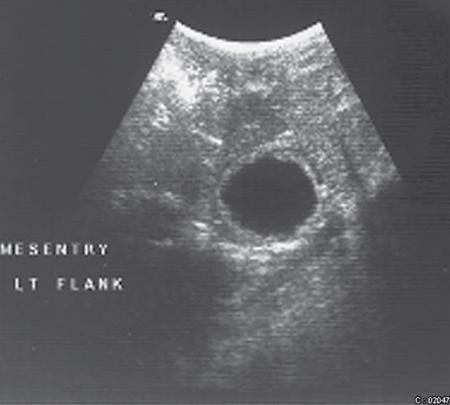
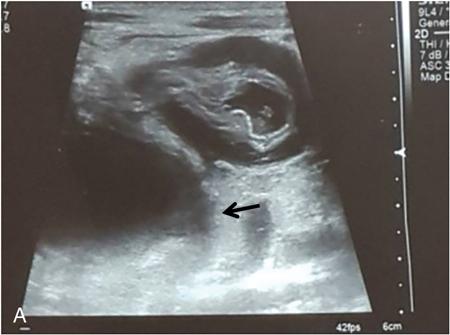
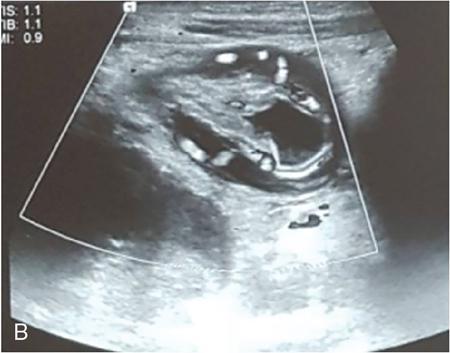
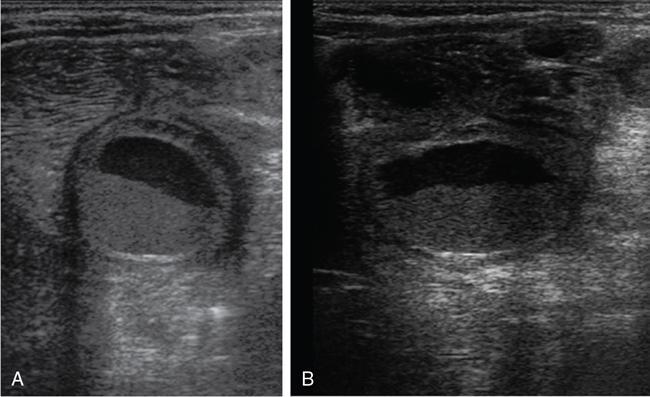
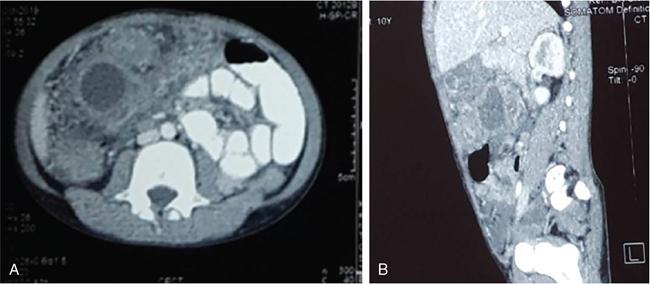
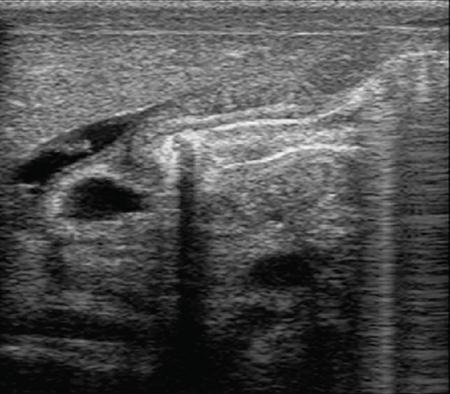
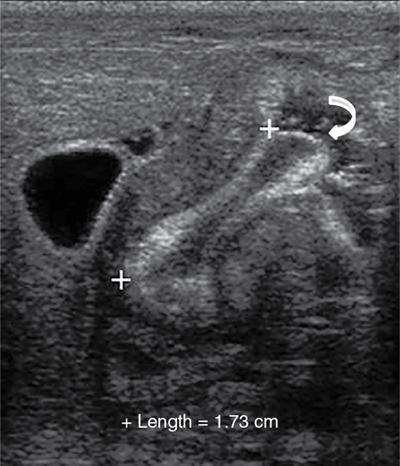
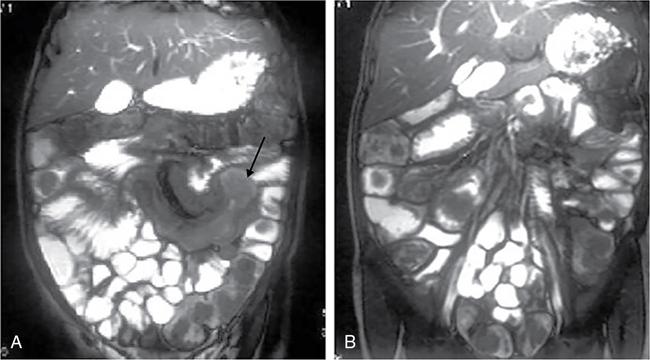
Rashmi Dixit, Anju Garg Abdominal masses are a common clinical problem in children, arising from virtually any abdominal organ. Masses arising from the gastrointestinal (GI) tract are not uncommon. They may be congenital or developmental, inflammatory, infective, idiopathic or neoplastic in nature. A high index of suspicion is necessary so as to ensure an early diagnosis and appropriate management. Imaging, therefore, plays a vital role in identifying the location, nature and extent of the mass lesion. Ultrasound (US) is the first-line imaging method for GI masses, like for most paediatric abdominal masses. It does not require sedation and is nonionizing which are important considerations in the paediatric population. US findings may be diagnostic in certain masses when no further imaging is required. Computed tomography (CT) plays an important role in complete evaluation of GI masses especially where US is either not diagnostic or the full extent and nature of the mass needs to be delineated, especially prior to surgery. It requires the administration of intravenous contrast and preferably oral contrast as well. Sedation may be required and it carries the risk of radiation exposure. Each study, hence, needs to be tailored to answer the clinical question at hand, while ensuring the lowest possible radiation exposure. Magnetic resonance imaging (MRI) provides excellent contrast resolution, which along with its nonionizing nature makes it a very attractive imaging technique. However, due to the relatively long imaging time requiring sedation, sensitivity to motion both respiratory and bowel and limited availability it is generally used as a problem-solving tool. Plain films and contrast studies currently have a very limited role in the evaluation of GI masses. Plain films may be diagnostic in masses like teratomas, while contrast studies may be required for mucosal abnormalities and small endoluminal masses like polyps. Enteric duplication cysts can occur along any portion of the GI tract from the pharynx to the rectum. They are found most often in the ileum (33%). The other sites are the oesophagus (20%), colon (13%), jejunum (10%), stomach (7%) and duodenum (5%). The incidence is reported to be around 0.2% of all children, with a slight male predominance. Duplication cysts are thought to arise between the 4th and 8th weeks of development; however, their aetiology is unknown and several different theories have been proposed. In 16%–26% cases there may be associated spinal defects, cardiac or urinary malformations. Other associated GI anomalies may be seen in about 10% of cases. Duplication cysts typically lie along the mesenteric border of the gut. They have smooth muscle wall which they share with the adjacent gut, as also the blood supply. On histopathology, three classical features are seen: an epithelial lining with GI mucosa, a smooth muscle covering and a close attachment to the GI tract due to a shared common wall. Although the mucosal lining does not necessarily match with the adjacent portion of the gut, nonetheless, duplications are named after the portion of the gut to which these are closely related. About 20%–30% of these duplication cysts contain ectopic gastric mucosa which is more common in oesophageal and small intestinal duplication cysts. Ectopic pancreatic mucosa may also be seen, most often in gastric duplication cysts. Morphologically they may be cystic or tubular with the former accounting for 80% and the latter for 20%. While cystic duplications mostly do not communicate with the adjacent gut, tubular duplications that run parallel to the GIT often do, hence in these cases connection with GIT must be demonstrated for operative planning. Duplication cysts may be multiple in 1%–7% of cases usually occurring in the same segment of the GIT. An atypical duplication cyst is an isolated duplication cyst which is completely separated from the GIT and has no communication or shared wall with the gut. They are extremely rare and thought to be the result of a vascular injury. Although duplication cysts may remain asymptomatic till adulthood they mostly present within the first year (70%). Almost 85% present by the second year. Clinical features depend not only on the size and location, but also on the presence of any ectopic mucosa and complications. Pharyngeal and oesophageal cysts may present with respiratory distress or dysphagia. Infection and rapid growth may cause retrosternal pain or haemoptysis. Gastric and intestinal duplications cause nonspecific symptoms like recurrent abdominal pain nausea, vomiting, distention or a palpable lump. High pressure inside the cyst consequent to accumulation of secretions is thought to be responsible for the recurrent abdominal pain. Obstruction due to intussusception or extrinsic compression may occur. Presence of gastric mucosa is associated with complications like inflammation, bleeding, ulceration and perforation. US is most useful for the diagnosis of abdominal duplication cysts and sometimes may demonstrate these on an antenatal scan. MR and CT are mainly used for oesophageal duplication and for planning surgery. Endoscopic trans oesophageal ultrasound may be informative for oesophageal duplications but is not a part of routine practice. Classical US features in uncomplicated duplication cysts include: a unilocular cystic structure in close proximity to the bowel. The cyst has a relatively thick wall which has a hyperechoic inner lining representing the mucosa and an outer hypoechoic rim produced by the smooth muscle layer (muscularis propria) – the so-called ‘gut signature’ sign. This sign is also referred to as the ‘double-wall’ or ‘muscular rim’ sign. (Fig. 7.7.1) Though, most characteristically described for duplication cysts, it may sometimes be seen in some other cystic lesions such as a complicated mesenteric cyst, Meckel’s diverticulum or torsed ovarian cyst. Some US signs described recently are: the ‘five layered cyst wall sign’ and ‘Y configuration of the muscle wall sign’. It has been shown that with the use of high frequency US transducers (12–18 MHz) the wall of the duplication cyst can demonstrate the same five layered wall structure as the normal GIT. From inside to outside these are: the innermost mucosa which is hyperechoic, muscularis mucosa (hypoechoic), hyperechoic submucosa, hypoechoic muscularis propria and the outermost serosa which is hyperechoic. If all these five layers can be identified in a cyst it is diagnostic of an enteric duplication cyst; however, the sign is difficult to demonstrate (Fig. 7.7.2A and B). The second sign arises because a duplication cyst shares its wall with the adjacent gut. Splitting of the common muscularis propria between the cyst and the adjoining bowel loop results in the Y configuration of the muscle layer on US, reflecting one of its important histological features. When this is seen, it is possible to confidently diagnose an enteric duplication cyst as this sign has not been demonstrated in other cysts. The ‘Y configuration sign’ can be particularly valuable in complicated cysts In addition, since US is a real time examination it also allows visualization of peristalsis of the cyst wall which is seen as a transient change in the cyst shape and contour due to contraction of the cyst wall (Fig. 7.7.3A and B). This requires the transducer to be kept stationary over the cyst for some time Most duplication cysts are anechoic but some echoes due to mucinous fluid or septations can be seen at times, and do not imply complications. Complications include haemorrhage (due to ectopic gastric mucosa), enzymatic destruction of the mucosal lining, inflammation (due to ectopic pancreatic tissue) and infection. In these cases, fluid levels or echogenic debris can be seen within the cyst with a thick hypervascular wall which may lack layers (see Fig. 7.7.2B). In these cases, the Y configuration sign can help to suggest the correct diagnosis. The inflammatory changes may extend to the surrounding mesenteric fat as well, which becomes hyperechoic. Duplication cysts near the ileocecal valve, can act as a lead point for intussusception. Atypical or isolated duplication cysts, may just produce the pseudokidney sign. The important US features of duplication cysts are listed in Box 7.7.1. Key Ultrasound Signs of Duplication Cysts CT is not performed for the diagnosis of duplication cysts, however, it can demonstrate the location, anatomical relationships, exact extent and the associated anomalies. On CT evaluation a cystic mass closely related to the adjoining GI wall is seen. The wall shows mild enhancement. A complicated duplication cyst showing internal high attenuation, air foci, thick enhancing wall and surrounding inflammation suggests infection (Fig. 7.7.4A and B). Internal high attenuation alone, however, could be due to proteinaceous contents or haemorrhage and does not imply infection on its own. MR is also not generally used as a diagnostic tool due to long examination times requiring sedation but is especially useful to demonstrate the cystic nature of thoracic duplications. Duplications cysts show hypointense signal on T1W sequences and very high signal on T2W images. Both CT and MR may be used prior to surgery. MR may have an additional value in assessment of foetal abdominal cysts. Differential diagnosis includes other cystic lesions such as mesenteric, omental, ovarian and choledochal cysts. In patients with an antenatal diagnosis, although the optimal time for resection is not defined, it is suggested that early resection within the first 6 months be considered. Treatment of asymptomatic duplication cysts remains controversial, however, since early elective surgery is associated with less morbidity and a shorter hospital stay than excision in symptomatic cases, it should be preferred. In addition, complications such as obstruction or massive bleeding may be life threatening and there is a potential risk for malignant transformation in adults. The cyst can be removed alone, but if there is a communication with the adjoining gut its resection will be required. Currently, minimally invasive surgery is becoming the procedure of choice. Complete excision is important to avoid cyst recurrence or subsequent malignant changes. Hypertrophic pyloric stenosis was earlier believed to be a developmental anomaly but is currently thought to be acquired disorder. It is characterized by hypertrophy of the circular muscle layer of the pylorus. This results in thickening and lengthening of the pylorus ultimately progressing to gastric outlet obstruction. The etiopathogenesis of this disorder is unknown but postulated mechanisms include abnormal innervation of the pylorus and duodenal irritation due to hypersecretion. Various genetic and environmental factors such as maternal smoking and use of erythromycin have also been implicated. Several ultrastructural anomalies have been identified in the muscle layer including abnormal nerve endings, decreased synthesis of nitric oxide, reduction in the number of cells of Cajal and an increased production of insulin-like growth factors. These factors are thought to cause muscle hypertrophy and failure of muscle relaxation. The incidence of HPS is about 3 per 1000 live births with male to female ratio of 4–5:1. Patients usually present between 2 and 6 weeks of age. The child is typically normal at birth and, subsequently develops non-bilious vomiting around 2–3 weeks of age. There may be a small lump palpable in the epigastrium, the so called ‘pyloric olive’, in nearly 80% of cases. Also, peristalsis may be seen traversing the epigastrium. If vomiting persists, dehydration and hypochloraemic alkalosis can develop. In the presence of a classical history and examination findings, the diagnosis is often made clinically and imaging is only used to confirm the diagnosis. US allows visualization of the pyloric canal morphology as well as behaviour during dynamic evaluation. Ultrasonography is performed with a high-frequency transducer ranging between 6 and 15 MHz. The child is placed supine and the examination is best performed with a moderately fluid-filled stomach. The first step is to localize the gallbladder as the pylorus lies posteromedial to the gallbladder. Another useful trick is to first localize the upper pole of the right kidney and then move the transducer medially towards the xiphoid to identify the pylorus. The transducer is then angulated so that the pyloric canal is well visualized in long axis. Correct positioning so that the pyloric canal is seen as a straight line is important to ensure that measurements are not performed in a tangential plane which can result in fallacious increase in muscle thickness. Sometimes a gas distended stomach may preclude visualization of the pylorus. In this case, shifting the child to an oblique position so that the right side is placed inferiorly allows fluid to move into the antrum which acts as an acoustic window. A markedly distended stomach can displace the pylorus posteriorly making it difficult to visualize. In this situation moving the child so that the left side is down or prone positioning may help localize the pylorus. Distention of the stomach with water rather than milk may be a better option as milk may at times also cause artefacts. A nasogastric tube may be used to fill up the stomach when necessary. The most important diagnostic feature of HPS is thickening of the muscle layer of the pylorus seen as hypoechoic curved bundles between the antrum and duodenal cap. A number of signs that have been described on sonography in HPS include the ‘empty cervix’ sign as the hypertrophied muscle mass indents the fluid filled antrum and duodenal bulb mimicking the appearance of cervix in longitudinal section (Fig. 7.7.5). The hypertrophied pylorus gives appearance of a ‘doughnut or a target’ in transverse section. The ‘antral nipple’ or ‘mucosal nipple sign’ refers to redundant pyloric canal mucosa protruding into the antrum. This can be seen as an echogenic structure protruding into the fluid filled antrum (Fig. 7.7.6). The fluid trapped between the mucosal folds in the centre of an elongated pylorus may be seen as two sonolucent lines in the centre referred to as the ‘double track sign’. Measurement of muscle layer thickness provides objective assessment of pyloric thickening. A muscle layer thickness of more than 3 mm is considered abnormal. This should be measured from the outer echogenic edge of the mucosa to the outer edge of the muscle on both transverse and longitudinal scans. A muscle wall thickness of less than 2 mm is considered normal while a thickness between 2 and 3 mm is considered equivocal as it can be seen in other conditions like pylorospasm or gastritis besides HPS. Pyloric canal length of less than 14 mm is thought to be unequivocally normal. A pyloric canal length of more than 15–17 mm is also considered diagnostic for HPS (Fig. 7.7.6). However, the measurement of canal length is much more difficult to perform and subject to variability, hence this should not be used alone to make the diagnosis of HPS. Total diameter of the pylorus over 13 mm and pyloric volume measurements have also been suggested, but muscle thickness measurement remains the most accurate In addition, real-time observation of the pyloric behaviour is also important. The stomach tends to be markedly distended and shows increased peristalsis with failure of gastric contents to pass into the duodenum. The other hand a wide-open pylorus with normal passage of contents into the duodenum excludes the diagnosis of HPS (Box 7.7.2). Key Ultrasound Features of Hypertrophic Pyloric Stenosis There is a higher incidence of renal anomalies in these patients and hence kidneys should be examined once diagnosis is established. Pitfalls in sonographic diagnosis include non-visualization of the pylorus due to an overdistended stomach pushing it posteriorly, tangential views of the pylorus or pylorus spasm producing a pseudo thickening of the pylorus. Hence it is important to make sure that the pylorus is correctly imaged and the entire pyloric length is visualized in longitudinal plane. As opposed to pylorus spasm the thickening and the appearance of pylorus in HPS tends to persist, while pylorospasm is transient and generally resolves within 30 minutes. If the muscle layer measures 2–3 mm in thickness and the pylorus does not relax during the US examination clinical follow up and repeat US examination may be advisable. Borderline measurements are more likely to occur in preterm infants. Some authors have provided measurements of the pyloric length and muscle thickness correlated with the age and weight of the child which may be helpful in small and premature patients. In equivocal cases, it is preferable to perform a repeat examination rather than erroneously make a false-positive diagnosis, as HPS is not a surgical emergency. A barium study may be performed if the US examination is inconclusive. Use of a nasogastric tube is preferred as it allows a controlled filling of the stomach with barium. The upper GI barium study shows delayed emptying of the stomach. The pyloric canal is elongated and narrowed visualized as a curved streak of barium directed upwards and posteriorly, referred to as the ‘string sign’. This combination of narrowed pyloric canal along with elongation is the most important feature of HPS on contrast studies. A ‘double string sign’ is produced when barium is caught between the folds of mucosa overlying the hypertrophied muscle. ‘Antral beaking’ refers to a mass impression upon the antrum with a streak of barium entering the narrowed pyloric canal. The hypertrophied muscle may indent the gastric antrum as well as the duodenal bulb producing the ‘shoulder sign’. Disruption of the antral peristalsis may result in a small outpouching along the lesser curvature of stomach producing the ‘pyloric teat’ sign. The stomach shows hyperperistalsis on fluoroscopic examination which is sometimes referred to as the ‘caterpillar sign’ (Box 7.7.3). These signs may sometimes be seen in pylorospasm but are not persistent. Therefore, it is important that the study is of sufficient duration to establish the persistence of findings. Any the residual barium must be aspirated if a nasogastric tube has been placed, in order to avoid aspiration. Key Upper GI Features of Hypertrophic Pyloric Stenosis The treatment of HPS is surgical, that is, Ramstedt’s pyloromyotomy. Postoperatively, in the first week, the muscles may remain same in thickness or even thicker than that prior to surgery. The muscle thickness gradually returns to normal with the anterior part of the muscle normalising first followed by the posterior portion. The anterior portion usually returns to a thickness of 3 mm within 3 months whereas the posterior portion may take up to 5 months to normalize. This is related to the usual anterior surgical approach. The gastric emptying, however, returns to normal within 2–3 days. An upper GI examination may also be used to assess gastric emptying postoperatively and to exclude gastro-oesophageal reflux if vomiting persists. GI polyps are commonly seen in childhood between 2 and 5 years of age and are the most common cause of painless rectal bleeding in children. Juvenile polyps are the most common type of polyps that are seen in children accounting for about 80% of the polyps. The term juvenile refers to the histology of the polyp rather than the age at which they occur. Histologically these lesions represent benign hamartomas. Diagnosis is made via rectal examination or sigmoidoscopy. Radiological examination is rarely required for diagnosis. Polyps in the distal colon and rectum can be easily resected via endoscopic polypectomy. Inflammatory polyps are commonly seen in children with inflammatory bowel disease. Most of these polyps are actually pseudo polyps formed by hyperplastic and inflamed mucosa in areas of inflammation and mucosal injury. They may reach large sizes but are not pre-malignant. Imaging reveals evidence of inflammatory bowel disease along with polyps. Juvenile polyposis syndrome is an inherited condition characterized by multiple juvenile polyps. The following criteria must be met for diagnosis of JPS (a) more than 5 juvenile polyps in the colon or rectum (b) juvenile polyps in other portions of the GI tract (c) any number of juvenile polyps with a positive family history. Single juvenile polyps are not a feature of juvenile polyposis syndrome. JPS has been categorized into 3 types depending on the clinical features and age of onset. Diffuse juvenile polyposis of infancy presenting in children up to 3 months of age. generalized juvenile polyposis with polyps throughout the GIT and juvenile polyposis coli (JPC) with polyps only in the colon beginning in children and adolescents. Children can present with diarrhoea, rectal bleeding, intussusceptions or anaemia. The diagnosis is usually established by colonoscopy. Double-contrast barium enema or CT colonogram can also demonstrate multiple polyps within the colon. Peutz-Jeghers syndrome is rare autosomal dominant disorder characterized by hamartomatous polyps throughout the GI tract. Mutation of ATK tumour suppressor gene is seen in some cases. Histologically these polyps are characterized by a smooth muscle core arising from the muscularis mucosa. Although polyps occur anywhere in the GI tract, they are commonest in the small bowel. They can be sessile or pedunculated and being firm in nature (due to the smooth muscle core) are particularly prone to intussusception. They may also present as GI bleed and anaemia Although the polyps of PJS are benign hamartomas the syndrome is associated with an increased risk of malignancy (adenocarcinoma). The diagnosis of PJS can be made if there are 2 or more histologically confirmed PJS polyps, any number of PJS polyps along with the typical mucocutaneous pigmentation, a family history of PJS with any number of PJS polyps or the presence of mucocutaneous pigmentation with family history of PJS. Because of the presence of small bowel polyps CT enterography, MR enterography and small bowel enema also have an important role to play in the diagnosis. The polyps are seen as sessile or pedunculated intraluminal filling defects on these studies. They may also be seen as the lead point of an intussusception (Fig. 7.7.7). Endoscopy and colonoscopy are used to establish the diagnosis. Video capsule endoscopy is especially useful to identify the polyp burden in the small bowel. The treatment is aimed at removing the larger polyps either surgically or endoscopically so as to avoid obstruction.
7.7: Gastrointestinal masses in children
Introduction
Imaging modalities
Ultrasound
Computed tomography
Magnetic resonance imaging
Congenital masses
Enteric duplication cysts
Imaging
Management
Hypertrophic pyloric stenosis (HPS)
Imaging
Ultrasound
Borderline measurements
Upper GI examination
Management
GI tract polyps and polyposis syndromes
Juvenile polyps
Inflammatory polyps
Polyposis syndromes
Juvenile polyposis syndrome (JPS)
Peutz-jeghers syndrome (PJS)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








