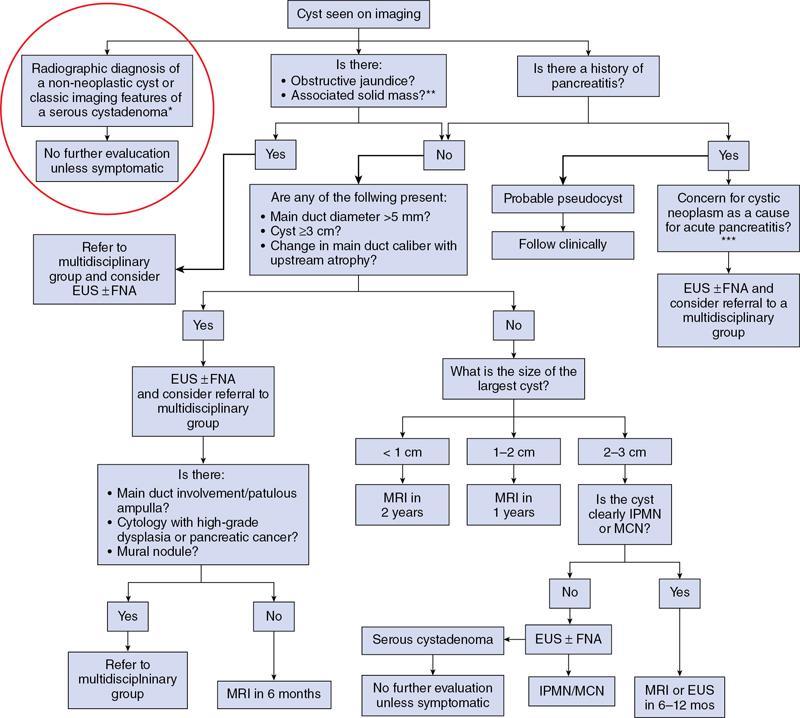
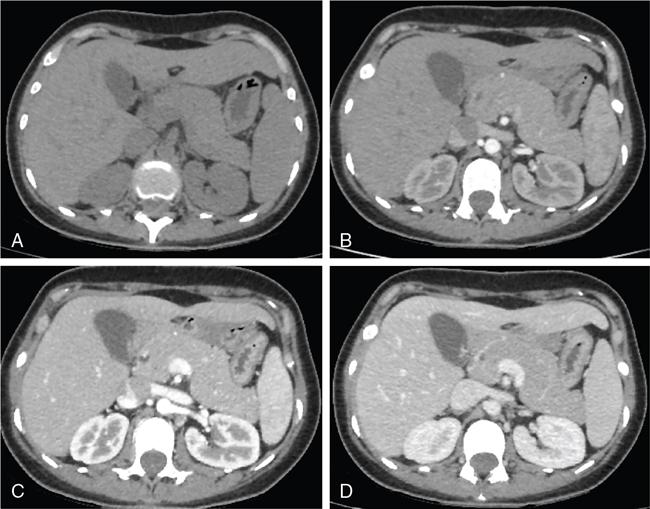
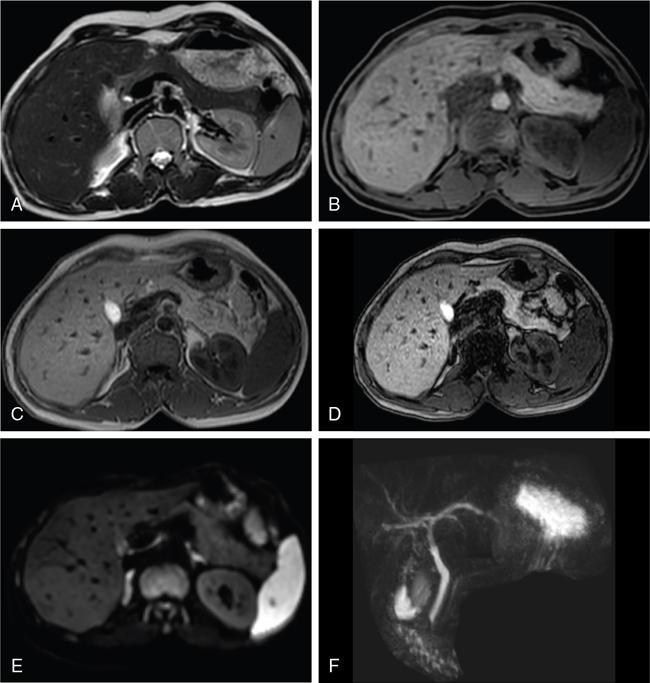
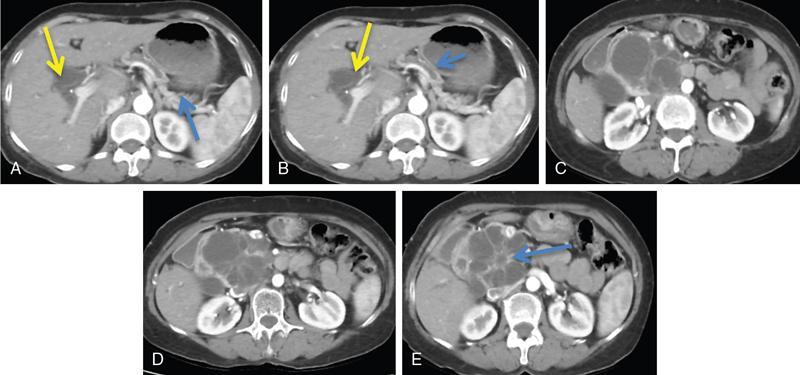
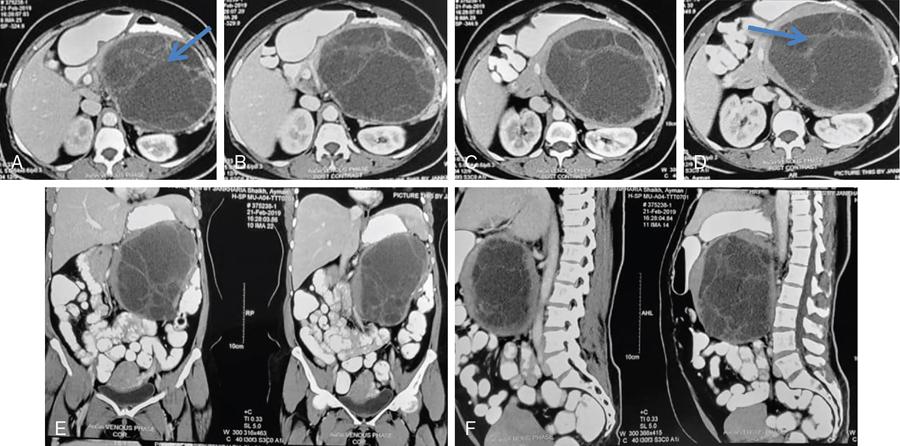
Ritu K. Kashikar, Shrinivas B. Desai, Pooja Punjani Vyas, Nilesh Doctor, Vivek Shetty Advancement and extensive use of imaging in recent times has to lead to increased detection and recognition of cystic pancreatic masses. Imaging however, plays a vital role in noninvasive diagnosis of these lesions, avoiding unnecessary intervention and planning management and follow up guidelines. The aetiology of pancreatic cysts ranges from primary cystic neoplasm to cystic degeneration of solid neoplasm to nonneoplastic cysts (Table 9.19.1). Cystic tumours of the pancreas comprise around 10%–15% of cystic lesions of the pancreas. The aim of this chapter is to educate the reader regarding imaging features of cystic lesions of pancreas, how to differentiate between cysts which can be left alone and those that require further management and also emphasize on latest international guidelines used in stratification of these lesions. Cystic pancreatic lesions are often incidentally detected on USG abdomen performed for other reasons. Although USG is an excellent modality for evaluation of cystic lesions elsewhere, it is unable to adequately characterize pancreatic cysts due to retroperitoneal location of the organ and other confounding factors such as obesity and gases. Most lesions detected on USG require characterization with either MDCT or MRI with MRCP. CEUS improves accuracy in the differentiation between a solid and a cystic lesion and also in determining whether enhancing septa or nodules are present within the cystic lesion. Currently USG also does not have a role in follow up algorithms of pancreatic cystic lesions. Multidetector CT evaluation of pancreatic lesions is best performed with a multiphasic technique (Table 9.19.2). The phases include a precontrast scan, an early arterial angiographic phase, a pancreatic parenchymal phase, and a portal venous phase. The precontrast scan is important for detection of calcification, which may be diagnostic of some lesions. Mucinous cystic neoplasms may be hyperdense on nonenhanced scan suggesting mucin content or haemorrhage. The arterial phase aids in detection of hyperenhancement neovascularity and arterial involvement in pancreatic masses. Optimal parenchymal enhancement of the pancreas is achieved at 35–45 seconds after initiation of injection of contrast agent. This is the pancreatic parenchymal phase. It is in this phase that the tumour pancreas contrast is maximum (Fig. 9.19.1, Table 9.19.3). Most hypoenhancing pancreatic lesions are best detected in this phase. The portal venous phase allows in detection of venous involvement and hepatic metastasis. Delayed phase is typically not required in cystic masses. MDCT has an accuracy of 56%–85% for characterization of cystic pancreatic lesions. MR imaging affords the best noninvasive means for the evaluation of cystic lesions of the pancreas due to superior soft tissue resolution. The helpful distinguishing characteristics of cystic pancreatic lesions, morphology of septae, relation to pancreatic duct are easier to detect at MR imaging and MR cholangiopancreatography (MRCP) than at CT (Table 9.19.4). Studies however indicate that MDCT and MRI are comparable in identifying malignant behaviour of cystic pancreatic lesions. The MRI protocol for evaluation of cystic lesions of pancreas includes T2-weighted single-shot fast spin-echo, T1-weighted in-phase and opposed-phase gradient echo, diffusion-weighted imaging, T2-weighted fat-suppressed fast spin-echo. Three-dimensional T1-weighted fat-suppressed spoiled gradient-echo T2-weighted MRCP. Postcontrast protocol includes dynamic three-dimensional T1-weighted fat-suppressed spoiled gradient-echo (in arterial, pancreatic and portal venous phases) (Fig. 9.19.2, Table 9.19.3). DWI has not found much utility in cystic neoplasms owing to overlap in ADC values. Some studies suggest role in distinguishing malignant from benign tumours in the case of mucinous cystadenoma and IPMN. Secretin is a peptide hormone produced in the intestinal mucosa, which stimulates the secretion of bicarbonate-rich fluid into the pancreatic ducts and transiently increases the tone of the sphincter of Oddi. The increased fluid distention of the pancreatic duct allows better study of ductal anatomy and identifying communication of pancreatic cystic lesions with the pancreatic duct. EUS is excellent in characterization of cystic lesions of pancreas. The proximity between the transducer and the lesions allows precise definition of the structural component of the cysts and components such as small mural nodules are better visualized with EUS than with other modalities. The other advantage of EUS is that cysts fluid aspiration and cytology can be performed. Tumour markers, genetic markers can be evaluated in the aspirated fluid. This allows comprehensive evaluation of cystic lesion. EUS also has therapeutic advantages allowing endoscopic draining of pseudocysts. Recently endoscopic ablation of cysts has been performed. These advantages have led to increasing use of EUS in recent years. This modality is however not indicated in all lesions and imaging should be able to stratify lesions requiring further invasive investigations. Studies have found PET CT comparable to PET alone or CT to determine presence of malignancy in cystic lesions. False positive findings may however be problematic. There is however no consensus for routinely using PET CT in characterization of cystic pancreatic masses. Serum CA19-9 and CEA are routinely done in all pancreatic masses. Though role in cystic lesions is still controversial. Serous cystadenoma is a benign neoplasm composed of glycogen-rich epithelial cells that form innumerable small thin-walled cysts containing serous fluid. It is the prototype microcystic pancreatic neoplasm. They occur frequently in older women (median age, 65 years) and is also called as grandmother lesion. Approximately 40% of pancreatic serous cystadenoma arise from the pancreatic head and uncinate process and 60% arise from the pancreatic body and tail. Serous cystadenomas are usually discovered incidentally at imaging; however, those that are large may cause symptoms. Patients may present with abdominal pain, palpable mass, anorexia, fatigue/malaise, or weight loss. Rarely the patient may present with jaundice. Genetic alterations similar to those in VHL are seen in sporadic SCA and include tumour suppressor gene VHL mutations and overexpression of vascular endothelial growth factor (VEGF). Allelic loss in chromosome 3 have seen in up to 40% cases of sporadic SCA. Serous cystadenomas are variable in size. The size ranges from <0.1 to 25 cm. The tumours are grossly well demarcated from the surrounding parenchyma. The gross appearance of the lesions is variable. It could be microcystic, consisting of innumerable small cysts giving the macroscopic appearance of a sponge or honeycomb. The presence of multiple, microscopic SCN is characteristic of von Hippel–Lindau (VHL). This variety show a centrally located area of fibrotic scar. In Macrocystic/oligocystic variant, borders with the surrounding parenchyma are less defined and the cystic lesion generally does not exhibit a central scar. The Solid variant, the lack of cystic spaces on gross inspection often gives the initial impression of a neuroendocrine or solid pseudopapillary neoplasm. Serous cystadenomas show nonviscous, clear-to-yellow cyst fluid, which provides an important clue to the distinction of SN from mucinous neoplasms. In contrast to intraductal papillary mucinous neoplasm, there is no connection with main or branch pancreatic ducts. These are clinically benign pancreatic epithelial neoplasms exhibiting serous morphology, characterized by a monotonous, cuboidal epithelial cell proliferation with marked cytoplasmic clearing due to intracellular glycogen. The centrally located epithelial cell nuclei are uniformly round to ovoid and not particularly large or hyperchromatic. Abundant diastase-sensitive cytoplasmic glycogen is highlighted by a periodic acid–Schiff stain. These lesions do not show evidence of necrosis, infiltrative architecture, or lymphovascular/perineural invasion, and mitotic figures are generally not observed. A variably prominent pseudocapsule of fibrosis divides parenchyma from the neoplasm On USG the microcystic variant shows lobulated contour with multiple tiny anechoic cysts separated by septae. the central scar containing calcification can be seen if present. Extremely microcystic, honeycomb variant may resemble a solid lesion at conventional US. The macrocystic type, can be mixed type with multiple large (>20 mm) and small cysts, and the unilocular type, which is more difficult to differentiate from mucinous cystadenoma (MCA). Enhancement of the intralesional sepatations is seen on contrast-enhanced USG allowing better characterization of the lesion. The central scar can show homogenous enhancement. Honeycomb variety appears as a hypervascular lesion owing to its extremely microcystic morphology and may resemble solid masses like neuroendocrine tumours. Pancreatic serous cystadenoma can have a varied appearance on CT depending on the morphologic patterns. Serous cystadenomas are typically solitary but may be multiple in von Hippel–Lindau disease, causing an appearance of disseminated involvement (Table 9.19.5). As mentioned previously, the polycystic pattern is the most common appearance of serous cystadenoma. A pattern of a bosselated collection of cysts that usually number more than six is seen. Each cyst may range from a few millimetres to 2 cm in size (Fig. 9.19.5). The other classic CT features include a lobulated contour and presence of a central stellate scar with calcification. This scar is seen in 30% cases and when present is strongly suggestive of the diagnosis. The intralesional fibrous septae show early enhancement after contrast administration. This is a distinguishing feature, as serous cystadenoma is the only hypervascular lesion among the cystic pancreatic neoplasms. Tiny cysts mimicking a honeycomb are the hallmark of this variant and may be poorly depicted as individual cysts on CT. On unenhanced CT, the honeycomb pattern may appear as a well-marginated lesion with soft tissue or mixed attenuation, depending on the size of the cysts and the amount of fibrous tissue. Moderate to strong enhancement is seen in these lesions (Fig. 9.19.6). This appears as a unilocular cyst or it may contain fewer large (>2 cm) cysts. Imaging Features include a lobulated contour, lack of a prominent thickened peripheral wall, and location in the head of the pancreas (Fig. 9.19.7). Owing to its supreme soft tissue resolution, MRI is the modality of choice in diagnosis of serous cystadenomas. The classic MRI features of microcystic variant includes a lobulated lesion with multiple small T2 hyperintense and T1 hypointense cysts with intervening hypointense fibrous sepate. These lesions do not communicate with the pancreatic duct; hence no dilatation is usually seen. Dilatation of pancreatic duct has however been reported in larger lesions. After the administration of gadolinium, the hypervascularization of the central scar and of internal septa may be seen. The morphology of the honeycomb pattern may also be better depicted on MRI. Multiple tiny T2 hyperintense cysts with intervening hypointense septae are seen (Fig. 9.19.8). The oligocystic variant shows fewer larger T2 hyperintense and T1 hypointense cysts and can mimic mucinous cystadenoma. However, the lobulated contour, together with the absence of wall enhancement and a wall thickness less than 2 mm, suggest the correct diagnosis (Fig. 9.19.9) (Table 9.19.6). The differential diagnosis of serous cystadenoma depends on the variety and are listed in Tables 9.19.7 and 9.19.8. Lesions with classic imaging features do not require further investigation or fluid analysis. The fluid in classic cystadenomas is yellow in colour and does not show elevated amylase, mucin or tumour markers. Approximately 20%–50% cases show cytological positivity for periodic acid-Schiff and cytokeratin AE1 and 3. Hemosiderin laden macrophages also do not have high diagnostic accuracy and are seen in only about 43% cases. Current management guidelines suggest (Table 9.19.9). Resection involves distal pancreatectomy or Whipples, depending on location of the tumours and is currently reserved for truly symptomatic cases. Mucinous cystic tumours are a rare subset of cystic neoplasms, constituting approximately 2.5% of pancreatic exocrine tumours. TABLE 9.19.10 The defining and characteristic histopathologic feature of mucinous cystic neoplasms (MCNs) is the presence of ovarian-type stroma similar to that observed in biliary cystadenomas. International Association of Pancreatology recommend that all suspected MCNs be surgically resected. A significant female predilection is seen with approximately 99.7% cases seen in women. As opposed to serous cystadenomas these lesions occur in middle aged women and hence the term mother lesions is used to describe these. The approximate age of occurrence is 50 years with a range from 20–82 years. Mucinous cystic tumours are a dominant cyst that is round or oval and is encapsulated. MCNs may grow slowly over time, at an average rate of 4 mm per year. The most common locations are the pancreatic body and tail (up to 75%). Stromal elements similar to ovarian stroma are the hallmark and key feature that aids in differentiation from IPMN, whose stromal elements are ductal in origin. Tall columnar cells with intracellular mucin arranged in a single row or vertically, forming papillary or polypoidal projections, constitute the epithelial elements. Portions of benign appearing epithelium can be seen adjacent to areas of invasive carcinoma in the same tumour. These lesions show smooth contour on gross appearance with few internal sepate. Peripheral calcification, mural nodule if present suggest malignant cyst (Fig. 9.19.11, Table 9.19.11). Lesions may be asymptomatic in 25% cases. Patients my present with pain in abdomen. Mucinous cystadenoma can also present with pancreatitis. Considering most lesions are located in distal body and tail involvement of splenic vein may lead to left sided portal hypertension. Mucinous cystic tumour is a well-circumscribed cystic mass in the pancreas. Lesions can have an irregular contour to the wall, septations, mural nodularity, and peripheral calcifications. The lesion may however be echogenic due to high by mucin content or haemorrhage which may impair the detection of other features. CEUS may improve detection rate of septa and mural nodules. Lesions are typically well-circumscribed with smooth contour. On unenhanced CT, mucinous cystadenoma may appear hypodense or slightly hyperdense content, due to the presence of variable amount of mucin and haemorrhage. Curvilinear calcifications occur along the periphery of the lesion and are seen in 15% of cases, in contrast study enhancement of the fibrous cyst wall along with enhancement of any septations or mural nodules is seen (Figs. 9.19.12 and 9.19.13).
9.19: Cystic pancreatic masses
Introduction
Imaging techniques
Ultrasonography (USG)
MDCT
Precontrast Scan
Arterial phase
18–20 seconds
Pancreatic parenchymal phase
35–45 seconds
Portal venous phase
60–70 seconds
MRI
Secretin-enhanced MRI
EUS
PET CT
Serum tumour markers
Serous cystadenoma
Location
Clinical features
Genetics
Histopathology
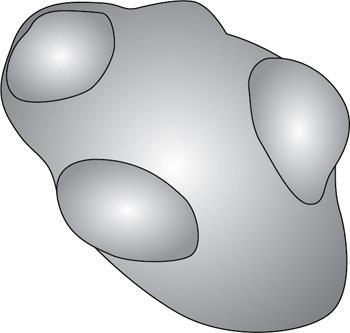
Gross appearance
Microscopic morphology
Patterns of pancreatic serous cystadenoma
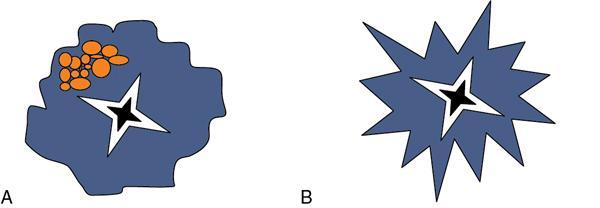
Imaging appearance
Ultrasound
Contrast-enhanced US
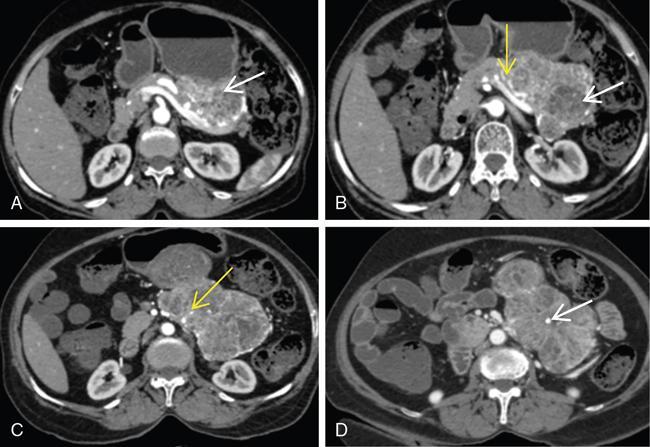
CT
Polycystic pattern
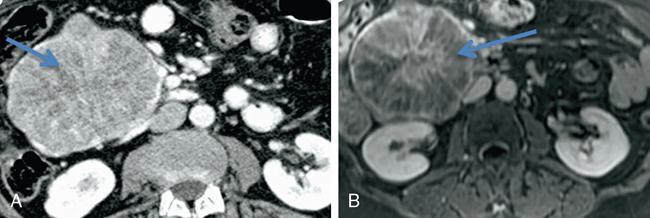
Honeycomb pattern
Macrocystic or oligocystic pattern
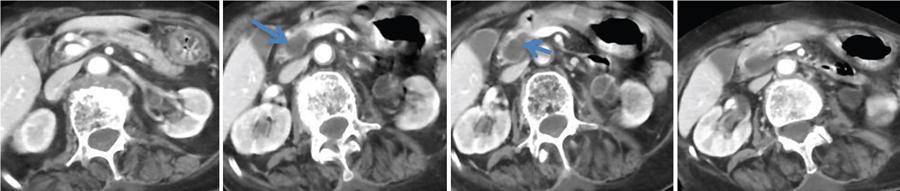
MRI features
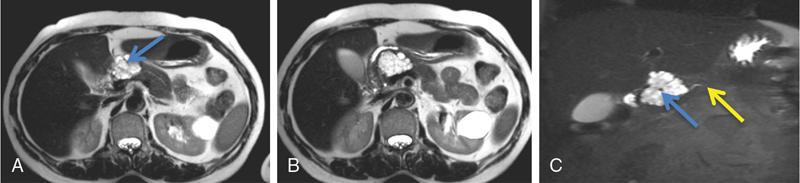
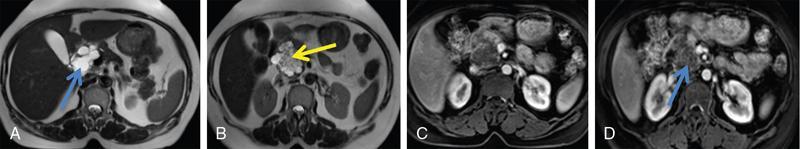
Atypical imaging appearance
Differential diagnosis
Serous Cystadenoma
Neuroendocrine Tumour
SPEN
Lobulated contour
Presence of hypervascular halo
Presence of haemorrhage
Multiple tiny cysts giving a sponge like appearance
Centre may be necrotic
Thick enhancing capsule
Serous Cystadenoma
Pseudocyst
Mucinous Cystic Neoplasm
IPMN
Lobulated contour
Smooth external contour
Smooth external contour
Pleomorphic and tubular external contour
No enhancing septae or wall
Peripancreatic stranding
Relatively thick enhancing wall, septations and mural nodule in the case of malignancy
Communication with main pancreatic duct or side-branch
No calcification in this variant
Clinical history of pancreatitis
Peripheral calcifications
Thick internal septations and nodularity suggestive of malignancy
Role for cyst fluid analysis
Management
Surgery
Mucinous cystadenoma
Epidemiology
Location
Histology

Presentation
Imaging
Ultrasound
CT
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree