Cervicothoracic Junction: Brachial Plexus Trauma
KEY POINTS
- Traumatic brachial plexopathy may benefit or be made worse by attempted repair.
- Repair or reconstruction of the brachial plexus has become more of an option.
- Imaging is critical to proper medical/surgical decision making with regard to the timing and likely success of a surgical repair or reconstruction.
- Imaging is essential in cases where there may be an acute vascular complication.
The general approach to conditions of the cervicothoracic junction (CTJ) is presented in Chapter 163 and should be reviewed at this time if necessary. This chapter presents examples of brachial plexus trauma. Patients with trauma causing a brachial plexopathy should come to imaging mainly when the timing and nature of the trauma might allow for useful therapeutic intervention.
ANATOMIC CONSIDERATIONS IN DIFFERENTIAL DIAGNOSIS
The general anatomy of the brachial plexus and its relationship to the CTJ is presented initially in Chapter 149. This anatomy is also presented in a more focused review in Chapter 163 as a foundation for the diagnostic approach to conditions of the CTJ region that involve the brachial plexus. Those sources of anatomy information should be reviewed at this time, if necessary.
IMAGING APPROACH
Techniques and Relevant Aspects and Pros and Cons
The detailed factors related to producing optimal imaging strategies and protocols for this region of anatomy are discussed in detail in Chapters 149 and 163. In summary, ultrasound is of limited use in brachial plexus trauma when it presents with possible vascular occlusion that might be monitored with Doppler techniques. It can also be used to screen for related internal shoulder derangement. Magnetic resonance imaging (MRI) is the primary diagnostic tool in most instances of traction, blunt, and penetrating injuries. If detailed views of the dorsal and ventral root entry zones within the thecal sac are required, then volumetric computed tomographic myelography may prove more consistently definitive than MRI in cases of subtle, preganglionic nerve root injury. MRI and volumetric computed tomographic myelography are sometimes complementary, and both may be necessary.
Penetrating CTJ injuries are by definition in Zone 1 (Chapter 154) and will typically also require computed tomography (CT) angiographic screening. Catheter angiography may be necessary for diagnosis but is more typically a more detailed prelude than CT angiography to an endovascular procedure when used diagnostically.
The ideal MRI pulse sequence choices for a brachial plexus study would be those done with homogenous fat suppression across the range of the required T1 and T2 weighting necessary for a definitive study. Unfortunately, this remains a goal even when optimal receiver coil design is available because of the inhomogeneity of the B0 magnetic field related to coil loading challenges in this odd-shaped anatomic region. Newer parallel imaging water/fat separation techniques show some promise for improvement over frequency-selective fat suppression, but widespread application and validation of this approach for brachial plexus evaluation is not currently available. So, while fat-suppressed images are often “recommended” for brachial plexus evaluation, they are often unacceptably degraded and considerably prolong the scanning session with limited additional yield of diagnostic information.
Axial acquisitions are the mainstay for basic analysis. Coronal images are useful. Sagittal images are usually not necessary. Images need only be of the injured side as long as the spinal cord is included. Acquisitions must include the entire axillary sheath whenever the injury might involve the distal brachial plexus. Studies can then usually be done with closely coupled receivers, such as a dedicated shoulder coils, to produce the best images.
DIAGNOSTIC APPROACH TO BRACHIAL PLEXUS INJURY
Pathophysiology and Patterns of Injury
Blunt trauma to the upper extremity and neck with sufficient energy can cause variable brachial plexus injury. Traction injuries in which the head and neck are moved violently away from the ipsilateral shoulder are most common. Shoulder dystocia during birth is a relatively common cause of such traction injury, and fortunately the majority of injuries recover good function spontaneously. Injuries may result from compression of the brachial plexus between the clavicle and first rib as well as penetrating injuries and direct blows. Other injuries in multitrauma situations, particularly to the spinal cord and head, may delay diagnosis.1,2
Brachial plexus injuries are often catastrophic, causing permanent loss of useful function of the upper extremity. Early repair and reconstruction are recent treatment advances that have created added utility for diagnostic imaging.1,2
In traction-type injuries, it is important to establish whether the injury is proximal (preganglionic) or distal (postganglionic) to the dorsal root ganglion. Preganglionic ruptures may be central at the root entry zone junction with the spinal cord or more distal than the root entry zone but still intradural. Since the axons remain in continuity with the cell bodies in the dorsal root ganglion, preganglionic injury does not cause wallerian degeneration or traumatic neuroma formation. However, since preganglionic root avulsion causes the cell bodies of the sensory nerves to be pulled from the cord, it greatly reduces the possibility of recovery or chance of successful surgical reconstruction. The development of an injury-related pseudomeningocele strong suggests a preganglionic injury.1,2
Stretch injury frequently results in axonotmesis as seen at surgery where the damage is only to the perineurium, with the axons of the nerve remaining viable but its anatomic disruption and ensuing scarring disconnecting the nerve from its muscle. The local blocking scar is a reactive change to initial hemorrhage and other damage at the time of injury that can partially or completely block conduction to the innervated muscle groups. The healing/regenerative response of the nerve produces a neuroma when the path of the regenerating axons is blocked by such scarring. The axons then cannot grow across the nerve-injured segment. Electrophysiologic studies at the time of exploration can confirm the extent of conduction passing through the neuroma as well and identify an avulsion rather than stretch if it was not diagnosed preoperatively.1,2
Distal, postganglionic stretch injuries leave the cell bodies in continuity with their axons, thus improving the chances of functional recovery. Postganglionic lesions are physiologically similar to peripheral nerve injuries.1,2
General Manifestations and Findings
From Clinical Data
Pain in the neck and shoulder is a common complaint, and a ruptured nerve may be painful; however, avulsion may actually cause the nerve to not be reactive and therefore not tender. Other sensory complaints may include paresthesias and dysesthesias. Upper extremity weakness or heaviness may occur. Traction injury with diminished pulses indicates an accompanying vascular injury.
Sensory nerve action potentials are helpful in differentiating preganglionic from postganglionic injuries by analysis of their localizing waveforms. Electromyography can be used to confirm neurapraxia given the appropriate clinical course of the injury and timing of the test.
Somatosensory evoked potentials (SSEPs) are used diagnostically and intraoperatively to guide the specific surgical approach. The presence of an SSEP suggests continuity between the peripheral nervous system and the central nervous system at the dorsal root ganglion. SSEPs are absent in postganglionic or combined pre- and postganglionic lesions.1,2
Magnetic Resonance Imaging and Computed Tomography
Assuming that high-quality MRI and CT myelography images are available, the following findings may be seen:
- Soft tissue swelling and/or blood products around the plexus (Figs. 168.1–168.6)
- Soft tissue swelling and/or blood products within the roots, trunks, and/or cords of the plexus (Fig. 168.1A–D)
- Blood products at the site of avulsion of dorsal and/or ventral nerve roots from the cord or within or around the plexus (Fig. 168.1)
- Partial or complete disruption of the roots, trunks, and/or cords of the plexus (Figs. 168.1–168.6).
- Avulsion of dorsal and/or ventral nerve roots from the cord (Figs. 168.1–168.5)
- Meningoceles or pseudomeningoceles (Figs. 168.1–168.5)
- Compressive effects of musculoskeletal injury (Figs. 168.6)
- Various manifestations of associated vascular injury (Chapters 154 and 165) (Figs. 168.4 and 168.7)
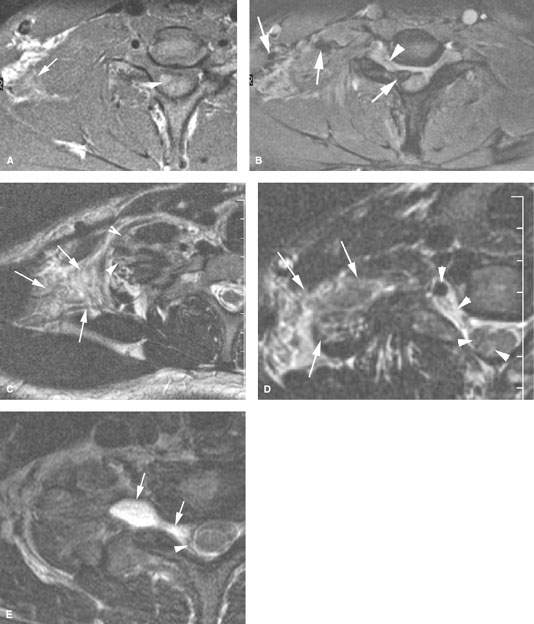
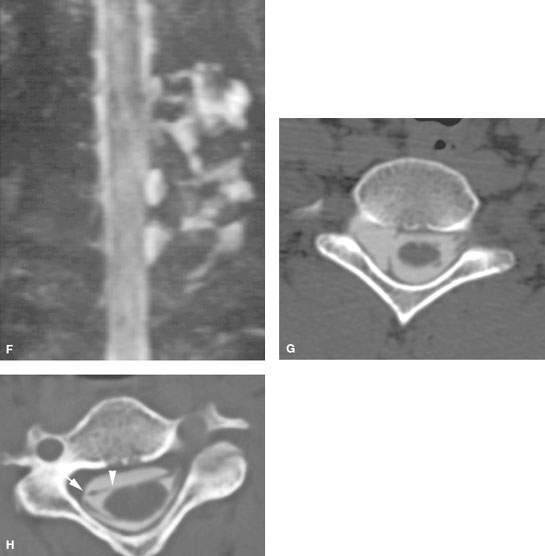
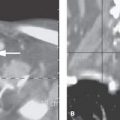
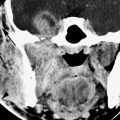
FIGURE 168.1. Five patients with preganglionic and some postganglionic brachial plexus injuries. A–D: Patient 1. The T1-weighted (T1W) image in (A) shows blood subacute products along the lateral margin of the scalene muscle (arrow) and generalized swelling within that muscle group. There is a small hematoma adjacent to the root entry zone of the cervical roots at the spinal cord (arrowhead) that is more conspicuous on some of the following images. The contrast-enhanced T1W fat-suppressed image in (B) shows blood products along the roots and trunks of the brachial plexus distally to the root entry zone at the spinal cord, where there is a discreet hematoma (arrows) proximally. An abnormal-appearing nerve root is visible within the nerve root sheath (arrowhead). The T2-weighted (T2W) image in (C) shows the extensive soft tissue swelling surrounding the trunks of the brachial plexus (arrows) and the swollen distal roots (arrowheads) as they emerge between the scalene muscles (arrowheads). The findings taken together suggest gross disruption of the root–trunk junction. The T2W image in (D) shows the extensive soft tissue swelling surrounding the trunks of the brachial plexus (arrows) as well as blood products and disrupted nerve roots within the nerve root sheath to the root entry zone, where there may be minimal cord edema (arrowheads). E: Patient 2. T2W image showing an acute posttraumatic meningoencephalocele (arrows) with some blood products at the thecal sac interface with the pseudomeningocele. F: Patient 3 with left-sided posttraumatic upper limb paralysis. Maximum intensity projection of T2W thin-sliced coronal magnetic resonance images shows posttraumatic meningoceles (arrowheads) and pseudomeningoceles (arrows) in the lower cervical and upper thoracic region; this finding strongly suggests avulsion of several brachial plexus nerve roots. G: Patient 4. The computed tomographic myelography to evaluate a complete sensory and motor dysfunction of the right arm shows a pseudomeningocele on the right (arrow). The anterior and posterior nerve rootlets visible as normal on the left (arrowheads) are not seen and presumed avulsed on the right. H: Patient 5. Computed tomographic myelography shows a blunted nerve root sheath on the right (arrow). The ventral root appears to be avulsed from the cord (arrowheads).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree