Indeterminate renal masses remain a diagnostic challenge for lesions not initially characterized as angiomyolipoma or Bosniak I/II cysts. Differential for indeterminate renal masses include oncocytoma, fat-poor angiomyolipoma, and clear cell, papillary, and chromophobe renal cell carcinoma. Qualitative and quantitative techniques using data derived from multiphase contrast-enhanced imaging have provided methods for specific differentiation and subtyping of indeterminate renal masses, with emerging applications such as radiocytogenetics. Early and accurate characterization of indeterminate renal masses by multiphase contrast-enhanced imaging will optimize triage of these lesions into surgical, ablative, and active surveillance treatment plans.
Key points
- •
Renal lesions not characterized as Bosniak I/II cysts or angiomyolipoma are indeterminate and require additional imaging characterization.
- •
Multiphase contrast-enhanced imaging has emerged as a means of characterizing indeterminate renal masses, with clear cell renal cell carcinoma (RCC), papillary RCC, chromophobe RCC, fat-poor angiomyolipoma, and oncocytoma as primary differential considerations.
- •
Relative corticomedullary enhancement greater than that of adjacent renal cortex of an indeterminate renal mass is a strong predictor for clear cell RCC.
- •
Multiphase contrast-enhanced imaging allows for quantitative analysis of renal lesions with many emerging applications, such as radiocytogenetics.
Introduction
Incidentally discovered renal masses that don’t require immediate treatment and are not definitively characteristic of angiomyolipoma (AML) or Bosniak I/II cysts at initial imaging are indeterminate and require further imaging characterization. For indeterminate renal masses, size is the leading determinant of renal mass triage. The larger an indeterminate renal mass, the greater the likelihood of high-grade/malignant disease. Smaller indeterminate masses, conversely, are more likely benign. Despite these observations, small (pT1a or <4 cm in size) indeterminate renal masses comprise 38% of surgically resected renal masses with 20% to 30% found benign on pathologic analysis. Furthermore, given slow growth and low rate of progression/metastasis of small renal cell carcinoma (RCC) lesions, Jewett and colleagues advocate for active surveillance with serial imaging for small RCC. Although these small indeterminate renal masses are traditionally presumed malignant with partial nephrectomy as preferred treatment, accurate image characterization of these lesions will optimize triage to biopsy or ablative/surgical treatment and allow for better selection for active surveillance and to optimize follow-up based on risk of malignancy/benignity. This review focuses on current and evolving methods to triage indeterminate renal masses at multiphase imaging based on assessment of their enhancement characteristics.
Normal anatomy and imaging technique
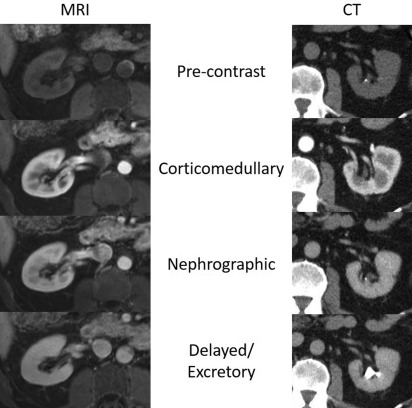
Current Society of Abdominal Radiology (SAR) computed tomographic (CT) protocol guidelines advocate for a minimum of unenhanced and nephrographic (120 sec) phases to assess for renal mass enhancement ( Fig. 1 ; Table 1 ). Unenhanced imaging provides a baseline to assess for lesion enhancement on subsequent phases and is also sufficient to assess for lesion homogeneity, macroscopic fat, and calcification. Uniform enhancement of the renal cortex and medulla during the nephrographic phase optimizes detection of heterogeneously enhancing lesions.
| CT | ||||
| Contrast : Low or isoosmolar, 35–52.5 g iodine (approximately 100–150 mL of 350 mg iodine/mL), rate: 2–5 mL/s | ||||
| Phase | Anatomic Coverage | Acquisition | Reconstructions | Additional reformats |
| Precontrast | Kidneys only | Axial | 3-mm slices, with or without 50% overlap | |
| Corticomedullary | Kidneys only | Axial, 40- to 70-s delay | 3-mm slices, with or without 50% overlap | Coronal/sagittal, 3-mm slices without overlap |
| Nephrographic | Kidneys only | Axial, 100- to 120-s delay | 3-mm slices, with or without 50% overlap | Coronal/sagittal, 3-mm slices without overlap |
| Excretory | Diaphragm to iliac crests | Axial, 7- to 10-min delay | 3-mm slices, with or without 50% overlap | Coronal/sagittal, 3-mm slices without overlap |
| MRI | ||
| Contrast : Extracellular gadolinium-based, 0.1 mL/kg, rate: 1–2 mL/s followed by 10–20 mL saline | ||
| Sequence | Planes/Thickness/Gap | Details/Alternatives |
| 2D T2 SSFSE | Axial/4–5 mm/no gap Coronal/5–6 mm/no gap | 2D T2 FSE/4- to 5-mm slice thickness/no gap |
| 2D T1 GRE in/out of phase | Axial/5–6 mm/0.5–1 mm | 3D Dixon/3 to 4-mm slice thickness/no gap |
| 3D T1 SPGR fat saturation, precontrast | Axial/3–4 mm/no gap Coronal/3–4 mm/no gap | |
| 3D T1 dynamic SPGR fat saturation, postcontrast | Axial/3–4 mm/no gap Coronal/3–4 mm/no gap | Postcontrast timing: 30, 90–100, 180–210 s, 5–7 min Obtain subtraction images as well |
| Diffusion-weighted imaging | Axial/5–6 mm/no gap | B values: 0–50, 400–500, 800–1000 s/mm 2 |
Corticomedullary and excretory postcontrast CT phases are optional per the SAR, but can be very helpful in delineating renal vascular and collecting system anatomy before any intervention that may be pursued. These additional phases also provide additional postcontrast time points to assess enhancement characteristics of renal masses.
Radiation exposure represents a significant risk for those undergoing CT surveillance, particularly given the prevalence of this modality in the medical community. Radiation dose of CT must be minimized, particularly in younger patients, because the equivalent of 8 lifetime CT scans theoretically increases lifetime cancer risk from 1/1000 to 1/82. As more is understood about differentiation of small indeterminate masses, directed measures must be taken to minimize radiation dose.
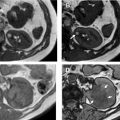
Dynamic contrast-enhanced MRI is a robust alternative technique for indeterminate renal mass characterization with similar lesion enhancement characteristics demonstrated as compared with CT (see Fig. 1 , Table 1 ). In addition to assessment of enhancement characteristics, MRI has the added benefit of T2-weighted, opposed phase, and diffusion-weighted imaging (DWI). Although generally tolerated well, MRI has unique limitations, primarily related to patient factors, such as motion, body habitus, claustrophobia, metallic implanted devices, and foreign bodies. In addition, MRI acquisition times are generally significantly longer than for CT, and the examinations are more costly.
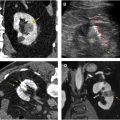
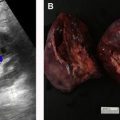
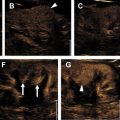
Contrast-enhanced ultrasound (CEUS) evaluation of indeterminate renal masses has been promising with less scan time than MRI, minimal risk of contrast morbidity, and no radiation exposure. Unlike CT and MRI, CEUS contrast boluses can be given multiple times during imaging acquisition, although this modality can be significantly limited in patients with large body habitus. CEUS image acquisition can also be difficult to reproduce, with a high level of interoperator variability.
Effective and safe active imaging surveillance of indeterminate renal masses requires judicious use of CT to minimize radiation dose. Periodic use of MRI and CEUS can allow for longer intervals between CT scans during the surveillance period with resultant decrease in patient radiation exposure.
Imaging findings/pathology
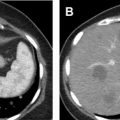
Incidentally discovered renal masses with macroscopic fat and absence of calcification on any precontrast or postcontrast phase enables confident diagnosis of AML, especially if the lesion is exophytic and of soft contour conforming to the surrounding renal parenchyma and perirenal tissues. Bosniak I/II cysts are characterized on unenhanced imaging based on homogeneity, CT attenuation values, or magnetic resonance (MR) T1 and T2 characteristics. Bosniak I/II cysts should also demonstrate absence of enhancement on all modalities. In lesions that enhance in the nephrographic phase and that lack macroscopic fat, the 5 primary differential considerations are clear cell, papillary or chromophobe RCC, oncocytoma, and fat-poor (FP) AML.
Under the current paradigm, these 5 subtypes of enhancing renal mass cannot be reliably differentiated based on enhancement characteristics derived only from unenhanced and nephrographic phases. If an enhancing small renal mass is detected, resection or ablation of the lesion with preservation of as many nephrons as possible represent the most definitive treatment options. Tissue diagnosis of the lesion is derived from the surgical specimen or a biopsy performed in conjunction with ablation. Accurate imaging-based subtyping of enhancing indeterminate renal masses will enable appropriate triage to biopsy, ablative or surgical treatment and will improve active surveillance.
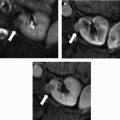
Given the broad differential diagnosis of small enhancing renal masses, which includes both malignant and benign entities, additional postcontrast phases are used to more specifically characterize indeterminate renal masses. Corticomedullary and excretory phases provide additional time points at which to assess lesion enhancement. Birnbaum and colleagues recognized the need for both corticomedullary and nephrographic phases to best assess for indeterminate renal mass enhancement, particularly for lesions that demonstrate peak enhancement later than the corticomedullary phase. In a small pilot study in 2000, Jinzaki and colleagues initially demonstrated differences between the most common renal mass subtypes based on their absolute peak enhancement and deenhancement in the corticomedullary and nephrographic phases, although these differences were not found to be statistically significant.
Generally, clear cell renal cell carcinomas (ccRCC) are round, oval, or lobular lesions that enhance heterogeneously and have a peak absolute HU greater than 160 on corticomedullary phase with unenhanced HU between 25 and 40, nephrographic HU at about 120, and excretory phase HU of approximately 100. Oncocytomas, the most common benign mimic of ccRCC, tend to be round or oval lesions that enhance more homogenously with peak corticomedullary HU 140 to 160 with deenhancement in the nephrographic phase near 120 HU and excretory phase HU values of about 100. Chromophobe RCC tend to be larger and more heterogenous compared with oncocytoma with peak enhancement in the nephrographic phase and excretory phase HU of 60 to 80. Patients with FP AML tend to be younger than patients with RCC. FP AML lesions tend to be ovoid- or mushroom-shaped space-occupying lesions that are typically 45 to 55 HU on unenhanced scans, with homogenous peak absolute enhancement of about 140 HU in the corticomedullary phase with deenhancement in the nephrographic and excretory phases at 100 to 120 and 80 to 100 HU, respectively. Finally, papillary RCCs tend to measure 40 to 50 HU on unenhanced scans and have absolute peak attenuation of 80 HU in the nephrographic phase with mild deenhancement to about 60 HU in the excretory phase. Unfortunately, there is overlap between several of the above characteristics that make indeterminate renal mass characterization difficult under the current paradigm ( Fig. 2 ).