FUNCTIONAL ENDOSCOPIC SINUS SURGERY
KEY POINTS
- Computed tomographic imaging is central to the diagnosis of disease that might require functional endoscopic sinus surgery.
- Computed tomographic imaging is central to the planning of functional endoscopic sinus surgery.
- Computed tomographic imaging is central to the evaluation of failed functional endoscopic sinus surgery or complications that might arise from such procedures.
- Magnetic resonance imaging plays an adjunctive role in these settings.
- Imaging must be properly integrated with the clinical evaluation to make the best strategic use of the costly examinations.
- Computed tomography must be used judiciously in children, especially under the age of 6 years, where the use of imaging is unlikely to alter medical decision making except under highly unusual circumstances.
The work of Messerklinger and Wigand in the late 1970s, together with the birth of computed tomography (CT) in the same decade, led to radical changes in the treatment of chronic sinusitis.1–5 These changes, based on a more proper understanding of sinonasal anatomy and physiology than available previously, have led to further applications of the then new concept of functional endoscopic sinus surgery (FESS). The initial combination of imaging and those new ideas about how to preserve the normal nasal physiology while extirpating disease has led to many more applications of FESS and when combined with real-time image guidance allows a more thorough and precise removal of disease as well as improved safety. FESS has led to dramatically improved success rates and related quality-of-life improvement in treating patients with diseases of the sinuses and/or nasal cavity. The fundamental assumption in FESS is that the surgical approach, to the extent possible, should preserve and enhance the normal patterns of mucociliary drainage, nasal airflow, and as much mucosa as possible. These techniques are now being extended to the central skull base by some very experienced practitioners6; such applications are not the focus of this chapter.
The role of imaging is defined by the clinical context. It should only follow a thorough history and a complete physical examination, including at least anterior rhinoscopy and often nasal endoscopy. The planned surgery should be reasonably linked to the patient’s symptoms, and imaging should corroborate those circumstances. In the great majority of cases, there should be a trial of conservative management, and imaging should be done only after the patient has reached maximum, if any, medical improvement with such a regime. Some conditions require more immediate imaging and perhaps surgical intervention.
Imaging then helps with the confirmation of disease and surgical planning. With regard to the latter, those interpreting these CT studies must understand and document circumstances that might raise the risk of a complication and/or present obstacles to a successful outcome.
Indications
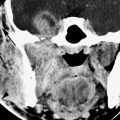
FESS is most commonly performed in patients with documented chronic rhinosinusitis, including CT scans if appropriate, and in those who have failed appropriate medical treatment (Fig. 83.1). For instance, those with nasal polyposis may have surgery sooner rather than later since medical therapy alone is less likely to be successful. It is clearly useful in patients with mucoceles, for epistaxis control, and for foreign body removal.
Endoscopic sinus surgery (ESS) for nasal masses and tumors must be selected based on the tissue type and the extent of disease as shown on imaging and nasal endoscopy. For instance, the endoscopic removal of an inverted papilloma has been controversial since careful control of bony margins and may require an external approach (Fig. 83.2). However, with more complex procedures being performed endonasally, greater preoperative imaging, and the use of image guidance, endoscopic removal of this disease is becoming a more standard approach.
Cerebrospinal fluid (CSF) leaks associated with CSF rhinorrhea can be managed with a high success rate with ESS, thus avoiding a neurosurgical intervention in over 80% to 90% of patients (Chapter 80).
Endoscopic approaches are currently being extended to include traditionally external approaches for ophthalmologic indications such as orbital decompression and dacryocystorhinostomy and even central skull base surgery in very experienced hands.
In children, especially those younger than 6 years of age, indications are more limited because of the very high normal incidence of upper respiratory infections in this age group. Some of these include management of extensive polyposis associated with chronic sinus disease in cystic fibrosis (Chapter 85), mucoceles, sinus infections complicated by orbital and intracranial extension of the infection (Chapter 84), fungal sinusitis (Chapter 86), and meningoencephalocele (Chapter 80) and choanal atresia repair (Chapter 81).
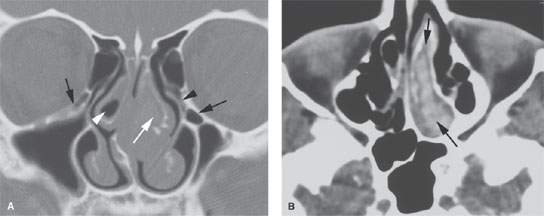
FIGURE 83.1. Computed tomography study of a patient being considered for functional endoscopic sinus surgery (FESS). A: There is a small concha bullosa on the right (white arrowhead) that contains mucosal thickening. The nasal septum is deviated to the right. Also on the right are Haller air cells that are a site of isolated disease. Compared to the Haller cell on the left side that is not diseased (black arrow), the concha bullosa on the left is diseased and expanded (white arrow). The left infundibulum (black arrowhead) is narrowed. B: Axial section at soft tissue shows essentially a mucocele within the left concha bullosa (black arrows) containing dried secretions. (NOTE: The findings on this study clearly helped to explain the patient’s sinonasal obstructive symptoms and provided an excellent road map for the FESS.)
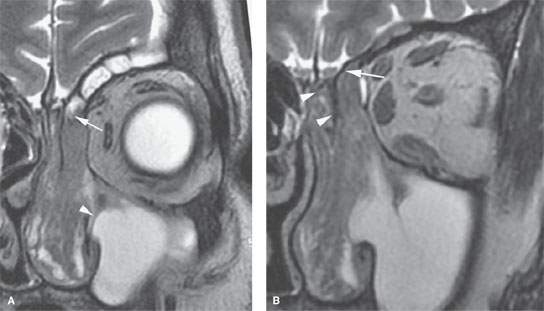
FIGURE 83.2. Magnetic resonance imaging study of a patient with an inverted papilloma showing that the papilloma involves the anterior skull base and likely is beyond the limits of appropriate gross total resection via functional endoscopic sinus surgery. A: T2-weighted (T2W) images showing the tumor high in the frontal recess (arrow) and its interface with postobstructive changes in the frontal sinus as well as the maxillary sinus (arrowhead) very well defined. B: T2W image showing the tumor clearly involving the ethmoid roof (arrow) and spreading between the ethmoid medial wall and vertical plate to the cribriform plate (arrowheads).
Contraindications and Possible Complications
External approaches may be preferable over ESS when complete, wider field treatment of disease is required, such as when sinus disease causes an orbital abscess or osteomyelitis or when an inverted papilloma extends to the anterior skull base (Fig. 83.2). Imaging with CT scans or magnetic resonance imaging (MRI) will help to guide decision making in these cases. Open procedures may also be preferred in failed CSF leak repair or when the frontal sinus must be obliterated or cranialized.
The most frequent major complications are those of orbital injury that might cause an orbital hematoma possibly leading to blindness or diplopia and nasolacrimal duct injury that could produce epiphora (Fig. 83.3). Penetration of the cribriform plate and/or ethmoid roof might cause a CSF leak or direct brain injury. Bleeding can be due to a lacerated anterior ethmoid artery (Fig. 83.3).
Scar formation can obstruct sinuses and/or cause the turbinates to become distorted and perhaps causing secondary obstruction or just poor airflow and recurrent symptoms. Forewarning of anatomic variants can help to avoid some of these unfortunate outcomes. Incomplete removal of disease or persistent disease may occur when anatomic variants, such as frontal cells, are not taken into account (Fig. 83.4).
IMAGING APPROACH
The general CT and magnetic resonance (MR) techniques used in this clinical setting are presented in Appendixes A and B. MRI is really only used for problem solving in highly selected circumstances, such as when FESS might be preferred if possible for resection of an inverted papilloma or to confirm the presence of an encephalocele. MRI is particularly valuable for surgical planning when used to better visualize the soft tissue interface between sinonasal structures and intraorbital or intracranial contents when the normal bony interfaces are absent or significantly altered on CT.
CT data sets should be obtained with 0.50- to 0.75-mm sections suitable for the highest-resolution multiplanar reconstructions and/or reformations. Contrast is only occasionally used with CT when tumor or a complicated acute infection is suspected. The most common applications of CT for uncomplicated sinus and facial study that might require FESS are done without intravenous contrast. Indication-specific CT protocols are presented in Appendix A.
APPLIED ANATOMY AND PATHOPHYSIOLOGY AS IT RELATES TO REPORTING
Reporting Rationale
The structures of the lateral nasal wall and paranasal sinuses are discussed in detail in Chapter 78.
FESS is a generally systematic approach to the anterior and posterior aspects of the sinonasal fields and related parts of the nasal cavity, orbit, and anterior and central skull base. The logic and approach of each operation should guide the thinking of those interpreting both diagnostic and preoperative planning studies. There are anatomic and physiologic principles related to sinonasal function that must be kept in mind when generating reports that are meant to diagnose conditions that might benefit from FESS. The fundamental assumption in FESS is that the surgical approach, to the extent possible, should preserve and enhance the normal patterns of mucociliary drainage, nasal airflow, and as much mucosa as possible. This must be done while accomplishing the goals of surgery, be it simply restoration of normal drainage pathways, correction or repair of some congenital or acquired abnormality, or extirpation of tumor or complicated acute inflammatory disease.
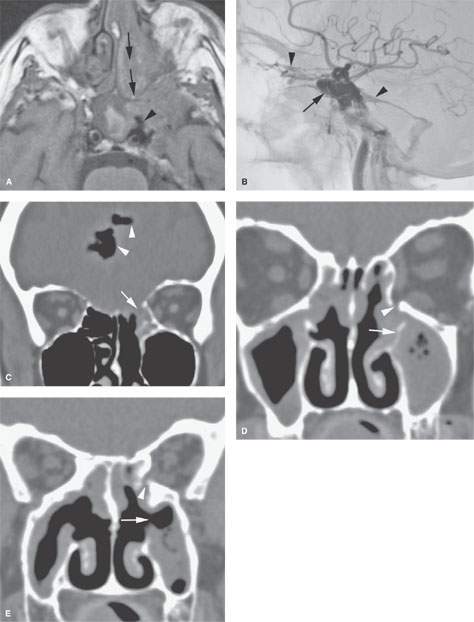
FIGURE 83.3. Illustrations of three patients experiencing complications of functional endoscopic sinus surgery (FESS). A, B: Patient 1. In (A), the non–contrast-enhanced T1-weighted magnetic resonance study shows the tract of surgery through the nose and into the sphenoid sinus (arrows) where the carotid artery was entered and a cavernous carotid fistula (CCF) created (arrowhead). In (B), catheter angiography shows the CCF draining to orbital and intracranial venous structures (arrowheads). C: Patient 2. Computed tomography (CT) study shows the patient to have relatively normal anatomy of the ethmoid roof and cribriform plate region, although the olfactory fossa is somewhat shallow and the overall cephalocaudal dimensions are slightly less than usual. During FESS, there was a substantial breach of the ethmoid roof (arrow), resulting in the intracranial gas (arrowheads). D, E: Patient 3. CT study done following FESS. The patient had recurrent sinus disease and double vision. In (D), there was an incomplete resection of the uncinate process (arrow), and the orbit was entered with the microdébrider during surgery (arrowhead). In (E), a section somewhat more posterior shows that the patient had a fairly wide medial antrostomy but did not have a complete uncinectomy (refer to A). Also, the orbital floor defect persists and the intraocular muscles are adherent to the area of the defect, explaining the patient’s double vision.
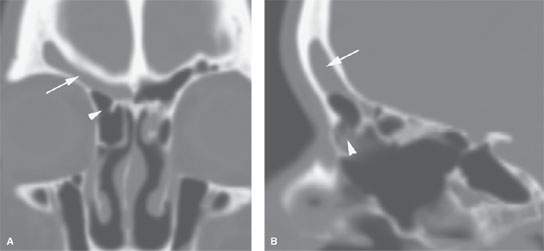
FIGURE 83.4. Computed tomography study of a patient with recurrent right frontal region symptoms following functional endoscopic sinus surgery. A: Coronal reformations showing recurrent mucosal thickening in the right frontal sinus (arrow) with an obstructing residual air cell (arrowhead). B: Sagittal reformation showing the persistent disease (arrow) and the likely suprabullar air cell obstructing the frontal recess (arrowhead).
Reports anticipating or analyzing the effects of FESS must reflect the related anatomic features that may be used to properly plan surgery and anticipate any potential hazards. The rigor of the report may be less intense in other clinical settings. This chapter, however, assumes that the study will at some point be used to plan FESS. Such reports are then inexorably linked to the following anatomic substrate, and the structure of the report should reflect that linkage.
Anterior and Posterior Ethmoid Complexes
There is an anterior and posterior ethmoid complex separated by the basal lamella of the middle turbinate. This separation of the two ethmoid complexes has its basis in the patterns of mucociliary secretion transport as well as embryologic development. Air cells and clefts that lie anterior and inferior to this lamella are of the anterior ethmoid complex, and those that drain posterior or superior, other than the sphenoid sinus, are of the posterior ethmoid complex (Fig. 78.1).
The basal lamella of the middle turbinate lies in three different planes. An anterior vertical segment lies in the sagittal plane along the lateral edge of the cribriform plate. The middle horizontal segment is fixed to the medial orbital wall, and a more posterior segment is attached to the medial orbital wall and the medial wall of the maxillary sinus or both. This imparts stability to the basal lamella of the middle turbinate. Variability in aerations of its middle segment will alter the boundary between the anterior and posterior ethmoidal complexes.
Reporting Significance
Reporting should be done with sensitivity to the boundaries of anterior and posterior procedures, and disruption of the stability of the middle turbinate attachments may impact the surgical outcome (Fig. 83.5).
Structures of the Anterior Ethmoid Complex, Including a Number of Structures of Very Variable Appearance
Ostiomeatal Complex or Unit
While ostiomeatal complex is a common term, it is not a specific anatomic structure. The term describes one functional entity of the anterior ethmoid complex that is a final common pathway for drainage and ventilation of the frontal, maxillary, and anterior ethmoid cells. Its usage in reports is, therefore, of little or no value. It is the specific anatomic structures that follow that make up the core observations that contribute to building a meaningful report.
Uncinate Process
This structure presents tremendous variations in its size and attachments that may influence drainage patterns and the likelihood of disease in the ethmoid infundibulum.
Reporting Significance
Unusual attachments of the uncinate process or less common variations such as presence of an uncinate air cell that might obstruct the infundibulum may be central to chronic recurrent disease and should be noted (Fig. 83.6). Absence of the uncinate should also be noted, as this is most commonly seen in postsurgical patients; however, erosive processes can also lead to destruction of the uncinate.
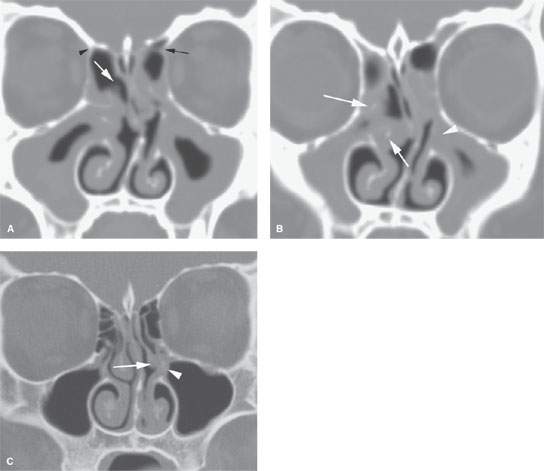
FIGURE 83.5. Computed tomography (CT) studies of two patients with recurrent disease following functional endoscopic sinus surgery (FESS). A, B: Patient 1. In (A), the CT study of a patient with recurrent disease following endoscopic sinus surgery is shown. The vertical attachment of the middle turbinate was removed during the surgery (white arrow). Also note the anatomic variations in the anterior ethmoid artery. In this case, the vessel appears to be on a mesentery on the left (black arrow) and not on a mesentery on the right (black arrowhead). In (B), a section somewhat more anterior to (A) shows that the destabilization of the middle turbinate caused it to become lateralized (arrows). The left side shows the region of the uncinectomy to be covered over by the recurrent disease (white arrowhead). (NOTE: This study reflects the importance of careful planning of FESS and preservation of as much normal anatomy as possible. In this patient, the vertical attachment of the right middle turbinate as well as part of its horizontal attachment were taken down, probably creating a substrate for recurrent disease. Also, the uncinectomy on the left side may have been incomplete, and there was not a medial antrostomy done. The nasal septum was left fairly markedly deviated to the left.) C: Patient 2. This patient had minimal endoscopic sinus surgery with an intended uncinectomy. The patient presented with recurrent left-sided symptoms. This study shows the uncinectomy to be incomplete (arrowhead) and the medial turbinate to be scarred into the attempted infundibulotomy site (arrow).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree