DISORDERS OF OLFACTION
KEY POINTS
- Magnetic resonance imaging is the primary tool when looking for a structural cause for first cranial nerve dysfunction along the entire conductive and sensorineural pathway.
- Computed tomography may be used if the pathology is likely to involve the sinuses or nasal cavity conductive pathway.
- Imaging cannot be trusted to confidently exclude all conditions that may lead to olfactory or taste dysfunction.
- The causative pathology may be of the end organ innervated by the cranial nerve rather than the nerve itself or its central nervous system origin.
- Diagnostic imaging, even performed optimally, fails to find a reason for olfactory and taste dysfunction in many patients.
Disorders of olfaction may affect the sense of smell and/or taste. Disorders of smell include anosmia, an inability to detect odors; hyposmia, a decreased ability to detect odors, as well as dysosmia, and parosmia and phantosmia, which are altered perceptions of odors. Disordered taste includes ageusia, the inability to taste; hypogeusia, a decreased ability to taste; and dysgeusia, a distorted sense of taste.
The most common causes of primary olfactory deficits are sinonasal disease, prior viral upper respiratory infections, and head trauma.
APPLIED ANATOMY
Since olfactory dysfunction can result from pathologic processes at any level along the olfactory pathway, from the nasal cavity to the brain, they can be thought of as conductive or sensorineural defects, similar to the organization of diagnostic thinking in otologic dysfunctions. This is based on the relative function of the anatomy in the linkage. Conductive losses are due to transport or transmission defects of the odorant stimulus to the olfactory neuroepithelium distributed along the upper nasal septum. Sensorineural losses are due to more central neural structure failure or the proximal connections of that neuroepithelium.
Peripheral Segment/Innervation End Organ
The olfactory tract (cranial nerve I [CN 1]) sends nerve endings through approximately 20 perforations in the cribriform plate. These innervate a dime-size area of neuroepithelium on both sides of the upper nasal septum (Fig. 78.13).
Olfactory nerve endings, by penetrating both the ethmoid and the anterior fossa dura, offer only minimal resistance to the spread of some infections and neoplasms from the upper reaches of the nasal cavity into the anterior cranial fossa. Also, the tumors arising from olfactory apparatus nerve endings, such as olfactory neuroblastoma, are likely to project both into the nasal cavity and across the cribriform plate.
Foraminal/Skull Base Segment
The olfactory tract and bulb are intradural while the nerve endings cross both the dura and the cribriform plate, finally residing in the mucosa of the olfactory cleft (Fig. 78.13).
Cisternal Segment and Projections to the Brain
The proximal olfactory tract emerges from the basifrontal portion of the frontal lobe in the olfactory sulcus. More proximally, the olfactory apparatus communicates with the limbic system. The distal part of the olfactory tract, the olfactory bulb, rests in the olfactory fossa and along the surface of the cribriform plate (Fig. 78.13).
Axons from the neuroepithelium travel to the five parts of the olfactory cortex that include (a) the anterior olfactory nucleus that connect the paired olfactory bulbs through the anterior commissure; (b) the olfactory tubercle; (c) the pyriform cortex, which is the main olfactory discrimination region; (d) the cortical nucleus of the amygdala; and (e) the entorhinal area that projects to the hippocampus.
IMAGING APPROACH
Techniques and Relevant Aspects
General imaging parameters used in CN I neuropathy or other causes of olfactory dysfunction require very high detail coronal images from the frontal sinus to at least the posterior corpus callosum. This will ensure that the entire pathway of olfaction and the limbic system, as well as the pituitary and hypothalamic region and anterior visual pathways, are studied definitively. Such techniques should include the sinonasal region, the “end organ” of olfaction, and the adjacent anterior and central skull base. Overall, magnetic resonance imaging (MRI) is best suited to the task of initial evaluation, with multidetector computed tomography (MDCT) held in reserve and focused on a specific area for problem solving when required.
Specific protocols for computed tomography (CT) and MRI studies for investigating a CN I neuropathy or disorder of olfaction appear in Appendixes A and B.
Diagnostic catheter angiography or radionuclide studies are almost never necessary.
Pros and Cons
MRI is particularly well suited to definitive evaluation of the olfactory bulbs and tracts, the gyrus rectus and olfactory sulcus, and the remainder of the limbic system. It is also the definitive examination of adjunctive areas of interest such as the anterior visual pathways, pituitary gland, and hypothalamus. MRI is exceptional in its ability to identify subtle brain changes such as migrational abnormalities that may contribute to accurate diagnosis. MRI is also far more sensitive to meningeal pathology compared to CT. MRI also provides an exceptional view of the olfactory sensory mucosa along the upper nasal septum and the nasal cavity with the exception of bone detail.
Since MRI does not provide definitive bone information, MDCT can be used when definitive information about the bone detail of the anterior skull base is necessary. CT also provides an exceptional view of the olfactory sensory mucosa along the upper nasal septum and the nasal cavity including bone detail.
In summary, if the condition is likely to be intracranial and either purely intra-axial or caused by leptomeningeal pathology, MRI is clearly superior to CT for the detection of those processes. If the disease is more likely sinonasal, CT is an appropriate starting point and would provide at least gross intracranial screening.
PATHOPHYSIOLOGY AND PATTERNS OF DISEASE AS SEEN ON MAGNETIC RESONANCE IMAGING AND COMPUTED TOMOGRAPHY
The specific pathologies that involve the cranial nerves are variable but can be grouped by three broad mechanisms of disease, including primary neurogenic problems of the brain or distal cranial nerve pathway of one of several possible etiologies as diverse as congenital, traumatic, degenerative, and neoplastic; pathology that causes a compressive neuropathy; and infiltrating neuropathies (Figs. 94.1–94.3). It is essential to first determine the specific localization of the offending pathology; this is logically and perhaps best approached by a systematic evaluation of the nerve from the cranial nerve brain origin to its end organ (s) that generate the signs and symptoms of disease. Following that, a morphologic evaluation of the disease process will almost always lead to a specific diagnosis or very short list of possibilities.
Cranial Nerve Origin
It is important to recall that the entire olfactory pathway will atrophy with age. This atrophy will parallel that of the limbic system and mesial temporal lobes. It is not surprising that neurodegenerative processes that affect those portions of the brain disproportionately will also cause more accelerated atrophy of the entire olfactory system (Fig. 94.2).
This intracranial segment is typically diseased due to developmental abnormalities, intra-axial or extra-axial brain tumors, effects of trauma, neurodegenerative disease, and rarely some unusual infectious disease (Figs. 94.1–94.3). The clinical localization may be aided by accompanying frontal lobe, endocrine, or visual dysfunction. When these additional circumstances are seen in combination with loss of olfaction, the pathologic process is likely more in the realm of central nervous system pathology than extracranial head and neck pathology.


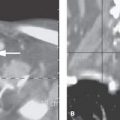
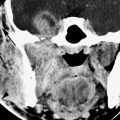
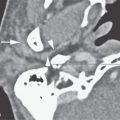
FIGURE 94.1. A–E: Computed tomography (CT) study in Patient 1, who has an absent septum pellucidum. In (A), the coronal CT shows the absent septum pellucidum. In (B), CT more anteriorly suggests poor development of the olfactory sulcus and gyrus rectus region (arrows). In (C) through (E), the series of T2-weighted images from anterior to posterior shows that the olfactory tracts and bulbs are severely underdeveloped or absent (arrows) and the olfactory sulcus is not developed properly (arrowheads). This should be compared to normal anatomy in this region seen in Figure 78.13. F–H: Magnetic resonance imaging of Patient 2 with Kallman syndrome. In (F), the anterior pituitary is difficult to identify, and the posterior pituitary (arrow) is ectopic. In (G), the olfactory sulcus is not developed (arrows). In (H), the coronal image confirms the lack of olfactory sulcus development and imperceptible proximal olfactory tracts.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree