Discoid meniscus (DM) is currently considered as a spectrum of disorders in meniscal shape and stability. Diagnosing DM on MR imaging has clinical and surgical implications. When diagnosing a DM on MR imaging, identification of tears and signs of instability is of utmost importance. The literature has focused on the diagnosis of DM based on morphology as complete, incomplete, and Wrisberg type, the latter lacking posterior attachments. This article reviews the relevant anatomy, histology, and clinical presentation of DM in the pediatric population.
Key points
- •
Discoid meniscus is a spectrum of disorders of meniscal shape and stability that have been classified as complete or incomplete, stable or unstable.
- •
In addition to the existing MR imaging criteria of diagnosing tears in nondiscoid meniscus, the presence of increased intrameniscal signal in discoid meniscus along with shape deformation may represent a tear.
- •
All tear types can be seen with discoid meniscus. However, the presence of a horizontal cleavage tear, especially in a child younger than 10 years, should raise the suspicion of a discoid meniscus.
- •
Shape deformation in discoid meniscus is an early sign of instability and may include surface changes, meniscal shift, and meniscal megahorn.
- •
“Pseudo–bucket-handle tear,” “crimped meniscus sign,” and parameniscal edema are all crucial imaging signs of meniscal instability.
Introduction
Discoid meniscus (DM) is a broad term used to describe a spectrum of disorders of meniscal shape and stability that can affect both men and women. Several theories have been proposed regarding the possible cause of DM. Initially, the abnormal shape of DM was thought to result from a failure of central resorption of its intermediate form during fetal development. However, a discoid shape was never found in embryologic specimens. Although associations that suggest familial predisposition, such as twin studies, have been recognized, no scientifically proven inherited risk of DM has been found. To date, the exact cause of DM remains unclear.
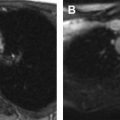
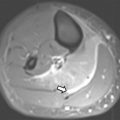
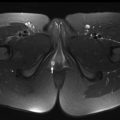
Japanese populations have the greatest incidence of DM (approximately 13%), whereas the incidence of DM in the United States has been reported as low as 3% to 5%. Because many DMs are asymptomatic or discovered incidentally, calculating the true incidence or prevalence is not possible. A discoid shape preferentially affects the lateral meniscus (0.4%–16.6%) in the general population and rarely affects the medial meniscus (0.1%–0.3%) ( Fig. 1 ). The reported prevalence of DM varies according to the patient population, selection criteria, and method of investigation. Hagino and colleagues in 2013 reported a prevalence of 17% lateral DM in a study of 94 symptomatic children at arthroscopy. Although many cases are asymptomatic or present with unilateral symptoms, the diagnosis of bilateral DM on arthroscopy ranges from as low as 3.8% to as high as 79% to 97% in young male Asian patients.

This article discusses an up-to-date MR imaging techniques, meniscal function and anatomy, classification of DM, clinical presentation, and the role of MR imaging and characteristic imaging findings of tear patterns and signs of instability in the DM in the pediatric population. In addition, up-to-date treatment information is also included.
MR imaging techniques
The MR imaging protocol to evaluate DM in pediatric patients should not differ from the routine protocol used to evaluate internal derangement of knee ( Table 1 ). However, some technical tips should be considered to optimize the study in children. Ideally and when possible, MR imaging of knee in pediatric patients should be performed on a 3-T magnet, which has the advantage of a higher signal-to-noise and contrast-to-noise ratio allowing to decreased examination time. This can minimize motion artifact, improve patient comfort, and enhance image quality by increasing spatial and contrast resolution. The MR imaging protocol on the 1.5-T magnet should include fast spin-echo (FSE) proton density sequences in all 3 planes and an FSE T2-weighted sequence with fat-suppression on the sagittal plane. FSE imaging technique, because of the shorter acquisition time, is advantageous in the less cooperative children. If the study is performed on a 3-T magnet, a 3-dimensional isotropic turbo spin-echo proton density sequence is obtained with multiplanar reformation capabilities instead of the FSE proton density sequences in all 3 planes.
| Sequence | Plane | Slice Thickness (mm)/Gap Size (mm) | Useful for |
|---|---|---|---|
| FSE PD FS | Cor | 4/0.4 | Best plane to evaluate meniscal shape/thickness by evaluating tibial plateau coverage Helpful confirming meniscal tears |
| T1 | Cor | 4/0.4 | Best plane to evaluate meniscal shape/thickness by evaluating tibial plateau coverage |
| FSE PD | Sag | 3/0.3 | Best sequence to evaluate meniscal tears Best plane to evaluate posterior meniscal attachments Helpful confirming meniscal thickness |
| FSE T2 FS | Sag | 3/0.3 | Most helpful to evaluate overall meniscal signal Helpful confirming meniscal shape and meniscal tears Helpful evaluating posterior meniscal attachments |
| FSE PD FS | Axial | 3/0.5 | Helpful characterizing complex meniscal tears and flipped meniscal fragments or meniscal shift |
| 3-D Isotropic TSE PD (3-T) | Sag | 0.6/0 | 3D capabilities allow to evaluate menisci in multiple planes Shortens scan time; however it may not be suitable in uncooperative patients Optional for 3-T scanner. It may replace FSE PD Sag and FSE PD FS axial planes |
In order to evaluate the meniscal shape and thickness, the coronal plane is probably the most useful. The sagittal plane can help confirming the shape of the meniscus but is most useful in evaluating the posterior meniscal attachments and meniscal tears. The axial plane can also be helpful in cases of radial tears or complex tears, with flipped fragments allowing good depiction of the abnormality. All 3 planes are useful in the evaluation of meniscal shift and/or displacement in the cases of instability.
Meniscal signal and tears are typically best delineated on short echo time sequences, especially proton-density sequences. The sagittal plane is the most useful; however, confirmation on the coronal plane increases confidence. The ability to detect meniscal tears is improved with high spatial resolution especially in the smaller size children. Measures such as maximizing matrix size and maintaining a small field of view and thinner slice thickness increase spatial resolution. Typical parameters required to achieve adequate resolution include a matrix size of at least 192 (phase-encoding direction) x 256 (frequency-encoding direction), a slice thickness no greater than 3 to 4 mm, and the smallest field of view possible.
The field of view in pediatric patients must be adjusted according to the age of the patient. In older adolescents with sizes similar to adults, a field of view of 16 cm or less should be used and decreased in the younger child. These modifications can decrease the signal-to-noise ratio but can be minimized by using a high field strength magnet (ie, 3-T) and increasing the number of acquisitions. Coil selection in pediatric patients of different sizes is also important to optimize the quality of the MR images. A dedicated high-performance extremity coil with 8 channels can be used in patients older than 10 years, which helps improving the signal-to-noise ratio. In younger children (4–8 years), the use of a multichannel high-performance flex coil is necessary to maintain the quality of the MR images.
Meniscal function and anatomy
The meniscus serves to lessen the impact of external forces on the knee joint and facilitate fluidity of movement to protect the specialized hyaline cartilage of the articulate surface. Its unique shape and ultrastructure are designed to relieve the stress of impact by providing structural support, joint stabilization, and shock absorption.
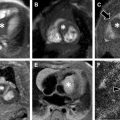
Unlike the DM, which is thickened centrally, the normal non-DM has a wedge shape that smoothly tapers centrally to form the inner free edge, partially covering and conforming to the tibial articular surfaces to allow for a smooth gliding of the femoral condyles. In normal individuals, the larger medial meniscus is more “c” shaped and has a larger posterior horn. On the other hand, the smaller lateral meniscus is more circular and maintains a more constant width, because both the anterior and the posterior horns are fairly equal in thickness ( Fig. 2 ). Except for the central free edge, both menisci have straight or slightly concave borders inward.

The lateral and medial menisci are anchored anteriorly and posteriorly by root ligaments. The root ligaments of both, the medial and lateral anterior horns, travel in the intercondylar fossa and insert on the tibia just anterior and posterior of the anterior cruciate ligament, respectively. The anterior medial root ligament is the largest meniscal attachment, whereas the strongest is the posterior medial root ligament.
The periphery of both, the medial and lateral menisci, is attached to the tibia by the coronary ligaments (also known as the meniscocapsular or meniscotibial ligaments). Unique to the medial meniscus is its firm attachment to the medial collateral ligament via extensions of its deep fibers. Conversely, the lateral meniscus has no attachments to the lateral collateral ligament. Instead, the lateral meniscus is anchored directly to the medial femoral condyle and additionally to the popliteal tendon as described later.
The popliteomeniscal fascicles are synovial bands that connect the posterior horn of the lateral meniscus to the popliteal tendon as they form the popliteus hiatus. These attachments are essential in stabilizing the lateral meniscus, acting as “meniscal struts” that fix the posterior horn in place. Specifically, the posterosuperior and anteroinferior fascicles attach the lateral-most aspect and the middle-third of the posterior horn to the popliteal tendon, respectively (see Fig. 2 ). The disruption of these fibers alone has been shown to cause a 50% increase in lateral meniscus mobility on cadavers and have been described with lateral meniscal tears when not visualized on MR imaging.
Additional support is provided by the meniscofemoral ligaments, which attach the posterior horn of the lateral meniscus to the medial femoral condyle. The anterior (or Humphrey ligament) and posterior (or Wrisberg ligament) meniscofemoral ligaments insert proximally and inferiorly to the posterior cruciate ligament on the medial femoral condyle, respectively.
Meniscus ultrastructure
Ultrastructure of the Normal Meniscus
The elastic nature and dynamic support provided by the meniscus is explained by the components of its ultrastructure. Longitudinal fibers in a circumferential arrangement composed of type I collagen bundles are concentrated at the periphery of the meniscus. These longitudinal fibers help to resist axial forces and prevent the extrusion of fibrocartilage (also known as hoop strength) during load-bearing, homogeneously distributing force. Surrounding the longitudinal fibers is a lattice of loosely organized perpendicular fibers known as radial fibers, which serve as support for the longitudinal fibers and aid in resisting longitudinal stress. This network of fibers forms a sturdy foundation for the adherence of glycosaminoglycans.
Ultrastructure of the Discoid Meniscus
DM ultrastructure differs from the nondiscoid type. In 2007, Atay and colleagues used electron microscopy to describe a heterogeneous and disorganized collagen fiber arrangement in DM, as well as a decrease in the number of collagen fibers compared with non-DM, predisposing risk factors to meniscal tears. Simultaneously, Cui and Min described 7 layers in a DM. They identified a central layer that is hypovascular, accounting for 75% of the meniscal thickness, as well as 3 symmetrically arranged layers (surface, outer, and inner) arranged in the superior-inferior direction. The central layer has a peculiar structure with peripheral fibrils that are bundled and arranged in a circular fashion, similar to non-DM, and are suited for resisting hoop stress. However, in the medial portion of the central layer, the collagen fibril arrangement is irregular and lacks the ability to provide either hoop strength or structural support. The central layer has fibrils with straight arrangement in the radial direction in the anterior and posterior zones, which strongly attach the meniscus to the tibia. This central layer also contains the middle perforating bundle, which further allows the DM to tightly anchor onto the joint capsule, thus providing stability ( Fig. 3 ).

Some of these observations were supported by Papadopoulos and colleagues, who, using light microscopy, concluded that there was no significant difference in the radial fibers in a complete discoid compared with a non-DM. The most pronounced mid-substance deformations were identified on the tibial aspect of the posterior third of discoid samples. Specific pathologic findings in the discoid collagen matrix included myxoid or cystic degeneration, osseous metaplasia, and void spaces.
Cui and Min also suggested that the presence of longitudinal fibers in the lateral middle zone of the DM, also seen in the non-DM, plays a crucial role in load-bearing and hoop strength; therefore, they should be spared when performing meniscectomy to prevent early degenerative joint disease.
Classification of discoid meniscus
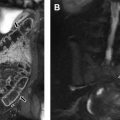
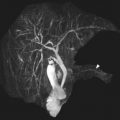
DM, by definition being a spectrum of shape and stability, is classified according to the morphology as complete or incomplete and according to stability as stable or unstable. The original Watanabe classification in 1978 described 3 types of DM based on their arthroscopic appearance. Type I DM (complete DM) is block shaped and covers the entire lateral tibial plateau ( Fig. 4 ). Type II DM (incomplete DM) is semilunar shaped and covers less than 80% of the tibial plateau (see Fig. 4 ). Type III DM (Wrisberg type) can be discoid or normal in shape but lacks posterior meniscal attachments (ie, popliteomeniscal fascicles) and is stabilized only by the ligament of Wrisberg, resulting in meniscal hypermobility ( Fig. 5 ). This meniscal hypermobility allows the Wrisberg DM to subluxate anteriorly on flexion, producing a clunk when reducing in extension, the so-called “snapping knee syndrome” ( Fig. 6 ).



More recently, the concept of stability or hypermobility has been included as one of the defining features in the DM classification. However, variability exists with respect to the definition of stability in the literature. Some investigators believe that hypermobility is the result of congenital deficiencies in the capsular attachments, as in the Wrisberg variant of DM, whereas some others believe that meniscal subluxation is the result of the posttraumatic peripheral tears of the DM or its attachments. Hamada and colleagues in 1994 suggested that DM can be symptomatic even when their surfaces remain intact due to intrasubstance tears (ie, horizontal tears not touching the articular surface) or due to intrasubstance degeneration, which causes meniscal shape deformation and abnormal meniscal motion. These, in turn, are responsible for the development of mechanical symptoms, such as locking or giving way.
The study by Klingele and colleagues in 2004 was the first one to overtly include stability as a feature in the DM classification based on arthroscopic findings. DM was classified based on the morphology as complete or incomplete, peripheral rim stability as stable or unstable, and the presence or absence of a meniscal tear ( Fig. 7 ). Klingele and colleagues defined instability as the evidence of hypermobility and peripheral detachment of a DM after systematic probing following saucerization. They found a prevalence of 28.1% for symptomatic DM, more common in the complete than incomplete type. They also analyzed the location of peripheral rim instability in DM (but did not include it in the classification) and found that it most commonly involved the anterior third (47.2%) followed by the posterior third (38.9%) and less frequently the middle-third (11.1%) of the meniscus ( Fig. 8 ). This comprehensive classification addresses the variability in the shapes, sizes, and locations of peripheral attachments and, therefore, in the degree of stability in lateral DM.


The inclusion of instability in the DM classification was echoed by Good and colleagues in 2007, which was also based on arthroscopy. They proposed that instability was present if it was possible to manually bucket or evert the meniscus or if it was possible to translate the anterior horn of the meniscus to the posterior half of the tibial plateau with a probe after saucerization. In 2007, Good and colleagues also found a higher incidence of anterior instability of 53% in DM and went further to suggest the inclusion of anterior and posterior instability in the classification. However, these classifications do not include the morphologically normal Wrisberg-type DM.
In 2009, Ahn and colleagues proposed an MR imaging classification, based on the concept of “meniscal shift” according to the anatomic location in the joint into which the meniscus is displaced. They considered this classification as complementary and helpful in treatment guidance. In addition, they defined meniscal instability in the presence of a tear larger than 1 cm, which is displaceable into the joint at arthroscopy. They postulated that meniscal instability was more frequently found with peripheral longitudinal tears that propagated anteriorly or posteriorly, allowing a large meniscal fragment to migrate freely in accordance with Smillie’s 1948 original explanation of snapping in DM. Yoo and colleagues in 2012 suggested that a DM was unstable when it could be deformed or displaced in the presence of a large enough tear. In 2016, Van Steyn and colleagues, based on arthroscopy and MR imaging findings, concluded that meniscal instability can be due to the congenital deficiencies of the meniscotibial attachments or due to posttraumatic tearing. Furthermore, they suggested that DM instability was age dependent, with congenital deficiencies probably more common in younger children, whereas posttraumatic tearing is more common in adolescents and adults.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree