagus. In various studies, 90% to 100% of all primary esophageal adenocarcinomas have been found to arise on a background of Barrett’s mucosa. Thus, adenocarcinoma of the esophagus is not only more common than was previously recognized, but the vast majority of these tumors appear to result from malignant degeneration in Barrett’s esophagus.
Barrett’s Esophagus
Barrett’s esophagus is a well-recognized condition in which there is progressive columnar metaplasia of the distal esophagus caused by long-standing gastroesophageal reflux and reflux esophagitis. In various studies, the prevalence of Barrett’s esophagus in patients with reflux esophagitis has ranged from 5% to 15%, with an overall prevalence of about 10%. In a widely quoted study, Barrett’s esophagus was found to occur in 4% of patients undergoing upper gastrointestinal endoscopy and in 9% of men older than 50 years. The clinical and radiologic aspects of Barrett’s esophagus are presented in Chapter 19 .
Various types of columnar epithelium have been described in Barrett’s esophagus, including a junctional-type epithelium, a gastric fundus–type epithelium, and an intestinal-type or specialized columnar epithelium. It is believed, however, that intestinal metaplasia is the major prerequisite for a pathologic diagnosis of Barrett’s esophagus. This intestinal metaplasia is characterized histologically by goblet cells with acidic mucin and, in some cases, enterocyte differentiation with brush border formation. The revised definition of Barrett’s esophagus is based on an emerging consensus that intestinal metaplasia represents the type of epithelium that usually predisposes these patients to the development of esophageal adenocarcinoma.
Investigators have also developed histopathologic criteria for Barrett’s esophagus in which patients are classified as having long-segment disease (i.e., extending more than 3 cm from the gastroesophageal junction) or short-segment disease (i.e., extending 3 cm or less from the gastroesophageal junction) based on the vertical extent of columnar metaplasia in the esophagus. Short-segment Barrett’s esophagus is even more common than long-segment Barrett’s esophagus, with a reported prevalence of 10% to 15% at endoscopy.
Risk of Adenocarcinoma.
In various series, the prevalence of adenocarcinoma in patients with Barrett’s esophagus has ranged from 2% to 46%, with an overall prevalence of about 10%. *
* See .
It should be recognized that prevalence data exaggerate the risk of cancer because many patients with Barrett’s esophagus remain asymptomatic until the development of a superimposed carcinoma. Nevertheless, it has been estimated that patients with Barrett’s esophagus have at least a 20-fold increased risk of developing esophageal adenocarcinoma. Current surveillance guidelines for Barrett’s esophagus assume that the absolute annual risk of developing cancer is about 0.5%, but one large study found that the actual risk was only 0.12%, calling into question the rationale for ongoing surveillance of asymptomatic patients who have Barrett’s esophagus without associated dysplasia (see later, “Surveillance and Management of Barrett’s Esophagus”).Dysplasia-Carcinoma Sequence.
Pathologic data strongly suggest that esophageal adenocarcinoma in Barrett’s esophagus evolves through a sequence of progressively severe epithelial dysplasia in areas of preexisting columnar metaplasia. usually intestinal metaplasia. These dysplastic changes can be detected on endoscopic biopsy specimens obtained from Barrett’s esophagus. Dysplasia may occur in all types of Barrett’s mucosa but is most likely to occur in areas of intestinal metaplasia. Dysplastic changes are classified as low grade or high grade on the basis of the histologic findings; high-grade dysplasia may subsequently progress to invasive adenocarcinoma. In various studies, the prevalence of high-grade dysplasia in patients with esophageal adenocarcinoma has ranged from 68% to 100%. Studies have also shown progression to cancer within 7 years in 22% of patients with high-grade dysplasia in Barrett’s esophagus.
When Barrett’s esophagus is stratified into short-segment and long-segment disease, it has been found that patients with short-segment disease are more likely to develop dysplasia than the general population but are less likely to develop dysplasia than patients with long-segment disease. Esophageal adenocarcinoma has also been found to develop in patients with short-segment disease. Additional studies are needed, however, to further elucidate the cancer risk in these patients.
Surveillance and Management of Barrett’s Esophagus.
Because of the increased cancer risk, many investigators advocate endoscopic surveillance of patients with known Barrett’s esophagus to detect dysplastic changes, so these individuals can be treated before the development of overt carcinoma. Endoscopic biopsy specimens are generally recommended from all four quadrants of the lower esophagus at 1- to 2-cm intervals, starting at the gastroesophageal junction. In patients with nondysplastic Barrett’s esophagus, the American Gastroenterological Association recommends surveillance endoscopy at 2- to 5-year intervals, depending on the patient’s age and health. In patients with low-grade dysplasia, endoscopy is recommended at 1- to 2-year intervals because of an increased but uncertain risk of developing cancer. If high-grade dysplasia or early cancer is detected, treatment should be undertaken to prevent the development of cancer or treat these cancers at a curable stage. In the past, a surgical esophagogastrectomy was the definitive treatment for high-grade dysplasia or early cancers in Barrett’s esophagus. Currently, however, various forms of endoscopic therapy are recommended as less invasive alternatives associated with lower morbidity and mortality rates than surgery. Radiofrequency ablation, photodynamic therapy, and endoscopic mucosal resection have all been shown to be effective and safe forms of therapy for treatment of high-grade dysplasia or early-stage cancer in Barrett’s esophagus, with a high rate of dysplasia and tumor eradication and a reduced risk of disease progression in these patients.
Despite these recommendations, there is continued controversy about whether endoscopic surveillance of patients with Barrett’s esophagus is a cost-effective approach for detecting esophageal adenocarcinoma because of the low incidence of neoplasia and the high cost of endoscopy. So far, endoscopic surveillance of patients with Barrett’s esophagus has not been associated with a substantially decreased risk of death from esophageal adenocarcinoma. Thus, many questions remain about the role of endoscopic surveillance and its ultimate value in patients with Barrett’s esophagus.
Attention has also been focused on the role of DNA flow cytometry and cellular genetics analysis to determine which patients with Barrett’s esophagus are at greatest risk for the development of adenocarcinoma. Some investigators have shown that a subset of patients with Barrett’s esophagus have genomic instability, with aneuploid populations of cells, predisposing to neoplastic transformation. In various studies, the findings on DNA flow cytometry have correlated directly with the presence of high-grade dysplasia or invasive carcinoma on endoscopic biopsy specimens. Thus, flow cytometry represents another potential diagnostic tool for surveillance of Barrett’s esophagus.
Much less frequently, the development of cancer in Barrett’s esophagus may result from an adenoma-carcinoma sequence similar to that found in the colon. Benign adenomatous polyps have occasionally been documented in Barrett’s mucosa, with or without focal areas of invasive adenocarcinoma. Because malignant degeneration of these adenomas represents another potential pathway for the development of adenocarcinoma, endoscopic resection of adenomatous polyps in Barrett’s esophagus may decrease the risk of cancer.
Relationship Among Scleroderma, Barrett’s Esophagus, and Adenocarcinoma.
Scleroderma, a connective tissue disease characterized by smooth muscle atrophy and fibrosis, affects the esophagus in about 75% of patients. Esophageal involvement is usually characterized by a patulous, incompetent lower esophageal sphincter and absent esophageal peristalsis, with poor clearance of refluxed peptic acid from the esophagus once reflux has occurred. As a result, patients with scleroderma often have reflux esophagitis. Because of the severity of esophagitis, these individuals have an even greater risk of developing Barrett’s esophagus than other patients with reflux disease. In one study, 37% of those with scleroderma who underwent endoscopy for reflux symptoms had biopsy-proven Barrett’s esophagus. Because Barrett’s esophagus predisposes to esophageal adenocarcinoma, patients with scleroderma also appear to have an increased risk of developing esophageal cancer. Thus, scleroderma should indirectly be considered to be a premalignant condition in the esophagus.
Pathology
Gross Features
Adenocarcinomas arising in Barrett’s mucosa appear grossly as infiltrating, polypoid, ulcerative, or varicoid lesions. These tumors tend to be located in the distal or, less commonly, middle third of the esophagus. Unlike squamous cell carcinomas, adenocarcinomas of the distal esophagus frequently spread subdiaphragmatically to involve the gastric cardia or fundus. Studies have shown that adenocarcinomas arising in Barrett’s esophagus account for up to 50% of all adenocarcinomas involving the gastroesophageal junction. The remaining lesions are primary carcinomas of the gastric cardia or fundus invading the esophagus. Whether they arise in the esophagus or in the stomach, these tumors have similar morphologic features in terms of pattern of growth, degree of differentiation, and depth of invasion. Nevertheless, it is important to ascertain whether the cancer has arisen in Barrett’s esophagus because it may be necessary to resect not only the primary tumor but all residual Barrett’s mucosa in these patients. Detection of malignant tumor at the cardia should therefore lead to a careful search for Barrett’s epithelium in the esophagus.
Histologic Features
At the time of diagnosis, most adenocarcinomas in Barrett’s esophagus are advanced, unresectable tumors. Occasionally, however, early, potentially curable lesions may be detected by radiologic or endoscopic surveillance of patients with known Barrett’s esophagus. Alternatively, early lesions may be detected fortuitously in patients who undergo barium studies or endoscopy because of their underlying reflux disease.
Routes of Spread
Like squamous cell carcinomas, esophageal adenocarcinomas invade local, regional, or distant structures via direct extension, lymphatic spread, or hematogenous metastases. Unlike squamous cell carcinomas, however, adenocarcinomas in Barrett’s esophagus have a marked tendency to invade the proximal stomach; involvement of the gastric cardia or fundus occurs in 35% to 50% of cases.
Clinical Aspects
Barrett’s esophagus is predominantly a disease of older white men, with a male-to-female ratio of 3 : 1 and a mean age of about 65 years at the time of diagnosis. Nevertheless, there has been a significant increase in the incidence of these tumors in younger patients between 45 and 65 years of age.
Patients with esophageal adenocarcinoma usually present with recent onset of dysphagia and weight loss. Other findings include upper GI bleeding, odynophagia, and chest pain. Because of their underlying reflux disease, some patients have long-standing reflux symptoms before the development of cancer. By the time these individuals develop dysphagia, however, they almost always have advanced, unresectable tumors. Thus, patients with esophageal adenocarcinomas have a poor prognosis, with overall 5-year survival rates less than 20%.
Most patients with early adenocarcinomas in Barrett’s esophagus are asymptomatic, but some may present with melena, guaiac-positive stool, or iron deficiency anemia as a result of low-grade bleeding from the friable surface of the tumor. Others may seek medical attention because of their underlying reflux disease, so early cancers may be detected as fortuitous findings in patients presenting with reflux symptoms.
Radiographic Findings
Early Adenocarcinoma
Like squamous cell carcinomas, early adenocarcinomas in Barrett’s esophagus may appear on double-contrast esophagograms as plaquelike lesions or as flat sessile polyps. Sessile or pedunculated polyps in the distal esophagus may also represent adenomatous polyps in Barrett’s mucosa with or without foci of invasive carcinoma ( Fig. 23-17 ). In patients with peptic strictures, the earliest manifestation of a developing adenocarcinoma may be a localized area of flattening, stiffening, or irregularity in one wall of the stricture ( Fig. 23-18 ). Other patients may have superficial spreading carcinomas, manifested by confluent nodularity or granularity of the mucosa without a discrete lesion. Although early adenocarcinomas are typically small lesions, some patients may have relatively larger polypoid masses that are indistinguishable radiographically from advanced cancers.