Introduction
This chapter has two components: the first reviews the ultrasound assessment of the scrotal contents, and the second describes the role played by ultrasound in the diagnosis of penile abnormalities, including erectile dysfunction and priapism. In both sections, emphasis is given to color Doppler imaging and Doppler spectral analysis.
The Scrotum
Anatomy and normal sonographic features
The anatomy of the scrotum, testicles, and epididymis is illustrated in Figs. 30.1 and 30.2 . As seen with ultrasound, each testicle is homogeneous and medium in echogenicity ( Fig. 30.3 ), with a smooth outer border encapsulated by the thin layer of tunica albuginea. In adults, each testicle measures 3 to 5 cm in long axis and 2 to 3 cm in short axis. The testicles are relatively hypoechoic before the age of puberty and achieve adult echogenicity thereafter. The mediastinum testis is visible as an echogenic band running along one side of the testicle. The epididymis is similar to or slightly less echogenic than the testicle. Its echotexture may be heterogeneous.


The arterial and venous anatomy of the testicles is illustrated in Figs. 30.4 and 30.5 . In postpubertal boys and adults, blood vessels are normally visible within and at the periphery of the testicle with color Doppler ( Fig. 30.6 ). The capsular arteries, which course around the periphery, give rise to the centripetal arteries, which penetrate the parenchyma. Thus blood flow in the centripetal arteries is from the capsule inward. Testicular veins follow the same pattern as the arteries and are generally readily visualized and can be differentiated from the arteries with spectral Doppler. In some normal individuals, one or more large artery/vein pair(s) may traverse the testicle obliquely from the mediastinum. These transmediastinal vessels may be visible on gray-scale imaging and should not be mistaken for pathology.



Arterial flow to the testicle and epididymis characteristically exhibits a low-resistance pattern on spectral Doppler, including continuous flow during diastole ( Fig. 30.7 ). In contrast, a high-resistance blood flow pattern is seen in extragonadal arteries, which are part of the cremasteric system. These arteries are occasionally visualized along the course of the spermatic cord. It is important not to mistake extragonadal Doppler flow signals for testicular flow. Peak systolic velocity in testicular arteries ranges from 4 to 19 cm/s (mean 9.7 cm/s), and end-diastolic velocity ranges from 1.6 to 6.9 cm/s (mean 3.6 cm/s). These values permit quantitative assessment of arterial flow when a sufficiently long arterial segment is visualized with color Doppler imaging, allowing for angle correction of the Doppler signal. When angle correction is not possible, spectral Doppler features are evaluated qualitatively.
- •
The normal intratesticular arterial Doppler waveform has a low-resistance pattern.
- •
The normal range of testicular artery peak systolic velocities is 4 to 19 cm/s.
- •
The normal rage of end-diastolic velocities is 1.6 to 6.9 cm/s.

Sonographic technique
A linear-array transducer with a frequency output of 10 MHz or higher is generally used for examining the testicles. A lower frequency setting may be needed if the scrotum is severely swollen. A towel is draped over the penis for the sake of modesty. The penis is then placed over the lower abdomen to keep it out of the examination field. For best results, another towel is placed between the patient’s legs to prop up and support the scrotum.
The first step in scanning the scrotal contents is to get oriented. Long- and short-axis images of each testicle and epididymis are taken. The testicles are measured in their long and short axes. A composite transverse view showing both testicles simultaneously is obtained so that testicular echogenicity can be compared. If both testicles cannot be viewed simultaneously on a transverse view, separate images should be recorded side by side, using identical ultrasound settings. Any pathologic finding should be imaged in whatever plane best documents the abnormality, but additional long- and short-axis views should also be obtained to aid orientation.
Blood flow signals can be evaluated with either color Doppler or power Doppler imaging. In either case, the pulse repetition frequency (color velocity scale) must be set to a low setting to detect very low velocity blood flow signals and the wall filter must also be at a low setting. A relatively high color Doppler gain setting is typically needed, as the testicular vessels are quite small and produce weak Doppler signals. One method to improve the visualization of blood flow signals is to increase the color Doppler gain until artifacts appear in the image and then decrease the gain slightly. It is important to maximize the size of the Doppler waveform display by using an appropriate spectral display scale because this facilitates the assessment of arterial pulsatility patterns, the measurement of blood flow velocities and velocity ratios, and the comparison of testicular blood flow from one side to the other.
- •
The history and physical examination offer important clues for the diagnosis of scrotal pain.
- •
Be sure to prepare the patient for scrotal examination by using towels to prop up and support the scrotum.
- •
Transverse views including both testicles with both gray-scale and color Doppler imaging are critical for detecting abnormalities related to infection and torsion.
- •
Evaluate testicular waveforms for subtle changes that indicate the cause of scrotal pain and swelling. Flow may be present with lesser degrees of torsion.
- •
Optimize color and pulsed Doppler imaging in infants and young children because of low-velocity flow. Look for asymmetry with gray-scale and Doppler imaging.
Scrotal masses
Masses and mass-like lesions within the scrotum include cysts, tumors, hematomas, inflammation, infection, abscesses, contusions, and focal infarction. The location of the pathology, the gray-scale appearance, and the Doppler blood flow features will, in many cases, help narrow down the diagnostic possibilities.
Testicular cysts
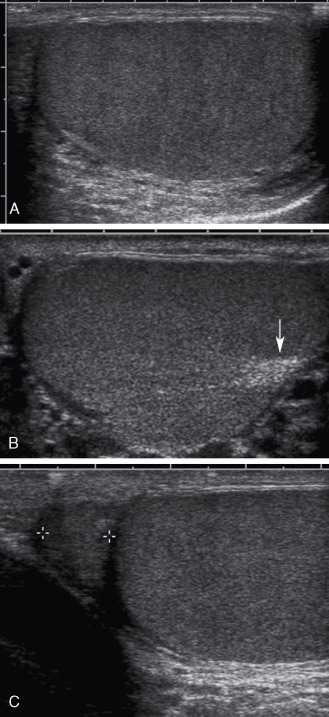
Testicular cysts are idiopathic and benign. They are fairly common, seen with increasing frequency with increasing age and are present in approximately 8% of adults at sonography. Most intratesticular cysts are located near the mediastinum testicle and are not palpable. They are typically small, measuring less than 1 cm in diameter, and are thought to arise from the rete testis, the convergence of intratesticular tubules at the mediastinum. These cysts may be single or multiple. Sometimes, in the presence or absence of discrete cysts, a region of dilated tubules is seen in the vicinity of the mediastinum, representing a dilated rete testis. Cysts located on the testicular surface are almost always tunical cysts, arising in the tunica albuginea, the fibrous layer that encapsulates the testicle. Tunical cysts may be palpable, prompting ultrasound investigation.
The most important point about testicular cysts and a dilated rete testis is distinguishing these benign lesions from other pathologies, including tumors and abscesses. Testicular cysts ( Fig. 30.8 ) have the following sonographic features: (1) anechoic contents, (2) sharply defined borders and an invisible wall, (3) enhanced through-transmission of ultrasound, and (4) no blood flow within or surrounding the cyst (other than normal testicular vessels). Cysts meeting these criteria are benign and inconsequential and require no follow-up. A dilated rete testis appears as small serpiginous tubular structures clustered in the mediastinum ( Fig. 30.9 ).


Testicular neoplasms
Testicular neoplasms are most often primary testicular tumors of germ cell origin ( Table 30.1 ). These neoplasms occur most frequently between the ages of 25 and 35 years and are almost always malignant. The prognosis is generally excellent with an overall 5-year survival of 95%, assuming timely treatment with surgery, radiation therapy, and/or chemotherapy. Less common testicular neoplasms arise from the stromal parenchyma and are either Sertoli or Leydig cell tumors. Rarely, nontesticular malignancies involve the testicle, including leukemia, lymphoma, and metastatic disease. Testicular tumors usually present in one of two ways: as an asymptomatic palpable mass or with sudden onset of pain and swelling caused by hemorrhage. It is not uncommon for the latter presentation to follow minor trauma. Tumors may also present with symptoms of epididymitis. A small number of patients can present with signs and symptoms caused by metastatic testicular cancer, for example back pain from retroperitoneal disease.
| Testicular Malignancies of Germ Cell Origin (95%) |
|
| Other Primary Testicular Malignancies |
|
| Metastases |
|
Ultrasound can distinguish intratesticular from extratesticular pathology with extremely high accuracy. Nevertheless, ultrasound generally cannot differentiate between different histologic types of testicular tumors, nor can it generally differentiate between malignant (common) and benign (uncommon) neoplasms. Most testicular tumors are well-defined hypoechoic intratesticular masses, but some may be poorly marginated or grossly infiltrating. a
a References .
They may exhibit some degree of internal heterogeneity because of hemorrhage and/or necrosis, and calcifications are occasionally present. Vascularity is evident within testicular neoplasms on the color Doppler flow examination, helping to distinguish tumors from cysts, hematomas, and abscesses, which do not have internal vascularity. Testicular tumor vascularity ( Fig. 30.10 ) is variable; however, most malignant tumors are hypervascular compared with the surrounding normal testicular parenchyma. bb References .
The distribution of tumor blood vessels is variable, with some lesions showing an orderly distribution of blood vessels and others, a chaotic distribution. Large avascular areas may be present in a testicular tumor when necrosis or hemorrhage is present. Spectral Doppler generally shows low-resistance blood flow within tumor vessels, a typical finding seen in malignant neoplasms regardless of location. Blood flow velocities may be substantially elevated in markedly hypervascular tumors. In general, the larger the testicular tumor, the more hypervascular it will be.
Testicular microlithiasis, scattered small calcifications in the testicular parenchyma as shown in Fig. 30.10A , is associated with an eightfold increased risk of testicular cancer, but the level of risk and follow-up requirements remain controversial. Recent publications recommend urologic referral in patients with concomitant risk factors for testicular cancer. In patients without other risk factors for cancer, monthly self-examination for masses is suggested.
Testicular tumor mimics
Lesions that can mimic the appearance of neoplasms include abscesses, inflamed areas (without frank abscess formation), contusions, hematomas, and infarcts. The sonographic appearance of these lesions may be nonspecific, as discussed later. Color and spectral Doppler features are of considerable importance in differentiating these etiologies. No flow is present in abscesses, infarcts, and hematomas, but hypervascularity may be present in the periphery of these lesions.
- •
Testicular neoplasms tend to be vascular, whereas cysts, abscesses, and hematomas are not.
- •
The larger a testicular tumor, the more likely it is to be hypervascular.
- •
Testicular lesions often need follow-up imaging to assess for resolution and to exclude malignancy.
Epididymal cysts
Epididymal cysts are much more common than testicular cysts, found in about 40% of adult males having an ultrasound examination. Most are located in the epididymal head, but cysts may occur anywhere in the epididymis: head, body, or tail. They may be single, multiple, unilateral, or bilateral. In some cases they are palpable, whereas in others they are found incidentally. Unlike testicular cysts, epididymal cysts may be septated or even multilocular. Most are 2 to 3 mm in diameter, but larger cysts are also common, and occasionally they may be several centimeters in size. The etiology of epididymal cysts is not entirely clear, although some are spermatoceles, which are encapsulated collections of sperm.
The great majority of epididymal cysts have the same sonographic features as testicular cysts, but there are other variations. Some may be septated or multilocular, and others may contain diffuse or dependent low-level echoes. When evaluating epididymal cysts sonographically, it is most important to document that the epididymal cysts have no solid components, have exquisitely thin walls, and show no internal blood flow on color Doppler examination ( Fig. 30.11 ).



Other epididymal masses
Other lesions of the epididymis include hematomas, abscesses, or inflammatory masses. Tumors of the epididymis are uncommon and usually benign. An important characteristic of any mass found is the presence or absence of blood flow, which should be detectable in a tumor but not present in a hematoma or abscess. Hematomas of the epididymis or spermatic cord are usually associated with trauma but may occur spontaneously or be associated with vigorous exercise. Abscesses and inflammatory masses are part of the differential diagnosis and are discussed later.
Epididymitis and orchitis
Infection is the most common cause of acute scrotal pain and tenderness. The great majority of cases in adults are because of an infection caused by sexually transmitted organisms (principally Neisseria gonorrhoeae and Chlamydia trachomatis ) that ascend through the genital tract. a
a References .
The tail of the epididymis is infected first, and then the infection spreads throughout the epididymis (epididymitis). The infection may then extend to the testicle (epididymo-orchitis) and finally to the scrotal cavity, generating an infected hydrocele.Ultrasound is a very useful method for confirming the diagnosis of epididymitis or orchitis and for excluding other pathologies that may cause acute scrotal pain or swelling. The key findings are enlargement and decreased echogenicity of the affected structures, accompanied by increased blood flow (hyperemia) on color or power Doppler examination. b
b References .
Because orchitis may be present by the time the patient seeks care, involvement of both the testicle and epididymis may be evident on ultrasound; however, in many patients, epididymal involvement will predominate ( Fig. 30.12 ). A hydrocele (excess scrotal fluid) is often present, and the scrotal wall may become edematous and/or inflamed. In some cases of epididymitis or orchitis, the ultrasound findings may be dramatic. In other instances, findings are less obvious and are based solely on side-to-side comparison of epididymal size, echogenicity, and blood flow patterns. Obviously, side-by-side comparison is not helpful in cases of symmetrical, bilateral infection; nevertheless, hyperemia of the scrotal contents may still be evident in such cases. It is noteworthy that focal orchitis may be seen as a focal, hypoechoic area of inflammation at the periphery of the testicle, adjacent to an infected epididymal head. This should not be mistaken for a testicular tumor but should be followed to resolution after antibiotic treatment.

The sonographic characteristics of epididymo-orchitis include hypoechogenicity of the involved portion of the testicle with hypervascularity, in addition to enlargement and hyperemia of the epididymis ( Fig. 30.13 ). With unilateral orchitis, the involved testicle is typically hypervascular with inhomogeneous areas of decreased echogenicity ( Fig. 30.14 ). These latter findings are similar to findings following detorsion of a testicle or with diffusely infiltrating lymphoma or leukemia.




The epididymis and testicle usually return to a normal sonographic appearance following an episode of infection, but in severe cases, testicular atrophy or infarction may occur. Atrophy is a noteworthy complication of mumps orchitis. Untreated or incompletely treated cases of epididymitis may present with symptoms and findings of chronic epididymitis, which may manifest as diffuse thickening and heterogeneity of the epididymis or as a focal, heterogeneous epididymal mass. Increased blood flow may not be a feature of chronic epididymitis. Hydrocele may also be present, and this may be septated or contain echogenic material.
Abscesses may occur in the epididymis or testicle in severe cases of scrotal infection. These appear as complex fluid collections with irregular walls, sometimes containing diffuse or dependent debris. Increased blood flow caused by hyperemia may be apparent in the surrounding tissues, either focally or diffusely. If an epididymal abscess becomes chronic, hyperemia may no longer be evident, making the abscess indistinguishable from other extratesticular lesions.
- •
Increased blood flow signals on color Doppler imaging and increased blood flow velocities are seen in cases of epididymitis, orchitis, or epididymo-orchitis.
- •
Localized orchitis can mimic the appearance of a tumor, and these lesions should be followed during and after antibiotic treatment.
Varicocele
Varicocele , or dilation of the pampiniform venous plexus, is a common cause of a palpable scrotal mass and pelvic discomfort. a
a References .
In some individuals, a varicocele contributes to low sperm count, decreased sperm motility, and infertility. These problems have been attributed to persistent, hyperemia-induced elevation of testicular temperature, but the real cause may be more complex and is not known with certainty.The veins of the pampiniform plexus, which drain the testicle and epididymis, are normally quite small, but these may dilate and become tortuous because of valvular incompetence and/or elevated venous pressure, forming a tangle of enlarged veins along the course of the spermatic cord and epididymis. In unusual cases, dilated veins may even extend into the substance of the testicle. Varicoceles are more common on the left side of the scrotum than the right, possibly because of elevated pressure in the left spermatic vein. The left spermatic (testicular) vein inserts into the left renal vein, which drapes across the aorta and may be compressed between the aorta and superior mesenteric artery, raising venous pressure, or it may be subject to higher hydrostatic pressure than the right because of its longer course. The right spermatic (testicular) vein drains directly into the inferior vena cava, is not subject to compressive effects, and is shorter than the left spermatic vein. Because of the left-side predominance of varicocele, spermatic vein obstruction as a result of an intra-abdominal mass should be considered whenever an isolated right scrotal varicocele is diagnosed.
Varicoceles are usually a clinical diagnosis, as the tangle of veins is easily palpated and feels like a bunch of worms. Ultrasound is required when the nature of the palpable mass is unclear or when pain or tenderness is present, as well as in men who experience infertility. A varicocele is diagnosed with color Doppler ultrasound when numerous veins of unusually large size are seen along the spermatic cord or epididymis ( Fig. 30.15 ). The extent of the varicosities should be documented, and the largest veins should be measured. The presence of reflux within the veins can be investigated by having the patient perform the Valsalva maneuver during color Doppler imaging. Normal pampiniform veins are barely detectable, so from one perspective, varicocele can be diagnosed whenever veins of unusual size and number are readily seen. More specific diagnosis may be important, however, in men with pain or infertility, in whom a decision must be made concerning the potential benefit of therapy. In this respect, veins 2 mm in diameter or less are generally regarded as not substantially dilated, whereas larger veins are considered varicose, especially those with a diameter of 3 mm or larger. This definition includes veins reaching this size with the patient in positions not typically used during ultrasound examination (e.g., standing, squatting) and in any state of respiration, including straining or performing a Valsalva maneuver. The demonstration of retrograde flow or reflux in the veins during a Valsalva maneuver is further evidence of potential clinical significance of a varicocele.
Varicoceles most often occur on the left side. Assess for spermatic vein obstruction by a pelvic mass when a varicocele is isolated on the right side.
- •
Veins of the pampiniform plexus are normally not seen on ultrasound examination.
- •
Varicoceles are considered present when a vein diameter reaches 3 mm or more.
- •
Retrograde flow or reflux seen in varicoceles during a Valsalva maneuver indicates greater clinical significance.

Testicular torsion
Torsion refers to twisting of the testicle within the scrotal sac, such that the arteries and veins are compressed and blood flow is compromised. a
a References .
Torsion usually occurs in children or young adults, and two peaks of incidence have been noted: the neonatal period and puberty. Torsion in neonates is typically extravaginal. That is, the entire contents of the scrotum twist on the spermatic cord, including the parietal and visceral layers of the tunica vaginalis. The torsion may occur prenatally and is seen in neonates of high birth weights. Torsion during puberty and in young adults is virtually always intravaginal, where the parietal tunica vaginalis remains intact, while the scrotal contents inside the parietal tunica twist.Intravaginal torsion results from abnormal mobility of the testicle because of narrow attachment of the testicle to the scrotal wall by the reflection of the tunica vaginalis (see Fig. 30.2B ). Normally, the parietal layer of the tunica vaginalis lines the inner wall of the scrotal sac until it reaches the edge of the testicle, where it reflects over the testicle, forming the visceral layer of the tunica vaginalis. The portion of the testicle that is not covered by the visceral layer of the tunica vaginalis is closely held against the scrotal wall and called the bare area. Vessels and tubules from the spermatic cord enter and leave the testicle across the bare area. When the bare area is abnormally small (an abnormality called the bell clapper deformity), the attachment of the testicle to the scrotal wall is narrow and the testicle is at risk of torsion because of twisting at this attachment. The pathologic sequence of events begins when the spermatic cord twists, with a rotation of at least 360 degrees, at the bare area. This causes venous obstruction, leading to swelling and increased pressure inside the testicle and within the spermatic cord. Subsequently, arterial flow becomes occluded and testicular ischemia results, progressing to infarction if detorsion does not occur surgically or spontaneously.
The pathologic process of torsion can be divided into acute torsion , during which the testicle suffers ischemia without permanent injury if detorsion occurs, and missed torsion , the stage after which testicular infarction has occurred to the point that the testicle cannot be saved even if detorsed. The testicle is almost always salvageable during the first 6 to 10 hours of torsion and is progressively less likely to be salvageable thereafter. In virtually all cases of torsion lasting more than 24 hours, the testicle cannot be saved.
Color Doppler ultrasound is the imaging method of choice for diagnosing testicular torsion. Gray-scale imaging and Doppler evaluation have high sensitivity (>85%) and virtually 100% specificity for diagnosing testicular torsion. In addition, ultrasound with Doppler is typically readily available in the emergency setting and can be performed quickly.
Soon after torsion occurs, the testicle may appear normal in echotexture. During this time, the only abnormal gray-scale sonographic findings may be in the spermatic cord and epididymis, which may appear as a thick echogenic structure with acoustic shadowing caused by twisting and the knot that forms. On Doppler imaging, blood flow will appear diminished or absent in the testicle and the knotted cord and epididymis. As the torsion persists and blood flow remains occluded, the testicle becomes enlarged and hypochoic due to swelling and edema. At this time, a small hydrocele is sometimes seen. With color Doppler imaging, blood flow remains diminished or absent in the testicle and epididymis. If there is further progression to the development of testicular necrosis, the testicle becomes mottled and heterogeneous with hypoechoic areas, and the scrotal wall becomes thickened and hypervascular. Once the testicle becomes heterogeneous, the likelihood of successful salvage is extremely low.
Gray-scale and Doppler findings are key to the diagnosis of testicular torsion. The examination should begin with gray-scale, including side-to-side comparison of testicular size and echogenicity and assessment of the epididymis to look for the knotted cord ( Fig. 30.16 ). Color Doppler should then be performed for side-to-side assessment of blood flow. With testicular torsion, differences in perfusion, including absence or marked diminution of blood flow will be found on the affected side. If blood flow is still present on the affected side, spectral waveforms typically demonstrate high-resistance flow, compared with the low-resistance flow in the normal testicle.