Eosinophilic diseases of the lung include a heterogeneous group of pulmonary disorders that characteristically feature peripheral or tissue eosinophilia. Patients with these illnesses have a variable clinical presentation and may be asymptomatic or may exhibit organ dysfunction or clinical symptoms of severe respiratory illness. Histologically, eosinophilic lung diseases demonstrate infiltration of eosinophils in the pulmonary interstitium and alveolar spaces with preservation of the lung architecture. As a result, these illnesses respond dramatically to corticosteroid treatment and typically heal without significant lung parenchymal damage.
The diseases are classified as idiopathic or secondary to a known primary disease process or well-defined cause. Established causes of eosinophilic lung disease include primary airways diseases, such as allergic bronchopulmonary aspergillosis (see Chapter 57 ), drug reaction (see Chapter 65 ), and toxin exposure and parasitic infections (see Chapter 14 ). The idiopathic eosinophilic diseases of the lung are simple pulmonary eosinophilia (Löffler syndrome), acute and chronic eosinophilic pneumonia, and the systemic conditions such as hypereosinophilic syndrome and eosinophilic granulomatosis with polyangiitis (formerly known as Churg-Strauss, which is discussed in Chapter 46 ).
The diagnosis of eosinophilic lung disease can be made in the presence of one of the following criteria: (1) peripheral eosinophilia (>1000/mm 3 ) in conjunction with pulmonary parenchymal opacities on chest radiography, (2) tissue eosinophilia on surgical or transbronchial lung biopsy, or (3) eosinophilia in bronchoalveolar lavage (BAL) fluid (differential cell count > 25).
Simple Pulmonary Eosinophilia
Etiology, Prevalence, and Epidemiology
Simple pulmonary eosinophilia (SPE) was originally identified and described in 1932 by Löffler. Patients with SPE typically have fleeting pulmonary opacities on chest radiography in the presence of blood eosinophilia. Approximately one-third of cases of SPE are idiopathic. The majority of cases occur secondary to drug toxicity or parasitic infection. The incidence of SPE in oncologic patients undergoing follow-up CT imaging has been reported to be 0.95%. These individuals typically lack respiratory symptoms and have subsolid nodules on chest CT.
Clinical Presentation
Individuals may be asymptomatic or may experience minimal respiratory symptoms, fever, or malaise. Symptoms and radiologic findings are self-limited and resolve spontaneously within 30 days with or without treatment.
Pathophysiology
Data is limited on the pathophysiology of this disease entity, but there is an association with alveolar infiltration with eosinophils and giant cells.
Manifestations of the Disease
Radiography
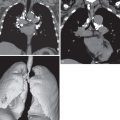
The characteristic radiographic findings are nonsegmental opacities, which are frequently ill-defined and peripheral in distribution. The opacities may be unilateral or bilateral and are characteristically transient or migratory ( Fig. 37.1 ).

Computed Tomography
Ground-glass opacities and consolidation, the most common CT findings, either showed a random distribution or upper and middle lung zone predominance ( Fig. 37.2 ; see Fig. 37.1 ). Pulmonary parenchymal nodules were reported to be present in 42% of cases. The CT features and distribution of opacities present in SPE and chronic eosinophilic pneumonia are similar; the important distinguishing feature is that the imaging findings in SPE will spontaneously fluctuate over a period of days as opposed to chronic eosinophilic pneumonia (CEP), where the consolidations and ground-glass opacities will persist for weeks to months and be accompanied by more significant respiratory symptoms. In individuals with SPE found at oncologic surveillance or routine screening, pulmonary nodules (either solitary or multiple) with a ground-glass halo are the most common CT finding.

Differential Diagnosis
The presence of consolidation on chest radiography can be seen in the setting of pneumonia, alveolar pulmonary edema, or diffuse alveolar damage. The absence of or minimal pulmonary symptoms associated with SPE, along with peripheral blood eosinophilia, strongly suggests the diagnosis. It is important to exclude known associations with pulmonary eosinophilia, the most common of which are parasitic infection and drug toxicity.
Synopsis of Treatment Options
No treatment is required because SPE is asymptomatic and self-limited.
- •
Approximately one-third of cases are idiopathic; infectious etiologies and drug toxicity are more common and should be excluded.
- •
Clinical course is mild and self-limited, with symptoms resolving spontaneously within a month.
- •
Radiography shows nonsegmental, peripheral opacities that are transient and/or migratory.
- •
Subsolid nodules, ground-glass opacities, and consolidation may be seen on CT.
Chronic Eosinophilic Pneumonia
Etiology, Prevalence, and Epidemiology
Idiopathic chronic eosinophilic pneumonia (ICEP) is a respiratory illness characterized by alveolar eosinophilia, typically greater than 25% at BAL and/or blood eosinophilia of at least 6%. Although blood eosinophilia is present in most patients, up to 12% of patients with ICEP do not have peripheral eosinophilia. The disease is more common among women and tends to occur around the fifth decade. There is an association of ICEP with asthma, which has been reported to occur in up to 53% of patients with ICEP. Unlike in eosinophilic granulomatosis with polyangiitis and hypereosinophilic syndrome (HES), extrathoracic symptoms and organ dysfunction are not present in ICEP.
Clinical Presentation
Systemic symptoms associated with ICEP, such as fever, weight loss, and night sweats in addition to dyspnea and cough, are typically present for 1 month on average before clinical presentation. The mean time to diagnosis is 7.7 months, reflecting the confounding factors that make the diagnosis of ICEP challenging. Other features seen in a minority of patients include anorexia, chills, and hemoptysis.
Pathophysiology
At histology there is an alveolar infiltration of eosinophils and to a lesser extent macrophages, lymphocytes, and plasma cells, and in approximately half of patients, there will be foci of interstitial fibrosis. In more rare cases, eosinophilic microabscesses and regions of intraalveolar necrosis will also be present.
Manifestations of the Disease
Radiography
The characteristic radiographic finding in ICEP is peripheral airspace opacities. In their 1969 article that first described the disease, Carrington and colleagues identified the unique presence of three features in ICEP: progressive peripheral dense consolidations, prompt resolution with corticosteroid treatment, and recurrence in the same unique locations upon relapse. The dense peripheral consolidation in ICEP has been described as the “photographic negative” of pulmonary edema. There is typically no lobar or segmental distribution to the opacities, which can be unilateral or bilateral and show ill-defined margins. An apical or axillary location is common ( Fig. 37.3 ), and opacities may be mistaken for loculated pleural fluid.

Computed Tomography
Subpleural and upper lung zone–predominant consolidation and areas of ground-glass opacity are present in the vast majority of patients ( Figs. 37.4 and 37.5 ). In some patients mediastinal lymphadenopathy will be evident on CT ( Fig. 37.6 ). Unlike in acute eosinophilic pneumonia, pleural effusions are rare. Less common CT findings include nodules, bronchiectasis, and interlobular septal thickening. In patients who have had at least 2 months of symptoms at the time of CT imaging, band-like opacities and lobar atelectasis can be seen.



Differential Diagnosis
The primary differential consideration in patients who have ICEP is an infectious pneumonia, given the airspace opacities and febrile respiratory illness that characterize the disease. Other diagnoses that may be considered include organizing pneumonia, drug toxicity, and sarcoidosis. The upper lobe–predominant opacities, constitutional symptoms, and rare hemoptysis may raise concern for tuberculosis. SPE may appear identical to ICEP on imaging; however, patients with SPE will have transient opacities that fluctuate over a period of days, a relative paucity of constitutional symptoms, and spontaneous resolution. The presence of blood and alveolar eosinophilia, dramatic clinical and radiographic response to steroids, and peripheral airspace opacities favor the diagnosis of ICEP.
Synopsis of Treatment Options
ICEP is extremely steroid sensitive, and opacities seen on imaging improve or resolve rapidly on initiation of corticosteroid therapy. After discontinuation or tapering of steroid therapy, there is a tendency for symptoms to recur and opacities to reappear, often in the same location and configuration as seen initially.
- •
Idiopathic chronic eosinophilic pneumonia features filling of the alveoli with a mixed inflammatory infiltrate, primarily of eosinophils.
- •
Peripheral eosinophilia is typically present, but absence of elevated blood eosinophils does not exclude the diagnosis.
- •
Systemic symptoms include fever, nonproductive cough, weight loss, and dyspnea. In contrast to acute eosinophilic pneumonia, hypoxemic respiratory failure is uncommon.
- •
As many as 50% of patients have a history of asthma or atopy.
- •
The characteristic pattern is peripheral opacities favoring the upper lobes: “photographic negative” of pulmonary edema.
- •
Response to corticosteroids is rapid, although recurrence is frequent with steroid withdrawal.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree