10
Central Nervous System Brachytherapy
Abigail L. Stockham and Nils D. Arvold
BRACHYTHERAPY FOR MALIGNANT BRAIN TUMORS
Primary malignant brain tumors are among the most aggressive of all human neoplasms. Despite significant efforts to improve outcomes in this patient population over the past four decades, dramatic advances in progression-free survival (PFS) and overall survival have remained elusive. Standard therapeutic approaches with aggressive surgery, radiotherapy, and chemotherapy rarely offer the potential for cure. For example, median survival times for patients with glioblastoma (GBM) remain around 12 to 18 months in most series (1–3). Recurrences among primary malignant brain tumors are nearly always local, within or in close proximity to the original tumor. This pattern of local recurrence has resulted in many radiation dose escalation efforts, following a rationale that increasing radiotherapy doses would result in increased local control when compared with standard radiotherapy doses, thereby enhancing clinical outcomes in this patient population. Dose escalation for GBM in particular has been studied with a variety of radiotherapy platforms including conventional radiotherapy, stereotactic radiosurgery (SRS), and brachytherapy (4–7). The physical properties of brachytherapy theoretically provide an ideal mechanism for central dose escalation within the tumor–tumor cavity, seemingly critical for maximizing local control and optimizing outcomes for patients with newly diagnosed and recurrent malignant brain tumors. This chapter reviews the role of brachytherapy in the management of malignant brain tumors from the standpoints of radiobiology, physics, and clinical results.
RADIOBIOLOGY IN MALIGNANT BRAIN TUMORS AND ROLE OF BRACHYTHERAPY
Recent radiobiological models generated from patient data have been proposed, which offer characterization of the radiosensitivity and repopulation characteristics of GBM (8), and imply theoretical advantages for the use of brachytherapy in management. The models suggest that GBM cells exhibit significant intrinsic radiosensitivity combined with substantial repair capacity, resulting in an α/β ratio of 8 Gy. The relatively low dose rates of many brachytherapy sources used in the brain permit repair of radiotherapy-induced sublethal and potentially lethal DNA damage (9), and thus dose escalation with brachytherapy as opposed to external beam radiotherapy (EBRT) is likely to be more feasible with regard to normal tissue tolerance. The investigators futher established a doubling time of 15.4 days and a relatively delayed time to initiation of accelerated repopulation of 37 days. The potential clinical impact of these data is that overall treatment time may not play as critical a role in tumor control as previously thought, which also suggests that repopulation during the course of low dose rate (LDR) brachytherapy administration may not be a significant issue. The authors propose that with administration of 74.8 Gy delivered in 34 fractions of 2.2 Gy (biological effective dose [BED] = 92.1 Gy) as conventional EBRT, PFS may be improved (to 0.86; time point not provided) when compared with historical PFS data. Although promising, this model, demonstrating increased treatment efficacy with increased dose, has several limitations. Different molecular phenotypes that impact prognosis and treatment response were not considered when the model was developed, nor was modern chemotherapy with temozolomide uniformly applied. Acknowledging these limitations, these data represent the most up-to-date understanding of the radiobiology of GBM. They also offer a rationale for revisiting dose escalation as a means of optimizing local control and PFS. Although the use of brachytherapy was not included in the patient populations utilized to develop the GBM radiobiological models previously discussed, its use alone and in combination with EBRT has recently been revisited in the management of GBM (10). Brachytherapy may offer a delivery mechanism to accomplish this dose escalation while obviating dose-limiting toxicity of EBRT.
PHYSICS OF BRACHYTHERAPY
Brachytherapy for tumors of the central nervous system (CNS) is most commonly performed using either temporary high-activity iridium-192 (192Ir) or permanent low-activity iodine-125 (125I) sources, although additional isotopes such as cesium-131 (131Cs) have been introduced in recent years (Tables 10.1 and 10.2) (11). The indications and technical considerations for various isotopes are reviewed subsequently. However, no ideal brachytherapy source has yet been identified, particularly in light of the recently developed and proposed GBM radiobiological model. Brachytherapy has been investigated both in patients with newly diagnosed and recurrent primary malignant brain tumors (10,12–18). Many of these investigations have identified ideal candidate tumors to be supratentorial without evidence of basal ganglia or diencephalon involvement, though other studies have proposed expanded inclusion criteria for brachytherapy candidacy, based on experience with brachytherapy in these eloquent regions. Application systems and their technical considerations are discussed subsequently.
Table 10.1 Characteristics of radionuclides commonly used in central nervous system brachytherapy

Table 10.2 Parameters of permanent and temporary 125I seeds and 131Cs seeds

Permanent Implants (Low Dose Rate)
A permanent brachytherapy implant begins with the neurosurgeon performing maximal safe surgical resection. Subsequently, low-energy 125I sources are placed along the walls of the resection cavity at 0.5 to 1.0 cm intervals as an intraoperative collaboration between the neurosurgeon and radiation oncologist to help define the boundaries of the surgical cavity and precise seed spacing. Accurate spacing is most commonly achieved by implanting seeds that are pre-embedded within suture strands at defined intervals, or inserting mesh implants embedded with radioactive seeds at defined intervals. These 125I sources have photon energies of 27 to 35 keV, resulting in very limited tissue penetration with rapid dose falloff, and are the most commonly used isotope for permanent implants, although 131Cs is being used with increasing frequency by some groups. This technique is most effective for treating microscopic disease, with relative sparing of surrounding normal tissue. Following implantation, a CT scan is performed to calculate final dosimetry. Figures 10.1 to 10.3 demonstrate the highly conformal dose distributions and sharp dose falloffs, which spare surrounding normal brain parenchyma. Clinical indications and parameters for permanent implants are discussed in detail subsequently.
Temporary Interstitial Implants (Low Dose Rate)
A temporary interstitial implant system entails the placement of a stereotactic head frame, followed by a contrast-enhanced brain CT. Catheter trajectories are then planned, and a minimum peripheral dose of 60 Gy is typically prescribed to the margin of the contrast-enhancing tissue. Following afterloading of the nylon catheters with dummy sources, a head CT is obtained and careful dosimetry is performed. The source positions and strengths can be optimized to reflect the desired dosimetry. The isotope-containing inner catheters are then loaded for the prescribed amount of time to achieve the correct dose. Once this time has been accomplished, the catheters are removed and the patient is discharged.
GliaSite (High Dose Rate)
Temporary cavitary brachytherapy may be delivered with the GliaSite device (IsoRay Corp., Richland, WA) (Figure 10.4A). At the time of tumor resection, an appropriate balloon size is selected to fill the resection cavity. The double-walled balloon portion of the GliaSite catheter is placed within the resection cavity (Figure 10.4B). The other end of the catheter serves as the injection port and is fixed on top of the skull, but concealed underneath the skin. After the surgery, a contrast material and saline are injected into the catheter. Following balloon size—and placement—verification with MRI, the saline/contrast material is retrieved and replaced with the active 125I liquid colloid source (Iotrex, IsoRay Corp., Richland, WA). The liquid source dwells for 3 to 7 days, delivering the prescribed dose of radiation. At the end of this period, the source solution and saline are withdrawn and the balloon catheter is removed during a brief surgical procedure.

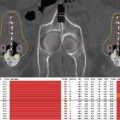
Figure 10.1 Images demonstrating brachytherapy implant for a gentleman with multiple recurrent atypical meningioma in the setting of prior resection and two courses of proton stereotactic radiosurgery (13 Gy/1 fraction and 15 Gy/1 fraction). At the time of surgical resection for a third recurrence, a brachytherapy implant was completed with 79 125I seeds for a total implanted activity of 29.7 mCi. Axial contrast-enhanced MRI demonstrating presurgical tumor volume (A), postoperative resection cavity demonstrating intracavitary seed placement (hypointense black regions, B), and isodose lines tightly conforming to 125I seeds in the axial plane on MRI (C) and CT (D). Sagittal imaging demonstrates preoperative tumor volume on T1 contrast-enhanced MRI (E), postoperative imaging demonstrating hypointense 125I seeds and isodose lines conforming to a highly complex surgical resection cavity (F). Sagittal imaging with CT (G) also demonstrates seed placement, highly conformal isodose lines, and avoidance of critical normal structures (midbrain and pons in kelly green, medulla in pale green, and optic chiasm in yellow).
Figure 10.2 Brachytherapy plan administered for multiply recurrent metastatic lung adenocarcinoma involving the brain after initial hypofractionated stereotactic radiotherapy (25 Gy/5 fractions), initial resection, and whole-brain radiotherapy (35 Gy/14 fractions). At the time of repeat surgical resection, 50 125I seeds were implanted for a total activity of 21.15 mCi. Axial contrast-enhanced MRI demonstrating presurgical tumor volume (A), intraoperative image demonstrating placement of stranded 125I seeds (B, courtesy of Dr. Alexandra Golby), postoperative resection cavity (C), and isodose lines tightly conforming to 125I seeds in the axial plane on MRI (D) and CT (E).
RESULTS OF BRACHYTHERAPY IN THE MANAGEMENT OF PRIMARY MALIGNANT BRAIN TUMORS
Newly Diagnosed High-Grade Gliomas (Grades 3 and 4)
Several retrospective and prospective series have characterized the efficacy of brachytherapy in the treatment of patients with newly diagnosed malignant glioma. One of the earliest and largest series, reported by Loeffler et al (6), included patients with supratentorial tumors of 5 cm or less in diameter, without involvement of the corpus callosum or ependymal surfaces, and a minimum Karnofsky performance status (KPS) score of 70. Thirty three of the 35 patients underwent surgical debulking, while the remaining two patients underwent biopsy for histopathologic evaluation. Patients were then treated with EBRT to a dose of 59.4 Gy, delivered in 1.8 Gy fractions, without concurrent chemotherapy. Two weeks after completion of EBRT, patients underwent stereotactic placement of brachytherapy catheters. Interstitial brachytherapy was administered via high-activity, LDR 125I sources (20–50 mCi, minimum dose rate of 0.4 Gy/hr; Figure 10.5), to a dose of 50 Gy. Survival at 1 year was 87% for the brachytherapy group, compared with 40% for historic controls treated with EBRT alone (P < .001). At 2 years, the survival in the brachytherapy group was 57% compared with 13% for the control group. Forty percent of the patients in the brachytherapy group required reoperation for neurologic deterioration, at a median time of 6 months from brachytherapy administration. Histopathologic evaluation at reoperation demonstrated residual microscopic disease in most specimens, although the viability of these cells was difficult to determine. The acute toxicity rate was 18%. Patients treated with brachytherapy generally experienced a decline in KPS, from 80 at the time of implantation, to 70 at 6 months, and 60 at 12 months. After this time, there did not appear to be any further decline of KPS. In an updated report, a median survival of 18 months was reported in the group treated with both brachytherapy and EBRT compared with 11 months for patients treated primarily with EBRT (6). The reoperation rate was 64% for the brachytherapy group and the median survival improved to 22 months in the patients undergoing reoperation, compared with 13 months for those who did not.



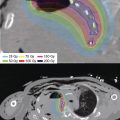
Figure 10.3 Brachytherapy plan administered for recurrent metastatic non–small cell carcinoma of the lung involving the right cerebellum. The patient had previously undergone initial surgical resection, whole-brain radiotherapy (30 Gy/10 fractions), and hypofractionated stereotactic radiotherapy (25 Gy/5 fractions) for recurrent disease. Preoperative axial T1 contrast enhanced MRI demonstrates recurrent tumor (A). At the time of re-resection, 50 131Cs seeds (average activity of 3.66 mCi for a total activity of 183.0 mCi) were implanted at the perimeter of the resection cavity as can be seen in axial (B), sagittal (C), and coronal (D) CT images. The infratentorial radiopaque seeds can also be seen on the planar imaging (E). Isodose distributions can be seen on CT images in the axial (F), sagittal (G), and coronal (H) planes.
Figure 10.4 GliaSite catheter is tested with saline before insertion into tumor cavity. Note the flexible catheter and balloon with central infusion channel (A). Schematic of relationship of intraparenchymal intracavitary balloon with extradural subgaleal infusion port (B). Images courtesy of IsoRay Corp, Richland, WA.

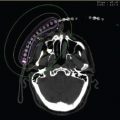
Figure 10.5 Schematic of temporary 125I catheter–based implants showing transcranial placement by a closed stereotactic technique (A). Neurosurgeon placing catheter for temporary 125I (B). Axial CT scan after the placement of the catheter with dummy markers into a left frontal lobe tumor (C). Panel A courtesy of Dr. Loeffler’s teaching file and Panels B and C courtesy of Dr. Normand Laperierre.
The Brain Tumor Cooperative Group 87–01 trial was another randomized trial examining the benefits of brachytherapy in patients newly diagnosed with malignant glioma (4). This study compared surgery, EBRT, and carmustine (BCNU) compared to the same plus the addition of brachytherapy implant. Until May 1989, all patients received whole-brain radiotherapy to a dose of 43 Gy in 25 fractions, plus a boost for an additional 17.2 Gy delivered in 10 fractions. After May 1989, all patients received 60.2 Gy in 35 fractions to the contrast-enhanced volume plus a 3 cm margin. Interstitial implantation was performed using high-activity temporary 125I sources, designed to cover the contrast-enhancing volume with up to a 1 cm margin. A total of 60 Gy was delivered to the tumor perimeter over 5 to 7 days. Factors associated with outcome were reported to be age, KPS, and histopathology. However, the slightly longer median survival for the brachytherapy group (68.1 weeks vs 58.8 weeks for patients treated with EBRT) was not statistically significant (P = .101).
An additional, more recent series of high dose rate stereotactic brachytherapy (HDR-STBT) involved 28 patients with newly diagnosed GBM treated with brachytherapy in combination with surgery and EBRT (24). Eligibility requirements included unifocal lesions, residual tumors less than 6 cm, supratentorial lesions, tumors not crossing midline and without subependymal spread, and KPS greater than 60. Brachytherapy was administered over 5 consecutive days with two fractions per day for a total median dose of 30 Gy. Twenty-eight GBM patients who were treated with surgery and EBRT alone served as matched controls. It was determined that the median survival times for the brachytherapy and the control group were 19.5 and 12.5 months, respectively. The 1- and 2-year survivals were 89% versus 42% and 61% versus 28%, respectively (P = .12). On multivariate analysis, age, KPS, and HDR-STBT were significant determinants of survival. It was ultimately found that the survival benefit was most significant for recursive partitioning analysis (RPA) Class V patients, compared with the control group.
In summary, although some series have suggested advantages to brachytherapy for newly diagnosed GBM, the largest prospective data from randomized trials do not demonstrate a significant survival benefit.
Recurrent Malignant Glioma
Outcomes of patients with recurrent malignant gliomas are known to be especially poor. Although there have not been phase III randomized studies reported on the value of brachytherapy for this patient population, there have been retrospective reports published that suggest potential benefit. Shrieve et al (25) reported on patients with recurrent malignant gliomas who were either treated with temporary high-activity brachytherapy or radiosurgery. The median survivals were found to be comparable in both groups (11.5 months for brachytherapy vs. 10.2 months for radiosurgery). The risk of reoperation was higher in the brachytherapy group at 64% versus the radiosurgery group at 33%, although the treatment volumes were significantly larger in the brachytherapy group.
Emerging data suggest that low-activity permanent implants may provide some advantages over high-activity temporary implants with the rates of symptomatic necrosis being lower. Some studies also suggest an improvement in median survival times of patients with recurrent malignant gliomas. Larson et al (26) reported on the outcomes of permanent low-activity sources in 37 patients with recurrent GBM. The dose administered approached 300 Gy at 5 mm depth, with an initial dose rate of 15 cGy/hr. The median survival of these patients was found to be 52 weeks, with a median time to progression of 16 weeks. Of the 37 tumors, 34 recurred. Given the relatively short time to tumor progression, the authors recommended options other than brachytherapy for patients with recurrent malignant gliomas (27).
NOVEL BRACHYTHERAPY APPROACHES FOR MALIGNANT GLIOMAS
A novel brachytherapy technique with 125I-labeled monoclonal antibodies (mAbs) has been explored in patients with newly diagnosed GBM. The radiolabeled antibodies are directly introduced into the resection cavity at the time of initial surgery, taking advantage of the molecular characteristics of these tumors. Tenascin, which is an extracellular matrix hexabrachion glycoprotein, has been found to be expressed in high frequencies in high-grade gliomas, but not in normal brain (28). A mAb 81C6 has been developed, which binds to an epitope within the alternatively spliced fibronectin type III region of tenascin, which is expressed in high frequencies in gliomas. In a phase II study from Duke, 120 mCi of 131I-labeled murine 81C6 was directly injected into the resection cavity of 33 patients with high-grade gliomas (GBM = 27; AA = 4; anaplastic oligodendroglioma [AO] = 2). Patients then received EBRT followed by 1 year of alkylator-based chemotherapy. Median survival for all patients was 86.7 weeks and for patients with GBM, in particular, it was 79.4 weeks. There were some patients who had longer term survival outcomes. In fact, 11 patients remain alive at a median follow-up of 93 weeks. The treatment was generally well tolerated, with nine patients (27%) having developed reversible hematologic toxicity. Neurotoxicity occurred in five patients (15%). One patient (3%) required reoperation for radiation necrosis. The authors concluded that the median survival achieved with 131I-labeled 81C6 exceeded that of historical controls, with significantly lower rates of reoperation.
In addition, nanoparticle technology has been evaluated for use as a platform for interstitial brachytherapy (29), with promising results in xenograft mouse models with GBM. Human application has not yet been reported. Additional nanoparticle platforms have demonstrated promise in animal models, but are not yet reported in humans (30). Use of this technology may further the role of brachytherapy in the management of CNS neoplasms in the future.
Summary
Of all human neoplasms, primary malignant brain tumors are one of the most treatment refractory types. Few therapies have been demonstrated to significantly improve outcomes. Unfortunately, phase III studies have not demonstrated success in identifying patients who derive a significant survival benefit with the addition of brachytherapy to aggressive standard treatment of malignant gliomas including surgery, EBRT, and chemotherapy. Further molecular–genetic profiling may identify patients with malignant gliomas who derive a benefit from the addition of brachytherapy into the overall treatment regimen. Novel strategies such as 131I-labeled murine mAbs and use of nanotechnology may improve the therapeutic ratio of brachytherapy through reduction of neurotoxic side effects in the future.
Low-Grade Glioma
The use of brachytherapy in the management of low-grade gliomas has been studied in several retrospective reviews, most frequently in pilocytic astrocytoma. Pilocytic astrocytoma, a well-circumscribed World Health Organization (WHO) grade 1 glioma often arising in eloquent regions of the brain, is diagnosed most frequently in the pediatric patient population. Standard of care for this patient population includes maximal safe surgical resection followed by observation. However, significant therapeutic challenges exist when tumors arise in eloquent regions such as in the optic chiasm, hypothalamus, diencephalon, or mesencephalon because surgical resection may be inadvisable. Additionally, the long-term sequelae of radiotherapy are of particular importance in the pediatric patient, in whom radiotherapy administration may result in significant long-term neurocognitive, neuroendocrine, and/or neuropsychiatric effects. The well-circumscribed nature of pilocytic astrocytoma, frequent location in eloquent regions of the brain, and typically pediatric population provide a strong rationale for evaluation of brachytherapy in patients with pilocytic astrocytoma. Moreover, studies have demonstrated safety and tolerability of brachytherapy in the management of tumors present in eloquent regions of the brain as well as in regions of the brain previously thought to be unsuitable for brachytherapy (31). The use of brachytherapy in the management of tumors located in highly eloquent regions of the brain was corroborated by a subset of a larger series of patients with low-grade gliomas treated with temporary 125I implants (32,33). In a subset of this large series, 45 patients whose tumors were located in eloquent regions, primarily the hypothalamus, underwent temporary implant with LDR (10 cGy/hr) 125I to a total dose of 54 Gy. No evidence of radiation-related complications was reported. Local control was excellent, with no evidence of progressive disease at a median follow-up of 31.5 months and five patients reported improvement in their neurologic symptoms (34).
These findings were supported by publication of a large, single-institution report in which 160 patients with inoperable low-grade glioma (WHO grades 1 and 2) underwent permanent 125I implantation to a cumulative dose of 50 to 65 Gray within 9 months of implantation (initial dose rate: 0.7 Gy/d) (35). The authors reported that the procedure was well tolerated, with no procedure-related mortality and an operative morbidity of 5.4%. At a median follow-up of 67.1 months, no significant difference was seen between WHO grades 1 and 2. A complete response was noted in 24.6% of patients, partial response in 31.0% of patients, stable disease in 29.6% of patients, and tumor recurrence was reported in 14.8% of patients. This study was consistent with prior studies in that neurologic status improved in 57.8% of patients and remained stable in 23.0% of patients. Survival at 5 years was 93%, and 82% at 10 years. The authors concluded that STBT with 125I seeds was a safe, effective initial treatment strategy for patients with inoperable low-grade glioma.
Brachytherapy in the management of low-grade glioma in eloquent regions has also been reported in other single-institution case series. Schnell et al (36) reported on their pilot series evaluating the role of definitive 125I implantation alone in the setting of small (< 4 cm) and complex recurrent grade 2 gliomas, as well as microsurgery plus STBT for grade 2 glioma tumors greater than 4 cm. Of the 31 patients who underwent LDR-STBT, 18 were treated de novo, while 13 patients were treated for recurrent tumors. Stereotactic LDR (median 10 cGy/hr) interstitial brachytherapy was administered to a reference dose of 54 Gy with 125I. The authors reported a 27.8% rate of combined microsurgery and brachytherapy-related morbidity, and a 6.4% rate of transient morbidity in the patients managed with brachytherapy alone. Complete response was noted in 8 patients, partial response in 9 patients, and stable disease in 14 patients, with 5-year PFS greater than 60%. No permanent treatment-related morbidity was reported.
Ruge et al (37) published their experience in a series of 47 patients with inoperable brainstem gliomas treated with stereotactic 125I brachytherapy to a median surface dose of 65 Gy. The treatment was determined to be safe, with no procedure-related mortality, a 13% rate of transient perioperative neurologic deterioration, and a 4% rate of permanent neurologic deterioration. The reoperation for space-occupying cysts was 13%, and was noted to occur at a median time of 28.5 months from permanent interstitial brachytherapy seed placement. Responses were split nearly evenly, with a complete response rate of 23.4%, partial response rate of 29.8%, stable disease in 27.7%, and progressive disease in 19.1%. PFS rates were 98%, 81%, and 62% at 1, 5, and 10 years, respectively. Overall survival rates at 1, 5, and 10 years were 100%, 98%, and 88%, respectively. These survival rates compare favorably with data evaluating the use of EBRT with concurrent and adjuvant temozolomide, with 1-year PFS and OS of 33% and 50%, respectively (38). These studies must be interpreted with acknowledgment of selection bias, as brachytherapy for brainstem glioma is only appropriate in those patients whose tumors are amenable to surgical access, which may be confounded by tumor location and grade. Future studies may seek to combine chemotherapy with brachytherapy in the patient population, or explore brachytherapy as a boost to EBRT.
Long-term toxicity in patients who undergo brachytherapy for CNS neoplasms is not well characterized. Sneed et al (39) retrospectively reviewed KPS in patients at UCSF treated with brachytherapy for low-grade glioma. The mean baseline KPS for the 28 patients undergoing brachytherapy was 88 (+/– 9). Three years following brachytherapy, the mean KPS for the group was 87 (+/– 7). Performance status was maintained over time in the 11 patients surviving more than 5 years following brachytherapy, whose mean KPS remained 87 (+/– 9) (39). Additional cohorts with available long-term follow-up suggest minimal long-term treatment-related toxicity. In their previously discussed cohort of 160 patients with low-grade glioma managed with 125I, Ruge et al (35) reported a 3% rate of treatment-related morbidity including permanent hemiparesis, visual field deficits, and trochlear nerve palsy at 7 to 18 months following brachytherapy. Patient-reported outcomes in another study of 51 patients treated with 125I for low-grade glioma as children were somewhat less favorable (40). Moreover, patients may develop vasculopathy many years following brachytherapy treatment for low-grade glioma. Tacke et al (41) reported magnetic resonance angiography (MRA) findings in 13 patients (four with neurofibromatosis type 1 [NF-1]) included as part of a larger cohort who underwent prior temporary 125I implantation for suprasellar low-grade gliomas. At a range of 3 to 13 years following brachytherapy, six patients were noted to have radiographic evidence of cerebral vasculopathy, though only one patient reported symptoms of cerebral ischemia. Given the small patient population and indeterminate clinical significance, the authors were unable to definitively report on vascular outcomes in patients treated with brachytherapy for low-grade gliomas, but advocated for consideration of vasculopathy in this patient population and inclusion of MRA in routine follow-up of patients to evaluate for vasculopathy regardless of symptoms.
Summary
STBT offers a potentially effective therapeutic strategy for patients with surgically inaccessible low-grade gliomas, with an acceptable toxicity profile based on institutional series. Moreover, the physical characteristics of brachytherapy offer a radiotherapy option with minimal dose to surrounding tissue, suggesting minimization of long-term radiotherapy sequelae in this patient population with high rates of overall survival. Multi-institutional, prospective studies with incorporation of patient-reported outcomes may offer additional insights regarding efficacy as well as late effects.
BRACHYTHERAPY APPROACHES FOR BRAIN METASTASES
Brachytherapy offers an appealing theoretical benefit in patients with solid tumor metastases to the brain. Often, patients have received prior courses of EBRT to the brain including whole-brain radiotherapy and SRS, limiting the feasibility of intervention with additional external beam radiotherapeutic techniques. Moreover, the classically well-circumscribed, noninfiltrative nature of metastatic disease to the brain is well suited for the highly localized, conformal dose delivery that brachytherapy provides with rapid dose falloff.
Theoretical risks to the use of interstitial brachytherapy as an adjunct therapy in the management of resected metastatic disease include the possibility for collapsing of the tumor cavity around the interstitial brachytherapy source, resulting in higher than intended dose to surrounding normal brain. However, this could also provide a potential benefit in the event microscopic disease extension exists beyond the area of contrast enhancement traditionally treated with external beam radiotherapeutic techniques, including SRS.
Several case reports and case series in the 1980s and 1990s reported on early experiences with 125I and 192Ir in the management of brain metastases, with promising anecdotal experience and minimal toxicity (42–45). In 2006, The Brain Metastasis Study Group published results from a multi-institutional phase II trial, in which patients with newly diagnosed single-brain metastasis underwent resection and placement of a GliaSite catheter and balloon followed by delivery of 60 Gy (40–60 cGy/hr) prescribed to 1 cm depth over a median of 114 hours (range: 93–162 hr) (46). The median tumor volume was 12.7 cm3 (range: 2–150 cm3). Actuarial 1-year local control in the 54 patients who completed brachytherapy treatment was 79% at 1 year, which was increased to 87% when two patients with histopathologically proven radiation necrosis in the absence of persistent tumor were excluded. Median time to local recurrence in the nine patients whose tumors progressed was 26 weeks (range: 5–66 weeks). Of the original nine patients considered as failures, four patients were treated with whole-brain radiotherapy, one underwent SRS, and six underwent surgical resection, which demonstrated radiation necrosis alone, as previously mentioned. CNS-related death occurred in four patients (11%). Similar to case series evaluating the role of brachytherapy in the management of primary malignant gliomas, median survival was increased in patients who developed radiation necrosis—overall survival was 83 weeks in patients who developed radiation necrosis versus 35 weeks in patients who did not develop necrosis. The authors concluded GliaSite brachytherapy offered comparable local control, duration of functional independence, and median survival time compared with surgical resection of a single metastasis and adjuvant whole-brain radiotherapy. It is noteworthy that the authors advocated for prescribing to a depth of 0.5 cm in future studies. Additional studies established the feasibility and safety of outpatient brachytherapy with GliaSite (47).
Management of a single-brain metastasis with resection and permanent 125I implant and omission of whole-brain radiotherapy was reported in multiple single- and dual-institution series (48–50). Each study reported rates of local control at 1 year of 90% to 95% in patients with newly diagnosed brain metastases, and a local control rate of 86% in patients with recurrent brain metastases (49). It is noteworthy that in the series from UCSF by Huang et al, the median dose delivered at 5 mm depth was 400 Gy, and the overall rate of symptomatic necrosis was 23%, most of whom required reoperation, suggesting that the doses used in that study may be associated with higher rates of toxicity. The authors in each series advocated for consideration of surgical resection and permanent 125I in patients with single-brain metastases amenable to surgical resection and omission of whole-brain radiotherapy in the initial treatment paradigm. Several authors emphasize an advantage to brachytherapy over SRS in that no size restriction exists for consideration of brachytherapy. Ruge et al (51) reported a retrospective review that compared outcomes in patients with single-brain metastases managed with linear accelerator-based SRS versus 125I (surface dose of 50 Gy delivered over 42 days). There were no differences noted in local control (93.6% for patients undergoing a SRS vs 96.7% in patients undergoing brachytherapy), nor were any differences noted in distant brain failure (42.4% for patients undergoing SRS vs 46.4% of patients undergoing brachytherapy). No treatment-related mortality was reported and no patients experienced permanent grade 3 or 4 CNS toxicity. The authors concluded that STBT offers a safe, minimally invasive, highly effective therapeutic intervention in the management of single metastases. They highlight advantages of brachytherapy over SRS, including histopathologic evaluation and treatment with one procedure, unrestricted tumor localization, and the ability to apply brachytherapy in metastases larger than may be appropriate with SRS.
Recently, Brachman et al (20) reported preliminary results with re-irradiation of 10 patients with recurrent intracranial neoplasms, including two patients with recurrent metastatic disease from breast cancer, with 131Cs (Proxcelan, Isoray Corp, Richland, WA). Other patients included in the study were treated for recurrent grade 3 meningioma (four), grade 2 meningioma (two), craniopharyngioma (one), and one patient with malignant spindle cell neoplasm. Of the two patients with metastatic breast cancer, one had been previously treated with SRS alone to 16 Gy, while the other patient had previously undergone surgical resection, SRS to 16 Gy, and a second course of SRS to 15 Gy. Patients were taken to the operating room where re-resection was performed. Carriers for 131Cs sources were created intraoperatively, adding 12 to 20 minutes per case, to deliver a dose of 60 Gy prescribed to a depth of 5 mm (4–47 seeds, 14–169 mCi). Of the 10 patients included in the study, only one patient met exposure requirements for radiation safety precautions for clinical staff at the conclusion of the craniotomy. No wound-healing complications, radiation necrosis, or tumor bed recurrences were reported at a follow-up time of 1 to 10.5 months; both patients with breast cancer were noted at 5 months of follow-up to have no evidence of recurrence at the resection cavity. This approach offers support for re-irradiation in patients who have undergone significant prior therapy, allows for dual-modality treatment in a single procedure, and results in minimal additional case time and radiation precautions by nursing. A prospective trial is currently underway to validate their initial findings and provide additional information to optimally define the role of 131Cs in the management of intracranial neoplasms, including brain metastases.
Wernicke et al (21) reported on their experience with 131Cs in the management of newly diagnosed brain metastases treated as part of a phase I or II study at Cornell. In this study, 24 patients with newly diagnosed brain metastases, including those with traditionally radioresistant histologies, were taken to the operating room where resection of the metastasis was performed. Subsequently, permanent implantation with 131Cs seeds was performed to a dose of 80 Gy to 5 mm depth. Implant characteristics were based on a physics nomogram and volumetric analysis of the resected metastasis. With a median follow-up of 5 months, the 1-year resection cavity freedom from recurrence was 100%. The procedure was relatively well tolerated; one patient required reoperation for a dural tear, while one patient developed seizures. No other additional implant-related side effects were noted, with evaluation of psychological function and quality-of-life correlates still pending.
The Cornell group also studied the cost-effectiveness of four different intervention strategies for the management of brain metastases (52). Single institutional data were retrospectively reviewed, with 74 patients having undergone one of four intervention strategies: surgery plus 131Cs brachytherapy implant, SRS alone, surgery plus whole-brain radiotherapy (10 fractions), and whole-brain radiotherapy alone (15 fractions). The authors concluded that surgery plus 131Cs brachytherapy implant was significantly less costly than the other treatment strategies. The authors then considered 131Cs versus whole-brain radiotherapy alone (10 fractions). Surgery and 131Cs implant remained less costly, at $19,271 for surgery and 131Cs versus $20,571 for whole-brain radiotherapy alone over 10 fractions. Of note, this analysis only evaluated the cost associated with interventions at one time point, and not overall cost of care for the management of brain metastases.
Summary
Brachytherapy may offer an attractive treatment strategy for histopathologic evaluation, symptom relief, local dose escalation, and condensed overall treatment time. Histopathologic evaluation may provide additional information valuable in directing subsequent therapies, and short overall treatment time may offer an opportunity for patients to resume systemic therapy shortly after resection of a symptomatic brain metastasis. These benefits are largely theoretical at this time and have not yet been verified with rigorous study, including comparison with existing techniques such as SRS to the surgical cavity. In summary, brachytherapy in the management of brain metastases is associated with apparent efficacy and low morbidity, though the optimal patient population remains to be defined in ongoing studies.
BRACHYTHERAPY FOR MENINGIOMA
Several brachytherapy techniques have been used in the management of intracranial meningiomas that exhibit aggressive histopathologic features that recur following prior resection and radiotherapy, and/or that cannot undergo resection. Several brachytherapy platforms have been described including permanent high-activity LDR permanent 125I seed implant (53–59), permanent implant with other radionuclides (gold-198 [198Au], phosphorous-32 [32P], yttrium-90 [90Y]) (60), and temporary 198Ir implant (61). The largest series evaluating the role of brachytherapy in the management of meningioma has reported on the use of high-activity LDR 125I seeds implanted at the time of surgery, or with stereotactic guidance (57,59,62,63).
In the 1980s and 1990s, Kumar et al (53–57) published a series of papers reporting on the use of high-activity LDR 125I in the management of recurrent and unresectable meningiomas, including those involving the skull base. Patients underwent implantation at a dose rate of 5 to 25 cGy/hr to a total dose of 100 to 500 Gy. The authors noted that patients with calcified meningiomas demonstrated lower rates of tumor regression, but noted no local recurrences in their series at an early median follow-up of up to 25 months. No complications were reported and the authors concluded that brachytherapy with 125I offered an effective, well-tolerated treatment option for patients with newly diagnosed and recurrent meningioma.
Slightly higher rates of toxicity were reported in 25 patients with parasellar–clival meningiomas, which also used permanent 125I, but to a lower total dose of 100 to 150 Gy, than in the series by Kumar et al (53,54,56,57). Procedure-related cranial nerve III injury was noted in 1 of 25 patients and new or worsening facial numbness was appreciated in 9 of 25 patients, while 8 of 17 patients with preexisting cranial neuropathies experienced resolution of symptoms. At a median follow-up of 19 months (range: 6–32 months), four patients demonstrated moderate radiographic response, 13 meningiomas were slightly reduced in size, and five tumors demonstrated stable radiologic findings on follow-up CT. In 19 elderly patients who underwent definitive brachytherapy, tumor response was more dramatic, as tumor volume at 12 months had reduced by 54%. The authors report an actuarial tumor-related survival rate of 78% at 1 year and 62% at 2 years and concluded that brachytherapy offers a relatively safe treatment modality in the management of parasellar–clival meningiomas and meningiomas in the elderly (63).
In addition, Ware et al (62) reported on the outcomes of 17 patients with recurrent malignant meningioma and four patients with atypical meningioma managed with permanent 125I implant at the time of surgical resection. The median number of sources implanted was 30 (range: 4–112 sources) of a median total activity of 20 mCi (range: 3.3–85.9 mCi). At a median follow-up of 6.6 years, median overall survival was 2.4 years after brachytherapy implant. The cohort of patients with atypical meningioma was noted at a median follow-up of 9.4 years to have a median survival of 2.4 years from the time of brachytherapy implantation. PFS for the combined group was 10.4 months.
Most recently, brachytherapy was studied in the management of recurrent grade 2 and grade 3 meningioma in patients who had undergone prior EBRT as part of a case series by Brachman et al (20). In this case series, recurrent meningioma comprised a majority of patients treated with surgical resection and permanent 131Cs seed implant. Two patients with recurrent grade 2 meningioma were included who had each undergone two prior surgeries and SRS to 15 Gy; four patients with recurrent grade 3 meningioma were included who had all undergone multiple prior radiotherapy courses to a total dose greater than 85 Gy. All patients were taken to the operating room for maximal safe surgical resection and permanent implant with 131Cs to a dose of 60 Gy prescribed to 5 mm depth (4–47 seeds/bed were implanted with 14–169 mCi of 131Cs). No patients in this series demonstrated evidence of disease recurrence or treatment-related complications, though follow-up was short at 6.5 months in the two patients with grade 2 meningioma, while two patients with grade 3 meningioma were followed to 10.5 months, one patient with grade 3 meningioma was followed to 7 months, and one patient with grade 3 meningioma was followed for only 1 month postprocedure. The authors of this series concluded that brachytherapy with 131Cs was well tolerated in a heavily pretreated patient population and report further investigation was underway.
Summary
The use of brachytherapy has been explored for its role in the management of meningiomas of a variety of grades and locations, with variable rates of toxicity reported. Over the past several decades, many patients previously considered for intervention with brachytherapy are now able to undergo surgical resection and/or EBRT as a result of technological advances. However, the role of brachytherapy in patients with high-grade recurrent tumors has recently been revisited, with promising results. Future studies will clarify the role of brachytherapy in the management of meningioma.
DURAL PLAQUE BRACHYTHERAPY FOR SPINAL TUMORS
Intraoperative dural brachytherapy by customized 192Ir or 90Y plaques has been developed at the Massachusetts General Hospital for spinal tumors. This technique was developed for the management of spinal–paraspinal sarcoma, which poses unique treatment challenges, given the radiation tolerance of the human spinal cord.

Figure 10.6 Polycarbonate dural plaque custom made for intraoperative implantation to spinal dura (A). The radioactive plaque in situ on the dura, stabilized with gauze packing for the duration of treatment (B). The radiation physicist carefully manages the timing of the implant. Courtesy of Dr. DeLaney’s teaching file.
Surgery is the primary treatment for most spinal tumors, but wide margins are rarely achievable and tumor cells may remain on the dura. Resection of the dura itself poses risks of contamination of cerebrospinal fluid (CSF) and CSF leak. Adjuvant EBRT in the management of microscopic residual disease typically requires at least 65 Gy be delivered for sarcoma, but this is challenging given the spinal cord tolerance of only 45 to 50 Gy. The main problem is one of spacing, as the dura and the spinal cord are separated by only 3 to 4 mm of CSF at most.
A National Cancer Institute (NCI)-supported protocol was designed to develop a dural brachytherapy applicator to preferentially treat the dural surface and spare the spinal cord. The initial challenge of establishing a mechanism for dose delivery was met with fabrication of a semicylindrical polycarbonate brachytherapy plaque of the appropriate size (Figure 10.6). Next, various isotopes were evaluated. The dural areas at risk were measured on preoperative MRI scan, allowing for creation of an appropriately sized nonradioactive plaque to confirm appropriate fit and placement. Once the fit and placement were confirmed, the associated live plaque was installed. The initial plaques were made with 192Ir. Subsequent plaques were made with 90Y because of their superior dose distribution.
The treatment protocol included EBRT (with or without protons) and best resection of vertebral, soft tissue, and/or epidural tumor. The nonradioactive plaque was fitted to the dura at risk. When the fit was approved, there was an intraoperative brachytherapy boost to dura by placing radioactive dural plaque for between 6 and 40 minutes to deliver an intraoperative radiation therapy (IORT) boost dose of 7.5 to 15 Gy. The timing of dwell was precalculated to ensure adherence to prescription and written directive and was independently monitored by the authorized user and physicist. After dose delivery, the plaque was removed and the surgery proceeded to additional vertebral reconstruction and stabilization as indicated by the primary resection. Postoperative chemotherapy was administered when clinically indicated (64).
PARASPINAL PERMANENT SEED BRACHYTHERAPY FOR METASTATIC SPINAL TUMORS
Rogers et al reported the follow-up on 24 of 30 patients implanted with 125I permanent implants at the time of spinal cord decompressive surgery. The study group consisted primarily of patients with metastatic disease who were undergoing decompressive surgery. The implant methodology employed 125I seeds in suture strand (Oncura, Inc., Arlington Heights, IL) with seed spacing set at 1 cm. The space between the strands was also 1 cm. The average activity per seed was 0.46 mCi, with an average of 20 seeds per implant for an average activity of 13.8 mCi. The mean minimal peripheral dose was 22 Gy (very low dose rate [vLDR]). Most patients received EBRT up to spinal cord tolerance, with a mean of 38 Gy (various fractionations).
The implant methodology was to fixate seeds in suture to the target area with a combination of methylmethacrylate, staples, suture, Gelfoam, and direct fixation to the implanted reconstructive devices (eg, screws, rods, and cages). The methodology avoided direct placement of seeds on nerve roots or dura by using a double-layer Gelfoam technique. The postoperative implant evaluation was carried out with orthogonal films and later with CT scan dosimetry. Implants were evaluated for dose and also for the risk of seed migration. At median follow-up of 42 months, the 2- and 3-year median local control rates were 87% and 73%, respectively. The functional outcome in this group was closely linked to the pretreatment level. No myelopathy or other adverse radiation events were observed (65).
Novel brachytherapy techniques are continuing to be developed, including use of implanted 125I seeds, as demonstrated by Yang et al (66–68) through a recent series of studies evaluating the role of percutaneous vertebroplasty with interstitial implantation of 125I seed in 50 patients with osteoblastic metastases, after first exploring toxicity and efficacy in a banna mini-pig model. The authors report a 98% rate of significantly relieved back pain, improvement in KPS scores, and advocate for further evaluation of the technique to augment the efficacy of percutaneous vertebroplasty.
Recent application of HDR brachytherapy in the management of metastatic disease to the spine has been reported. Folkert et al (69) reported a series of five patients with spinal metastases who had undergone at least two prior courses of radiotherapy who were managed with 192Ir brachytherapy. Two patients underwent intraoperative placement of brachytherapy catheters, while three patients underwent percutaneous placement. A median of four catheters (range: 2–10 catheters) were required to deliver a median dose of 14 Gy (range: 12–18 Gy), while respecting cord–cauda equina dose constraints. Reported median cord–cauda equina Dmax was 7 Gy (range: 5.8–10 Gy), with a median cord–cauda equina dose of 4 Gy (3–5.6 Gy). At a median follow-up of 9 months (range: 4–17 months), four patients reported reduction of pain, with complete resolution of pain in two patients. Of particular note, no patients developed local recurrence–progression and no patients developed brachytherapy-associated toxicity.
Summary
The role of brachytherapy in the management of primary and secondary tumors of the spine is not well characterized. Advances in the techniques of brachytherapy, imaging, surgery, and interventional radiology have allowed for development of novel brachytherapy applications for patients with spinal tumors, and there are currently limited but promising data for this population.
CONCLUSIONS
The role of brachytherapy in the management of CNS neoplasms has held appeal for more than 100 years (70), over which time this modality has fallen in and out of favor for the promise held by its physical dose distribution and biological characteristics, balanced by the potential for toxicity. The application of brachytherapy in the management of patients with primary and secondary tumors of the CNS remains an area of active research, with the potential widening of the therapeutic ratio made possible by ever-advancing surgical techniques, imaging modalities, and brachytherapy systems.