Abstract
Cystic fibrosis is a single-gene autosomal recessive disorder characterized by chronic airway infection, pancreatic insufficiency, gastrointestinal dysfunction, and male infertility. Cystic fibrosis is caused by a mutation in the cystic fibrosis membrane conductance regulator (CFTR) protein. Mutations in the responsible gene, located on chromosome 7, lead to an absent, nonfunctional, or partially functional protein, with resultant abnormal fluid and electrolyte transport across cells. Over 1000 different mutations in the CFTR gene have been identified with an expansive ethnic and racial distribution as well as varying phenotype and penetrance. The current prenatal detection rate of cystic fibrosis carriers in the general population using a standard 23 mutation panel ranges from 30%–97% depending on racial/ethnic origin. Greater than 90% of cystic fibrosis patients manifest symptoms in early childhood, often leading to an increased mortality rate in the third and fourth decades of life. Sonographic findings linked to cystic fibrosis include echogenic bowel, nonvisualization of the gallbladder, and dilated bowel; however, none are diagnostic. Prenatal carrier screening should be made available to all patients regardless of ethnicity, and invasive prenatal diagnostic testing should be considered in cases of two known parental mutations, family history of cystic fibrosis, and/or the sonographic finding of echogenic bowel.
Keywords
cystic fibrosis, CFTR, echogenic bowel, meconium ileus
Introduction
Cystic fibrosis (CF) is a single-gene autosomal recessive disorder characterized by chronic airway infection, pancreatic insufficiency, gastrointestinal dysfunction, and male infertility. Symptoms typically manifest during childhood, leading to early mortality in the third and fourth decades of life. Greater than 1000 different mutations in the CF transmembrane conductance regulator (CFTR) gene have been identified with an expansive ethnic and racial distribution as well as varying phenotype and penetrance. With advances in medical care, there are a growing number of CF patients who are advancing into childbearing years, thereby heightening awareness of strategies for prenatal carrier screening and diagnosis.
Disorder
Definition
CF is caused by a mutation in the CFTR protein, which is a cyclic adenosine monophosphate regulated chloride channel located on the apical membrane of epithelial cells. Mutations in the responsible gene, located on chromosome 7, lead to an absent, nonfunctional, or partially functional protein, with resultant abnormal fluid and electrolyte transport across cells. Direct consequences of this defect include dehydrated secretions, decreased mucous clearance in the lung, deficient secretion of pancreatic enzymes, gut dysmotility, and increased sodium chloride levels in sweat. These result in the clinical manifestations of CF, which can range from severe pulmonary and pancreatic insufficiency to an otherwise normal phenotype with only male infertility or chronic sinusitis.
Prevalence and Epidemiology
Approximately 30,000 individuals in the United States (70,000 worldwide) are currently affected with CF, and approximately 1000 new cases are diagnosed each year. CF is one of the most common single-gene disorders in the Caucasian population, with an incidence between 1 : 3000 and 1 : 3300 individuals. Both the Ashkenazi Jewish and the Hispanic population also have a relatively high incidence of the disease, whereas African American and Asian populations have a much lower disease incidence. Prenatal carrier testing for this disorder poses a challenge, as there is an expansive ethnic/racial distribution of the greater than 1000 detectable mutations. The current detection rate of CF carriers in the general population using a standard 23 mutation panel ranges from 30%–97% depending on ethnic/racial origin. Parental carrier screening is typically performed in a stepwise manner using a standardized panethnic mutation panel. Expanded mutation panels as well as complete CFTR gene sequencing are available for patients with a personal or family history of CF.
Etiology and Pathophysiology
CF is inherited in an autosomal recessive manner. The most common mutation in the CFTR gene is ΔF508, a frameshift mutation caused by a three base-pair deletion at codon 508 in exon 10 of CFTR , resulting in the absence of a phenylalanine residue. This mutation causes a protein misfold that inhibits migration of the CFTR protein from the endoplasmic reticulum to the cell membrane. Other mutations result in a spectrum of protein dysfunction ranging from the production of unstable ribonucleic acid to CFTR cell surface instability. These mutations in the CFTR protein lead to abnormal fluid and electrolyte transfer across epithelial cell membranes, causing dehydrated and viscous secretions in major end organ systems. In the lung, these secretions interfere with mucociliary clearance, obstruct airflow, and provide a growth medium for pathogenic bacteria. In the pancreas, exocrine enzymes are retained, causing progressive damage and, ultimately, fibrosis of the pancreas. The function of CFTR has been most extensively studied in the sweat gland, in which dysfunction of CFTR leads to failure of chloride resorption. This leads to a change in the normal electrochemical gradient of the cell, which typically drives sodium across the cell membrane. The resulting increase in sodium and chloride concentration in sweat is the basis for the diagnostic “sweat test” often used in children with clinical manifestations suspicious for CF.
Manifestations of Disease
Clinical Presentation
Greater than 90% of CF patients manifest symptoms in early childhood. Of them, 15%–20% present at birth with meconium ileus. Both the constellation and severity of symptoms are highly variable and somewhat dependent on the responsible mutation. While pulmonary complaints are the hallmark of this disease, they are not necessary for diagnosis. Common clinical manifestations by organ system are listed subsequently.
Respiratory tract
- •
Chronic sinusitis
- •
Chronic airway infection ( h. Influenza, s. Aureus, p. Aeruginosa, b. Cepacia )
- •
Bronchiectasis
- •
Cor pulmonale
- •
Respiratory failure
- •
Gastrointestinal tract
- •
Meconium ileus (failure to pass stool, abdominal distention, emesis)
- •
Exocrine pancreatic insufficiency (malabsorption of protein, fat, and fat-soluble vitamins)
- •
Focal biliary cirrhosis
- •
Failure to thrive
- •
Endocrine system
- •
Insulin resistance/diabetes mellitus (destruction of pancreatic islet cells)
- •
Osteoporosis (vitamin D deficiency)
- •
Reproductive tract
- •
Delayed onset puberty
- •
Azoospermia (congenital bilateral absence of the vas deferens)
- •
Imaging Technique and Findings
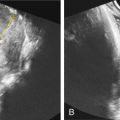
Ultrasound.
Ultrasonographic markers have been linked to the prenatal diagnosis of CF, although none are diagnostic. Echogenic bowel, defined as bowel with a sonographic density equal to or greater than that of bone, is the most well studied of these markers and is observed in 0.1%–1.8% second-trimester ultrasounds ( Fig. 141.1 ). The magnitude of risk association between echogenic bowel and CF is reported to range from 0%–33%. Other sonographic findings associated with CF include mildly dilated loops of bowel, nonvisualization of the fetal gallbladder, and complete bowel obstruction with severely dilated loops of bowel and polyhydramnios. Some studies have also suggested that meconium ileus may be detected on routine prenatal ultrasonography, with the characteristic appearance of echogenic bowel ± dilated intestinal loops. This appearance is thought to be secondary to the dehydration of intestinal secretions, which then leads to increased bowel viscosity and subsequent obstruction.