Chapter Outline
Abdominal hernias are commonly encountered in clinical practice, and they account for approximately 750,000 operations per year in the United States. Most herniations involving the anterior abdominal wall or groin can be easily diagnosed by inspection and palpation. In these cases, the radiologic examinations are useful for preoperative demonstration of the hernia’s contents and associated complications, such as bowel obstruction or ischemia. Diagnostic radiology is the principal means of detecting the internal, diaphragmatic, and other nonpalpable or unsuspected hernias.
Multidetector computed tomography (MDCT) can show several features of hernias that are critical to diagnosis and management: the precise anatomic site of the hernia sac; the shape of the sac and its connections; the contents of the sac; the hernia cuff and surrounding wall; and complications of the intestinal, vascular, omental, and mesenteric wall and cavity of the hernia sac. The multiplanar capabilities of MDCT offer exquisite detail of the anterior abdominal and pelvic wall. It can identify wall hernias and their contents, detect postoperative complications, and characterize hematomas, abscesses, and neoplasms.
This chapter describes the clinical and radiologic features of various abdominal herniations. In many instances, small and reducible hernias are discovered incidentally during gastrointestinal barium studies or CT of the abdomen performed to investigate unrelated conditions. Asymptomatic hernias do not warrant surgical repair, but their presence should always be brought to the attention of the patient and the referring physician. If intestinal obstruction attributable to an incarcerated hernia develops at a future time, it can then be treated without undue delay while the underlying cause is sought.
Hernia Classification
Abdominal hernias can be classified as three major types. Internal hernia denotes protrusion of gut through a peritoneal or mesenteric aperture of omentum, mesentery, or peritoneal ligament, leading to its encapsulation within another compartment of the otherwise intact abdominal cavity. An external hernia (i.e., abdominal wall hernia) is caused by prolapse of an intestinal loop, omentum, or mesentery through a defect in the wall of the abdomen or pelvis. Diaphragmatic hernias, in which gut, omentum, or mesentery herniates into the chest, are usually considered a separate category.
The responsible hernia orifice is often a preexisting anatomic structure, such as the epiploic foramen of Winslow, inguinal canals, or esophageal hiatus. Pathologic defects of congenital, postsurgical, or traumatic origin are also potential sites of herniation. Mobile segments of the small or large bowel are usually the content of abdominal hernias, but the greater omentum and various other viscera or pelvic organs are occasionally involved.
The nomenclature used for specific hernias indicates the anatomic location of their orifice rather than the nature of the protruding organ. The classification is therefore based on topographic distribution of the relatively common types of hernias ( Box 112-1 ).
Internal Abdominal Hernias
Paraduodenal
Foramen of Winslow
Pericecal
Intersigmoid
Transmesenteric
Retroanastomotic
External Abdominal or Pelvic Hernias
Abdominal wall
Umbilical
Ventral
Spigelian
Lumbar
Incisional
Pelvic walls and groin
Inguinal
Femoral
Obturator
Sciatic
Perineal
Diaphragmatic Hernias
Esophageal hiatus
Foramen of Bochdalek
Foramen of Morgagni
Acquired defects
Hernias
Internal Abdominal Hernias
The autopsy incidence of internal hernias is in the range of 0.2% to 0.9%. They are becoming more common with increasing use of bariatric surgery and liver transplantation. Figure 112-1 shows their typical location. Internal hernias may remain clinically silent if they are easily reducible, but the larger ones often cause vague epigastric discomfort, colicky periumbilical pain, and recurrent episodes of intestinal obstruction. Physical examination may reveal a palpable mass of herniated loops with localized tenderness. In these patients, the correct diagnosis can be made if barium study or CT of the abdomen is performed during the symptomatic periods. Otherwise, the hernia may not be recognized after it is reduced spontaneously or after bowel decompression with a nasogastric tube.

By far, the most common presentation of an incarcerated internal hernia is acute small bowel obstruction. In a large series of patients who underwent operation for bowel obstruction, the underlying causes proved to be internal hernia (4.1%), external hernia (17.5%), neoplasms (18%), or adhesions (32%).
Each type of internal hernia has specific radiologic features ( Table 112-1 ), which are described later in this chapter. As a general rule, barium studies and CT show certain diagnostic hallmarks shared by various internal hernias, including abnormal location of an intestinal segment in susceptible regions, such as the lesser sac; encapsulation and crowding together of several small bowel loops within the confines of the peritoneal cavity; stasis of contrast material in the lumen and dilation of more proximal bowel; and apparent fixation of the herniated loops, preventing their separation or dislodgment during fluoroscopic manipulations or by changing the position of the patient.
| Hernia Type | Subtype | Incidence * | Characteristic Clinical Findings | Radiography and Barium Studies | CT Findings | Key Vessel |
|---|---|---|---|---|---|---|
| Left paraduodenal | Congenital, normal aperture | 40% of all hernias, 75% of paraduodenal hernias | Postprandial pain, may date back to childhood | Encapsulated cluster of the jejunum in LUQ, lateral to the ascending duodenum; may have mass effect, indenting the posterior wall of stomach or displacing the transverse colon inferiorly | Clustered dilated small bowel loops between the stomach and pancreas, behind the pancreas itself, or between transverse colon and left adrenal gland | IMV in the neck of the hernia sac with anterior upward displacement of the IMV |
| Right paraduodenal | Congenital, normal aperture | 13% of all hernias, 25% of paraduodenal hernias | Postprandial pain, may date back to childhood | Encapsulated loops lateral and inferior to the descending duodenum; associated with small bowel nonrotation | Encapsulated loops lateral and inferior to the descending duodenum; associated with small bowel nonrotation | SMA displaced anteriorly |
| Pericecal | Congenital or acquired, abnormal aperture | 13% | RLQ pain, differential diagnosis of appendicitis; high incidence of occlusive symptoms | Clustered small bowel loops (usually distal) posterior and lateral to the cecum in the right paracolic gutter | Clustered small bowel loops (usually distal) posterior and lateral to the cecum in the right paracolic gutter | None |
| Foramen of Winslow | Congenital, normal aperture | 8% | Symptoms of proximal obstruction because of mass effect on stomach; symptom onset often preceded by changes in intra-abdominal pressure (i.e., parturition, straining); relief of symptoms with forward bending | Circumscribed loops medial and posterior to the stomach; differential diagnosis of cecal volvulus | Loops in the lesser sac between the liver hilum and IVC | None; vessels stretched through the foramen of Winslow |
| Intersigmoid | Type 1: congenital, normal aperture Types 2 and 3: acquired, abnormal aperture | 6% | None | U- or C-shaped cluster of small bowel posterior and lateral to the sigmoid colon | U- or C-shaped cluster of small bowel posterior and lateral to the sigmoid colon | None |
| Transmesenteric † | In children: congenital, abnormal aperture In adults: usually acquired, abnormal aperture | 8% | Two typical patient populations: children and postsurgical adults In adults, less vomiting because fewer secretions in proximal gastric pouch, onset more acute | Variable, air within gastric remnant; may simulate a left paraduodenal hernia | Small bowel lateral to the colon; displaced omental fat with small bowel directly abutting the abdominal wall | None |
| Retroanastomotic † | Acquired, abnormal aperture | 5% | Usually within the first postoperative month; less vomiting because fewer secretions in the proximal gastric pouch | Variable | Variable | None |
* Incidences for the first six types are from Meyers ; they are historic data but are the only major source available. Incidences for these first six types of internal hernias total only 93% because the perivesical hernias reported are not true internal hernias, so they were not included in this review.
† Probably more transmesenteric and retroanastomotic internal hernias currently because of the number of liver transplantations and gastric bypass operations being performed throughout the United States during the past decade. The 5% refers to the incidence after Roux loops were used during surgery for reasons other than liver transplantation or gastric bypass.
Preoperative radiologic diagnosis or a high index of suspicion for an internal hernia is important because at laparotomy, spontaneous reduction or inadvertent traction of the herniated loops may cause it to be overlooked. The usual abdominal exploration is inadequate for evaluating all peritoneal fossae and mesenteric defects that could serve as potential sites of herniations.
Paraduodenal Hernia
Paraduodenal hernias are the most common type of intra-abdominal herniation, and they account for 53% of the reported cases. They are more common in men than in women, with a ratio of about 3 : 1. Approximately 75% occur on the left and involve the paraduodenal fossa of Landzert. This peritoneal pocket is observed at 2% of autopsies. It is located just lateral to the ascending, or fourth, segment of the duodenum, beneath a peritoneal fold elevated by the inferior mesenteric vein and ascending left colic artery. Small bowel loops enter the sac from behind, where the duodenum emerges from its fixed retroperitoneal position. They protrude farther posteriorly and to the left, essentially herniating into the descending mesocolon and distal portion of the transverse mesocolon.
Twenty-five percent of paraduodenal hernias develop on the right side of the abdomen and typically involve the mesentericoparietal fossa of Waldeyer. This abnormal pocket in the jejunal mesentery is found at 1% of autopsies. Its orifice is located immediately behind the superior mesenteric artery and inferior to the transverse segment of the duodenum; however, the peritoneal pocket itself extends to the right and downward, directly in front of the posterior parietal peritoneum. Accordingly, the right paraduodenal hernia can be viewed as small bowel herniation into the ascending mesocolon.
The clinical manifestations of paraduodenal hernias range from intermittent and mild digestive complaints to acute intestinal obstruction. Recurrent periumbilical cramps or postprandial epigastric pain and distention are frequently experienced before the onset of incarceration. Contrast examinations of the gastrointestinal tract are most likely to provide the correct diagnosis during symptomatic periods. After the hernia is reduced spontaneously, however, results of barium studies tend to be negative, and the patient may be mislabeled as psychoneurotic.
The characteristic radiographic features of left paraduodenal hernias have been described by several investigators. A circumscribed, ovoid mass of multiple jejunal loops occupies the left upper quadrant immediately lateral to the ascending duodenum ( Fig. 112-2 ). The hernia indents the posterior gastric wall and depresses the distal transverse colon. Fluoroscopy and serial radiographs reveal a separation of encapsulated loops from the remaining small intestine. Dilation of the involved segments and stasis of barium may also be evident. Because the duodenum is retroperitoneal and the herniated proximal jejunal loops enter the fossa of Landzert posteromedially, only one loop of distal small intestine exits through the hernia orifice. The inferior mesenteric vein and ascending left colic artery lie in the anteromedial border of the left paraduodenal hernia. These findings are best appreciated during laparotomy, but CT scans of the upper abdomen may help demonstrate them before operation.


Right paraduodenal hernias are manifested as a similar ovoid grouping of several small bowel loops just lateral and inferior to the descending duodenum ( Fig. 112-3 ). They are usually more massive and fixed than those on the left side. Afferent and efferent intestinal loops pass through the hernia orifice, where they are closely apposed and narrowed. Lateral radiographs are particularly useful for demonstrating the retroperitoneal displacement of the hernia contents. Barium enema examination shows that the ascending colon lies lateral to a right paraduodenal hernia, and the cecum retains its normal position.


The superior mesenteric artery and its ileocolic branches are situated in the anterior wall of the right paraduodenal hernia sac. The passage of herniated loops behind these vessels causes arteriographically detectable changes. The jejunal arteries, which normally arise from the left side of the superior mesenteric artery, abruptly reverse direction to the right and course behind the parent vessel to supply the herniated loops in the fossa of Waldeyer.
Operative techniques for the correction of paraduodenal hernias are described elsewhere. Preoperative diagnosis and demonstration of their radiographic anatomy assist the surgeon to better understand the extremely confusing laparotomy findings. Blind division of the hernia sac should be avoided because it carries the risk of injury to the vital mesenteric vessels contained within its wall. It may create a mesenteric defect that could later serve as a potential site for an iatrogenic internal hernia.
Foramen of Winslow Hernia
The lesser sac communicates with the greater peritoneal cavity through the epiploic foramen of Winslow. This small opening can serve as a pathway for protrusion of viscera into the lesser sac, where 8% of all internal hernias occur. The small intestine is the herniated segment in 60% to 70% of cases. The terminal ileum, cecum, and ascending colon are involved in about 25% to 30%. The transverse colon, gallbladder, and omentum account for the remainder. Predisposing factors include an enlarged foramen of Winslow and excessively mobile intestinal loops because of a long mesentery or persistence of the ascending mesocolon. Herniation into the lesser sac may be provoked by a sudden increase in intra-abdominal pressure, like that experienced while lifting heavy weights or during parturition. An elongated right lobe of the liver can also be a contributing factor by directing the mobile intestinal loops toward the foramen of Winslow.
This type of internal hernia usually affects middle-aged patients and is manifested as progressive upper abdominal pain and acute onset of small bowel obstruction. Physical examination usually reveals localized tenderness and distention in the epigastric regions. A helpful sign is the relief of pain with forward bending or in the knee-chest position.
Several radiologic features of hernias through the foramen of Winslow are characteristic:
- 1.
Plain radiographs of the abdomen show bowel loops containing gas within the lesser sac medial and posterior to the stomach, usually in conjunction with markedly dilated proximal small intestine. The right iliac fossa may appear empty if the cecum and ascending colon are the herniated segments.
- 2.
Upper gastrointestinal examination demonstrates displacement of the stomach to the left and anteriorly because of extrinsic compression by the mass of herniated loops in the lesser sac. The first and second portions of the duodenum may be compressed and deviated to the left side.
- 3.
Small bowel series show dilation and hyperperistalsis of the intestinal loops that indicate distal mechanical obstruction. Serial radiographs and fluoroscopy can localize the site of obstruction to the right upper abdomen, corresponding to the anatomic location of the foramen of Winslow between the duodenal bulb and the hilum of the liver.
- 4.
If the herniation involves the cecum and ascending colon, barium enema examination may reveal a tapered narrowing or obstruction near the hepatic flexure.
- 5.
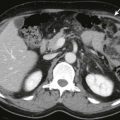
CT scans of the abdomen show an aberrant position of the bowel loops between the liver, the stomach, and the pancreas ( Fig. 112-4 ). CT is the optimal imaging technique to demonstrate the presence and content of herniations involving the lesser sac.


Figure 112-4
Foramen of Winslow hernia.
A. Graphic illustration of foramen of Winslow hernia shows bowel about to prolapse ( arrow ) into lesser sac, behind hepatoduodenal ligament, the free edge of the lesser omentum. B. CT scan shows small bowel ( arrows ) herniated into the lesser sac, causing a closed loop obstruction. C. Scan obtained several hours later shows that these loops have become ischemic and hemorrhagic.
( A from Martin LC, Merkle EM, Thompson WM: Review of internal hernias: Radiographic and clinical findings. AJR Am J Roentgenol 186:703-717, 2006. Reprinted with permission from the American Journal of Roentgenology.)
- 6.
Gallbladder herniation into the lesser sac can be diagnosed when the opacified gallbladder appears elongated and its fundus is malpositioned on the left, indenting the gastric antrum or body of the pancreas. Manual compression of the epigastrium can reduce the hernia if the foramen of Winslow is widely patent. Gallbladder strangulation and perforation in the lesser sac may occur if the condition is not diagnosed and treated promptly.
Besides the foramen of Winslow, there may be other openings for herniation into the lesser sac. For instance, a defect in the gastrohepatic ligament forming the anterior wall of the lesser sac may follow partial gastrectomy. A congenital or acquired defect may also be present in the transverse mesocolon or gastrocolic ligament, which constitutes the floor of the lesser sac. The intestinal loops can prolapse through these openings to occupy the lesser sac or reenter the greater peritoneal cavity by the foramen of Winslow.
Pericecal Hernia
Four peritoneal fossae located in the ileocecal region as well as congenital and acquired defects in the mesentery of the cecum or appendix may lead to development of a pericecal hernia. The various other terms used to classify these hernias (e.g., ileocolic, retrocecal, ileocecal, paracecal) appear to have limited practical value for radiologic differential diagnosis and surgical management.
In a collective review of 467 cases of internal hernia, 13% involved the ileocecal region. The clinical manifestations are usually intermittent episodes of colicky right lower abdominal pain associated with small bowel distention, nausea, and vomiting. Chronic incarceration may produce symptoms compatible with periappendiceal abscess, regional enteritis, or intestinal obstruction caused by adhesions.
In most cases, an ileal segment herniates through a defect in the mesentery of the cecum and occupies the right paracolic gutter. The correct diagnosis may be suggested on plain radiographs of the abdomen ( Fig. 112-5 ), provided the unusual relationship of the ileum to the cecum is recognized in association with small bowel obstruction. More useful are delayed radiographs of a small bowel series or a barium enema examination with retrograde opacification of the terminal ileum. Careful fluoroscopic evaluation and filming in lateral and oblique projections are particularly valuable for the demonstration of the fixed position of the herniated ileal loop posterolateral to the cecum.


Intersigmoid Hernia
Intersigmoid hernias involve the intersigmoid fossa, a peritoneal pouch located between the two loops of the sigmoid colon and its mesentery. This pocket is found at 65% of autopsies.
Intersigmoid hernias are usually reducible and are an incidental finding during laparotomy. Radiologic diagnosis is best made on barium enema studies in which retrograde filling of the small bowel has been achieved. The examination typically shows a portion of jejunum or ileum to be encapsulated between the sigmoid loops ( Fig. 112-6 ).


In this context, two similar entities deserve a brief mention:
- 1.
In transmesosigmoid hernias , a defect involving both layers of the sigmoid mesentery allows herniation of the small bowel loops toward the left lower abdomen, posterolateral to the sigmoid colon. The hernia ring is usually a long slit whose fibrous edge is bound by branches of the inferior mesenteric vessels.
- 2.
In intramesosigmoid hernia , a congenital defect may be present in only one of the constituent leaves of the sigmoid mesentery. The small bowel loops passing through this orifice are incarcerated in a hernia sac, which is formed by separation of medial and lateral leaves of the mesosigmoid.
Radiologic differentiation of these three types of hernias involving the mesosigmoid is often difficult, and in terms of their ultimate surgical management, it is irrelevant.
Transmesenteric Hernia
Approximately 5% to 10% of all internal hernias occur through defects in the mesentery of the small bowel. They have no limiting sac, but their functional significance is similar to that of true internal hernias.
Almost 35% of transmesenteric hernias affect the pediatric age group, in which they constitute the most common type of internal herniation. The underlying mesenteric defects are usually 2 to 5 cm in diameter and located close to the ligament of Treitz or the ileocecal valve. A causal relationship to prenatal intestinal ischemic accidents seems likely because such mesenteric apertures and associated hernias are frequently found in infants with atretic bowel segments. In adults, however, most mesenteric defects are probably the result of previous gastrointestinal operations, abdominal trauma, or intraperitoneal inflammation.
In the absence of a limiting hernia sac, a considerable length of small intestine may protrude through the mesenteric aperture. Volvulus may further complicate the process and cause rapid strangulation and intestinal gangrene. Patients present with severe periumbilical cramps accompanied by hyperactive bowel sounds and progressive distention. A tender abdominal mass, representing the Gordian knot of herniated intestine, may be palpable. Plain radiographs of the abdomen demonstrate a mechanical small bowel obstruction and occasionally show a single, distended closed loop ( Fig. 112-7 ). Small bowel examination may disclose a constriction around the closely approximated afferent and efferent limbs of the herniated intestine. The superior mesenteric arteriogram shows abrupt angulation and displacement of the visceral branches as they pass through the mesenteric defect to supply the herniated loops. These findings signal a surgical emergency, although clinical and radiologic differentiation of the mesenteric hernia from other closed loop obstructions (e.g., small bowel volvulus or entrapment beneath peritoneal adhesions) may be impossible.

Defects in the mesenteric attachments of the colon are also the potential sites of internal herniation. For example, a congenital or postsurgical opening in the transverse mesocolon permits small bowel loops to herniate into the lesser sac. A somewhat similar situation can be present in cases of congenital persistence of descending mesocolon. A defect in this structure may allow small bowel herniation into the left paracolic gutter, causing medial displacement of the descending colon.
Transomental Hernia
Approximately 1% to 4% of internal hernias occur through defects in the greater omentum. Fewer than 100 cases have been reported from observations made at surgery or autopsy. The hernia orifice is usually a slitlike opening up to 10 cm long, located in the periphery of the greater omentum. Most have a congenital origin, but trauma and inflammation may also produce omental perforations or weak areas. These defects can subsequently serve as potential sites for transomental herniation of the small intestine ( Fig. 112-8 ) and other mobile segments, such as the cecum or sigmoid colon. The clinical and radiologic findings are almost identical to those of transmesenteric hernias.

Retroanastomotic Hernia
Retroanastomotic hernias are a well-recognized and preventable complication of gastrointestinal surgery. They usually develop after partial gastrectomy and gastrojejunostomy. About 75% of cases involve incarceration of the efferent jejunal segment in the retroanastomotic space, which is created during antecolic or retrocolic anastomosis. The herniation is usually from right to left, so that the efferent jejunal loop or an excessively long afferent segment occupies the left upper quadrant of the abdomen. Less commonly, the ileum, the cecum, or the omentum is also involved.
Approximately half of these hernias occur in the first postoperative month, and the others occur within 1 year or several years after surgery. The presenting symptom is cramping abdominal pain, and the signs are those of high small bowel obstruction. The herniated loops are occasionally palpable as a tender mass in the left upper abdomen. These nonspecific findings may be mistaken for gastric outlet obstruction caused by stomal edema, dumping syndrome, or postoperative pancreatitis. Delay in establishing the correct diagnosis may lead to strangulation of the herniated loops; the associated mortality rate is up to 30% for surgically treated cases and almost 100% if the problem is not corrected.
Radiologic diagnosis of a retroanastomotic hernia requires careful fluoroscopic evaluation of the gastrointestinal tract after administration of barium or water-soluble contrast medium. The examination reveals that the site of obstruction is not the gastric stoma but is more distal in either of the anastomotic limbs. Gradual opacification of the partially obstructed efferent loop discloses its abnormal position lateral and posterior to the gastrojejunostomy ( Fig. 112-9 ). The herniated segments of jejunum appear clumped or fixed in the left upper abdomen, usually associated with some degree of dilation and stasis.

The diagnosis of afferent loop hernia, which occurs primarily after an antecolic anastomosis, is more difficult. The clinical findings include persistent epigastric pain and tenderness, nonbilious vomiting, and elevated serum amylase level. Contrast studies of the upper gastrointestinal tract show patency of the anastomosis and the efferent loop; however, opacification of the herniated afferent limb may not occur or may be delayed. CT scans or sonograms of the upper abdomen usually disclose the obstructed afferent loop as a fluid-filled and markedly distended tubular structure. Scintigraphy is another diagnostic method for this entity. Radionuclide agents excreted by the biliary tract into the duodenum permit visualization of the dilated afferent loop and the site of obstruction.
Like any other symptomatic internal hernia, hernias involving the retroanastomotic space also require surgical correction. However, this iatrogenic hernia should be prevented during initial gastric operation by the use of a short afferent loop and the closure of the retroanastomotic space with sutures placed between the jejunal mesentery and the transverse mesocolon.
Abdominal Wall Hernias
Herniations through the walls of the abdominal cavity develop in approximately 1.5% of the population and usually involve specific sites of congenital weakness or previous surgical incision. An outpouching of the peritoneum forms a hernia sac, which surrounds the protruding parts of the greater omentum, intestine, or other abdominal organs. The presenting symptoms are variable and depend on the size, location, and content of the hernia.
Umbilical Hernia
In infants and children, a patent umbilical ring is a common site of herniation. It often is manifested as a soft, asymptomatic bulge that tends to disappear spontaneously. In some instances, however, the development of umbilical tenderness and obstructive symptoms caused by incarceration necessitate surgical repair. The congenital entity known as an omphalocele is not a true umbilical hernia because the bowel loops never return to the peritoneal cavity during gestation in this disorder.
In adults, umbilical hernias occur predominantly in women with a history of multiple pregnancies and in patients with obesity or with increased abdominal pressure resulting from ascites and chronic bowel distention ( Fig. 112-10 ). The hernia contents are usually the greater omentum and various segments of the small or large intestine. Adhesions often develop between the protruding viscera and the peritoneal sac and cause digestive symptoms and other complications.

The clinical diagnosis of an incarcerated umbilical hernia should be suspected when a patient presents with intestinal obstruction and umbilical tenderness, even if there is no obvious bulge on the surface. Radiographs of the abdomen usually show the distended bowel loops and a tumor-like density, the umbilical hernia. A coned-down lateral or cross-table view of the anterior abdominal wall using low peak kilovoltage can be helpful in detecting the presence of omental fat or bowel loops in the umbilical hernia. Barium studies of the small or large intestine can show obstruction or protrusion of gut in the umbilical region. Ultrasound and CT studies of the abdomen establish the diagnosis when they demonstrate a “knuckle” of bowel protruding through an umbilical defect.
Ventral Hernia
The term ventral hernia encompasses several types of herniations through the anterior and lateral aspects of the abdominal wall ( Fig. 112-11 ). Most occur in the midline and emerge through the aponeurosis forming the linea alba. They are referred to as epigastric or hypogastric hernias, depending on their location above or below the umbilicus, respectively. The hernia aperture is often a small, firm defect that allows portions of the greater omentum, properitoneal fat, or a bowel loop to protrude anteriorly. Incarceration and strangulation of the contents may occur frequently and produce symptoms out of proportion to the objective findings. The severity of abdominal pain may simulate a perforating peptic ulcer. The pain of a ventral hernia, however, is aggravated by exertion and associated with focal tenderness of the abdominal wall.