Overnight in-house radiology has rapidly become an important part of contemporary practice models, and is increasingly the norm in pediatric radiology. MR imaging is an indispensable first-line and problem-solving tool in the pediatric population. This has led to increasingly complex MR imaging being performed “after hours” on pediatric patients. This article reviews the factors that have led to widespread overnight subspecialty radiology and the associated challenges for overnight radiologists, and provides an overview of up-to-date imaging techniques and imaging findings of the most common indications for emergent MR imaging in the pediatric population.
Key points
- •
Overnight subspecialty radiology coverage is now the norm. Simultaneously, MR imaging has increasingly supplanted computed tomography as the dominant modality in cross-sectional pediatric imaging. This has led to increasingly complex pediatric MR imaging performed “after hours.”
- •
Overnight radiologists face physiologic challenges stemming from disruption of circadian and homeostatic processes. Some practical strategies exist for managing sleep-cycle disturbance.
- •
General anesthesia is infrequently available and younger children may struggle with breath-holding. Strategies include short overall scan times, early acquisition of critical sequences, rapid sequences, respiratory triggering, and anxiolytics.
- •
Familiarity with the MR imaging appearance of the most common neurologic, thoracic, abdominopelvic, and musculoskeletal pediatric pathologies is critical when consultation with clinical colleagues overnight is limited.
- •
Future study is necessary to elucidate the impact of overnight subspecialty imaging on patient outcomes, trainee education, and radiologists’ health.
Introduction
Overnight in-house imaging is the fastest growing component of contemporary practice models for both private practice and academic radiologists. Whereas earlier in the decade, finalized overnight interpretations were a rarity, today they are increasingly the norm. The drive to attending overnight coverage stems from several factors, which include demand from emergency departments and administration, perceived clinical outcome improvement, and quality metrics data. Although the high accuracy of preliminary resident interpretations has been repeatedly demonstrated, overnight attending coverage may increase emergency room turnaround times, improve clinical outcomes, and increase clinician confidence in interpretation. Particularly in the emergency department, even a few discrepancies between preliminary and finalized interpretations may have a major clinical impact. It is self-evident that accurate, real-time interpretation of overnight imaging is crucial to patient care.
At the same time, the volume of MR imaging studies has continued to increase nationwide. This is particularly true with respect to overnight imaging. It was previously acceptable for MR imaging technologists to be “on call” in the event that MR imaging was required. However, now, most large imaging centers staff several magnets at all hours. This is particularly pertinent in pediatric imaging, in which the ionizing radiation of computed tomography (CT) is avoided when possible.
The combination of increased MR imaging volume in pediatric radiology and overnight subspecialty coverage has led to increasingly complex imaging “after hours.” This, in turn, has led to new challenges for after-hours radiologists.
Radiologist challenges and practical strategies in overnight imaging
There is increasing interest in the effects of sleep and sleep-deprivation, as it is estimated that approximately 20% of workers in industrialized nations undergo some disruption of the sleep-wake cycle. Sleep is primarily controlled by the suprachiasmatic nucleus of the hypothalamus, an internal clock that regulates sleep through the production of the hormone melatonin. This internal clock is in turn regulated by various external stimuli, most notably light exposure. Alteration of normal circadian rhythms results in physiologic, cognitive, and psychological disturbances.
Although there has been no formal study of the effects of overnight shift work on radiologists and radiologic interpretations, the consequences of circadian disruption has been studied in other medical and nonmedical disciplines. Prospective studies and several meta-analyses have demonstrated higher rates of coronary heart disease, primary malignancy (breast, lung, and digestive), stroke, and metabolic syndrome in overnight workers. Psychological and mental health challenges are posed as well. A longitudinal study of 819 overnight emergency room physicians reported increased fatigue, mood decrement, irritability, and worsened job satisfaction. Socially, overnight workers may feel isolated from family and friends and find it difficult to integrate with their daytime peers.
There is strong evidence that disruption of the sleep-wake cycle has deleterious cognitive effects as well. Field studies in nonmedical high-reliability organizations demonstrate decreased performance on objective cognitive tests following an overnight shift. This also appears pertinent in medical fields. Nurses on overnight shifts are noted to have a higher number of patient-related errors, especially if shifts exceed 12 hours in duration, and faculty anesthesiologists demonstrate decreased memory after overnight call. Although there are no data on radiologist cognition and perceptive error during an overnight shift, the negative impact of fatigue on radiologists’ performance has been repeatedly demonstrated.
Given these physical, psychological, and cognitive challenges, it is clear that evidence-based, practical strategies are required, as recently described by Rohatgi and colleagues. Briefly, these strategies include those that promote wakefulness during the overnight period (through light exposure, brief exercise, or caffeine) and restfulness during the day (through avoidance of light, bedroom optimization, avoidance of caffeine, or exogenous melatonin). The optimal shift schedule for overnight radiologists is an essential direction of future inquiry.
Imaging technique
Patient preparation is crucial for successful pediatric MR imaging in the overnight setting. Because anesthesia and child life services are infrequently available, both patients and sequences must be selected to maximize the likelihood of obtaining a diagnostic study. Children younger than 7 years may struggle with longer examinations, breath-holding, or both during MR imaging examination. Successful MR imaging in this setting thus limits study length, acquires the most crucial sequences first, uses short sequences potentially with limited K-space sampling, and uses respiratory triggering when feasible. Parents are usually able to indicate which pediatric patients can tolerate MR imaging, but the child also can be assessed by the radiologist (frequently during a preceding ultrasound) or MR imaging technologist. Video goggles and anxiolytics may be beneficial in some pediatric patients. However, those unable to tolerate MR imaging may have to be evaluated by other modalities.
Spectrum of MR imaging findings
Neurologic Disorders
Ventricular shunt failure
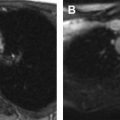
Hydrocephalus represents the most common surgically correctable neurologic problem in the pediatric population, with an incidence of 1 in 500 newborns. The underlying causes of hydrocephalus are numerous, ranging from congenital defects, such as X-linked aqueductal stenosis, to acquired postinfectious or posthemorrhagic etiologies. From the point of view of the overnight or emergency imager, however, the child with hydrocephalus is generally already undergoing treatment, and the question is whether or not that treatment has failed. It is notable that children with treated hydrocephalus represent a disproportionate number of hospital admissions nationwide. It is estimated that ventricular shunts fail at a rate of 30% to 40% in the first year, and 5% every subsequent year. At our institution, a so-called “ventricle-check” is the most common indication for overnight neuroradiology with MR imaging. Affected children typically present with symptoms of worsening hydrocephalus, most commonly headache, nausea, vomiting, lethargy, confusion, or seizure.
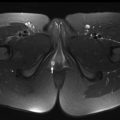
Characteristic MR imaging findings of shunt failure have been described. Most commonly, the ventricles are enlarged, but it is worth recognizing that an interval increase in ventricular size is much more important than size in isolation. Morphologically, children with worsening hydrocephalus tend to expand their occipital horns before frontal horns, and a comparison of occipital-to-frontal ratios may be beneficial, especially when comparing between different imaging modalities. A shunt also may fail without ventricular enlargement or distortion. In this setting, secondary signs of failure include enlargement of old ventriculostomy tracts, increasing fluid around the shunt, transependymal T2-prolongation (edema), or increasing subgaleal fluid ( Fig. 1 ). The catheter itself should be assessed for its integrity, intraventricular placement, and the presence of any loculations that may have formed around the tip. An optimally placed catheter tip is either centered in a lateral ventricle or superior to the Foramen of Monro, with catheter tips abutting the ventricular surface or septum pellucidum associated with higher rates of failure. Shunt migration is particularly problematic in young children, in whom normal growth may alter the position of the tip.

Several pitfalls exist when imaging suspected shunt failure in children. First, because children are growing, ventricular size also may increase naturally with time. In this setting, the radiologist or clinician may correlate with rapid changes in head circumference (so-called “growing off the curve”), ventricular disproportion can be assessed subjectively, or 2 studies may be coregistered and fused to account for the change in circumference. Coregistration and fusion also may be helpful when comparing examinations obtained in different imaging planes. Finally, it is critical to recognize that ventricular size alone is of little significance without reference to prior studies, which may be unavailable overnight. Shunted children have altered ventricular volume and compliance, and their baseline ventricular size may be slitlike or ballooned. For such children, “normal” ventricular volumes commonly coexist with shunt failure.
Failed endoscopic third ventriculostomy
Endoscopic third ventriculostomy (ETV) is increasingly performed as a first-line treatment of pediatric hydrocephalus due to the higher risks of failure and infection with ventricular shunts. In ETV, a surgical incision is created endoscopically at the floor of the third ventricle anterior to the mammillary bodies, placing the third ventricle and interpeduncular cistern in continuity. This is particularly used in the setting of aqueductal stenosis or posterior fossa tumor. Most (90%) children with failed ETV typically present within the first 16 days following surgery, with symptoms of worsening hydrocephalus.
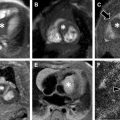
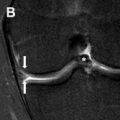
MR imaging findings of failed ETV are similar to those of a failed ventricular shunt: enlarging ventricles, particularly in the occipital horns, and transependymal T2-prolongation. A highly T2-weighted, sagittal sequence or reformat is required to assess for turbulence-related susceptibility artifact across the defect in the third ventricular floor. The absence of transspatial susceptibility in the setting of worsening hydrocephalus suggests ventriculostomy closure. A significant pitfall is that, even in the setting of a closed ETV, turbulence may occur at but not across the third ventricular floor; for this reason, a sagittal sequence or reformat is highly recommend ( Fig. 2 ).

Pediatric hyperacute and acute stroke
Pediatric stroke is an increasingly recognized cause of morbidity and mortality among children worldwide. Although rare overall, with an incidence of 2.3 to 13 in 100,000 children, pediatric stroke remains one of the most common causes of childhood death, and is particularly common in the neonatal period (1/5000 children). Boys are affected more than girls, and black children in particular.
A full classification and description of pediatric stroke is complex and beyond the scope of this article, but familiarity with common schema is helpful. Briefly, neonatal stroke occurs in the first 29 days following birth, and childhood stroke occurs between 29 days and 18 years. Unlike the adult population, in which ischemic stroke predominates, pediatric stroke is evenly distributed between ischemic and hemorrhagic subtypes. Ischemic stroke is then further subdivided into arterial ischemic stroke (AIS) and cerebral sinovenous thrombosis (CSVT). Clinically, neonates with perinatal stroke have subtle symptoms and may appear asymptomatic, whereas children with hyperacute or acute AIS present with a focal deficit localizing to an arterial distribution. In contrast to AIS, most children with CSVT and hemorrhagic stroke present with nonspecific symptoms, including headache, papilledema, nausea, and seizures.
MR imaging findings of childhood stroke are well described. Hyperacute ischemic arterial stroke demonstrates elevated signal on diffusion-weighted imaging (DWI), generally in the anterior circulation, with involvement of both anterior and posterior arterial circulations suggesting a proximal cause such as carotid dissection. In the late hyperacute and acute phases territorial T2-prolongation can be detected, initially more conspicuous on fluid-attenuated inversion recovery (FLAIR) sequences than conventional T2-weighted sequence ( Fig. 3 ). Cortical vessels may enhance conspicuously in the hyperacute phase, due to slow flow, with cortical enhancement typically present after 5 days. In hyperacute and acute CVST, the thrombosed venous sinus may demonstrate an absence of normal flow void on conventional spin-echo sequences. During the subacute phase, a degree of T1-shortening is typically present within the clot. Susceptibility-weighted sequences typically demonstrate prominent susceptibility, increasing sensitivity. Addition of 2-dimensional time-of-flight venography and 3-dimensional (3D) contrast-enhanced gradient-echo further increases both sensitivity and specificity for thrombus, in which a filling defect can be visualized in the otherwise enhancing dural venous sinus ( Fig. 4 ). Parenchymal changes of CVST include parenchymal edema or infarct in a venous or watershed distribution. Last, hyperacute or acute hemorrhagic stroke demonstrates parenchymal or subarachnoid blood products, with the former showing prominent T1-shortening and susceptibility with surrounding edema, and the latter demonstrating sulcal susceptibility artifact and loss of cerebrospinal fluid suppression on FLAIR. A causative arteriovenous or cavernous malformation also may be identified.


The imaging findings of childhood and neonatal stroke overlap, with 2 caveats. First, the brain of a neonate is incompletely myelinated and appears edematous on T2-weighted images or FLAIR, which also influences T2 shine-through on DWI. As such, apparent diffusion coefficient (ADC) maps and susceptibility-weighted sequences are particularly useful. Second, findings of neonatal stroke evolve rapidly on MR imaging, and care should be given with respect to “dating” perinatal injury given the potential medicolegal ramifications.
Craniocervical junction injury
Children are particularly susceptible to craniocervical junction (CCJ) injury due to their disproportionate head-sizes, ligamentous laxity, and underdeveloped skeletal articulations. The younger the child, the more likely they are to undergo craniocervical injury, and beyond 10 to 12 years, a child’s likelihood of CCJ injury is equal to an adult’s. The most common cause of CCJ injury in young children is motor vehicle collision. Injury may occur at the atlantoaxial and/or atlanto-occipital joint. Affected pediatric patients typically present with neck pain, torticollis, or neurologic signs of nerve root or cervicomedullary impingement. Severe injury at the atlanto-occipital joint is often fatal.
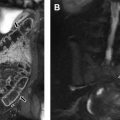
MR imaging is typically performed in conjunction with cervical spine CT to characterize soft tissue or spinal cord injury, the presence of which alters prognosis and changes management. When MR imaging is performed, it frequently shows injury where CT does not. Imaging findings of ligamentous injury include a thickened ligament that may be discontinuous and outlined by fluid. In the setting of atlantoaxial distraction, the alar ligament is abnormal and the odontoid-lateral mass interval may be widened on the ipsilateral side ( Fig. 5 ). In atlanto-occipital dislocation, the basion-dens interval becomes widened (typically >10 mm), fluid fills the widened (>5 mm) atlanto-occipital joints, and the alar ligament and/or tectorial membrane are abnormal. Direct spinal cord injury can be implied by parenchymal T2-prolongation (edema), edema accompanied by susceptibility artifact (contusion), or cord discontinuity (transection).

Thoracic Disorders
Pulmonary embolism
Pulmonary embolism (PE) is underrecognized in the pediatric population. Although rare overall, with an incidence of 1 per 100,000 children annually, PE occurs in hospitalized children at a rate of 8.6 to 57 per 100,000. Although there are no validated clinical tools for predicting the likelihood of PE in children, several risk factors are known, including obesity, immobility, congenital heart disease, surgery, trauma, central line placement, and prolonged total parenteral nutrition. These risk factors vary by the age of the child, and multiple risk factors are often present simultaneously. In children, classic symptoms, such as pleuritic chest pain, dyspnea, and hemoptysis, are frequently absent. Pediatric PE may even be clinically silent.
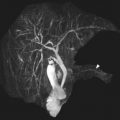
MR imaging characteristic of PE vary with technique. These most commonly include pulmonary MR angiography (MRA) and pulmonary perfusion MR imaging ( Fig. 6 ). In conventional MRA, which may be performed either as a postgadolinium or time-of-flight acquisition, PE appears as a filling defect within the enhancing pulmonary arteries. In pulmonary perfusion MR imaging, dynamic T1-weighted acquisitions are acquired iteratively after gadolinium administration, and then a precontrast sequence is subtracted from the postcontrast sequence with maximal lung parenchymal signal. In this technique, PE appears as a wedgelike hypointense defect surrounded by enhancing lung parenchyma. MR imaging can also add value over CT by visualizing the physiologic consequences of PE. Quantitative measurements of the pulmonary circulation, right ventricular ejection fraction, and pathologic cardiac motion all may characterize clinical severity, helping to guide management.

Treatment options in children with PE include thrombolysis and anticoagulation with heparins and vitamin K antagonists. In patients for whom thrombolysis is contraindicated, mechanical or surgical thrombectomy can be performed. It is worth noting that, although the sensitivity and specificity of MR imaging for PE (85% and 95%) are slightly lower than CT angiography, particularly in the distal pulmonary arteries, this discrepancy had no impact on adult mortality in a recent prospective study.
Aortic dissection
Aortic dissection is exceedingly rare in the pediatric population, with 3 notable exceptions: children with traumatic chest injury, connective tissue disorders, and congenital heart disease. Dissection is more common in adolescence rather than early childhood. Clinically, affected children present with confusion, weakness, neurologic disturbances, and hypertension. Migratory pain, which changes as the dissection propagates, may be the most specific clinical clue.
MR findings of aortic dissection have been described. Diagnosis is based on detection of the intimal flap that separates the aorta into true and false lumina. The flap appears as a hypointense line that is straight, arclike, or S-shaped in the axial plane. Generally, the true lumen is smaller than the false, and demonstrates higher signal intensity owing to the higher concentration of contrast material in the arterial phase. Thrombus typically appears as a filling defect with intrinsic T1 shortening, most commonly in the false lumen near the distal reentry point of the dissection. Mural thrombus also typically demonstrates T1 shortening. Three-dimensional reconstructions may be helpful in assessing the relationship of the dissection to arteries originating from the thoracic and abdominal aorta.
Aortic dissections that involve the ascending thoracic aorta (Stanford type A) require emergent surgical intervention to prevent extension into the coronary arteries, aortic root, or pericardium. Dissections isolated to the descending thoracic aorta (Stanford type B) may be managed medically, unless complicated by organ damage or uncontrolled pain.
Vasculitis
Pediatric vasculitides are multisystem processes with protean clinical manifestations and variable severity. Vasculitis is rare among children, with an overall incidence of 23 per 100,000, and the most commonly encountered variants are Henoch-Schönlein purpura (49%), Kawasaki disease (KD, 23%), and Takayasu arteritis (TA). Both KD and TA have thoracic manifestations, with KD (or suspected KD) more commonly imaged in the overnight setting due to the threat of coronary artery aneurysms and possible rupture. KD has a triphasic clinical course. In the acute setting, children with KD present with lengthy fever, conjunctivitis, mucosal erythema, gastrointestinal upset, and rash. In the nonfebrile, subacute phase of the illness, children are more likely to develop aneurysms, and are at the highest risk for sudden death. The convalescent phase of KD may last months to years. The presentation of TA depends on the arterial distribution.
Emergent thoracic MR imaging can be performed for vasculitis if KD is suspected and ECHO is limited. This is particularly helpful in the distal coronary arteries, for which the acoustic window is small. MR imaging findings of KD vary by clinical phase. In the acute phase, the myocardium, pericardium, endocardium, valves, and arteries may be involved, with patchy T2-prolongation and increased enhancement. During the subacute phase, coronary artery aneurysms predominate, and nonvascular abnormalities regress. Due to diminished spatial resolution, MR imaging is probably less sensitive than CT for small aneurysms. Findings of TA include vessel wall thickening, mural edema, and enhancement in the aorta and aortic arch branches ( Fig. 7 ).

The goal of treatment in KD is to reduce systemic inflammation and the risk for aneurysm. The recommended regiment of the American Heart Association is high-dose aspirin (80–100 mg/kg per day) and intravenous immunoglobulin (2 g/kg) within the first 10 days of disease. Unlike most vasculitides, the role of steroids in KD is uncertain. By contrast, TA is treated with high-dose steroids and angioplasty as necessary.
Abdominopelvic Disorders
Appendicitis
Acute appendicitis is the most common indication for abdominal surgery in the pediatric population, and a major cause of hospitalization. Appendicitis has been increasing in frequency in most recent population studies, occurring at an annual rate of 9 per 10,000, and most commonly affecting patients between 10 and 19 years old. Despite the increasing incidence of this entity, clinical diagnosis remains challenging. Right lower quadrant pain, nausea, vomiting, fever, and leukocytosis are typical signs and symptoms of acute appendicitis, although at least 50% of patients present atypically. Moreover, difficulty in eliciting history in younger children and broader differential considerations could lead to a delay in diagnosis and substantial morbidity and mortality. Various risk stratification algorithms, including the Pediatric Appendicitis Score and the Alvarado Score, are helpful in determining which patients warrant immediate surgical and imaging evaluation.
The diagnostic accuracy of MR imaging in cases of suspected appendicitis has been well documented, with a sensitivity of 96.5% and specificity of 96.1%. The MR imaging findings most strongly associated with acute appendicitis include an enlarged appendiceal diameter, focal periappendiceal inflammation, and diffusion restriction of the appendiceal wall. The presence of an appendicolith and/or a periappendiceal abscess are also very specific. Although an appendiceal diameter greater than 6 mm is present in most positive cases of appendicitis, size alone is not a sufficient criteria for diagnosis, as normal appendiceal size is highly variable in the pediatric population. Periappendiceal fat-stranding is a particularly helpful sign in early or mild appendicitis, and appears as irregular, intermediate-intensity signal around the appendix on T2-weighted sequences without fat saturation. By contrast, focal fluid is best seen on T2-weighted sequences with fat suppression. An appendicolith, if present, appears as a round signal void, usually at the junction of the normal and inflamed appendix ( Fig. 8 ). In the case of perforated appendicitis, the appendicolith may be intraperitoneal or within a mature or developing abscess. Wall thickening greater than 2 to 3 mm and submucosal hyperenhancement on postcontrast imaging are also helpful findings when present.


Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree