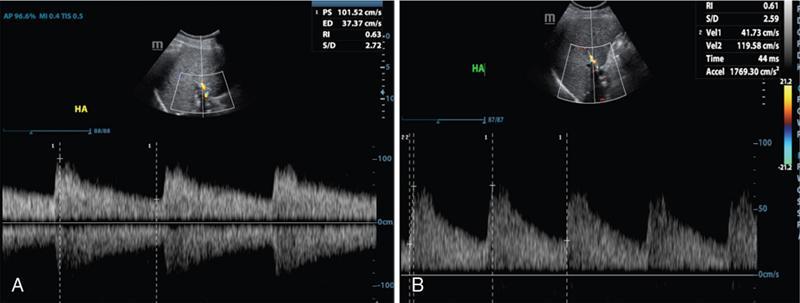
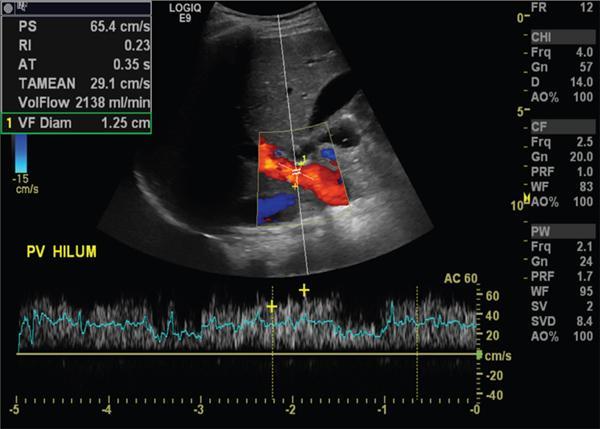
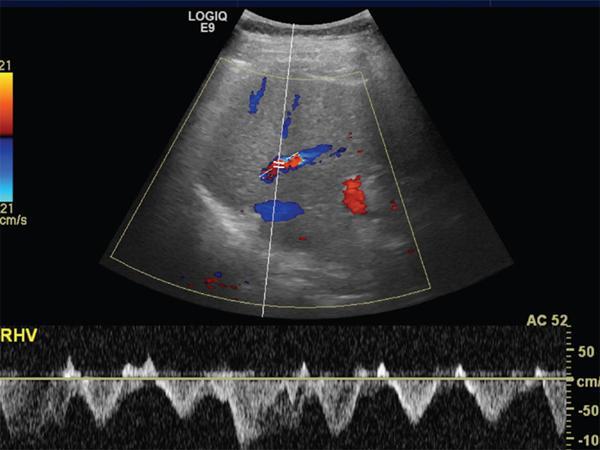
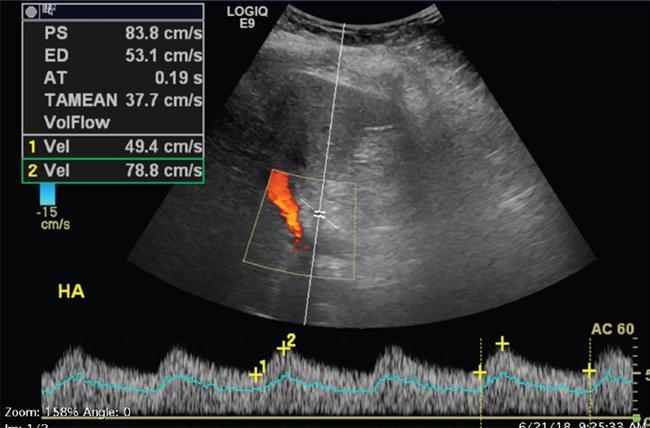
Imaging plays a significant role to make an early diagnosis and establish the best treatment options for posttransplant complications as clinical manifestations and laboratory parameters in many of the complications are usually nonspecific and overlapping. The imaging evaluation of postoperative complications remains the same in both types of liver transplants (DDLT and LDLT). Ultrasound with colour Doppler is usually the first modality of choice in the immediate postoperative period due to its easy and bed side availability with high accuracy and ability to pick up all major posttransplant complications, though it has a drawback of being operative dependent. It is usually part of regular postoperative assessment of the recipient for initial few postoperative days, playing an important role in picking up early complications. It is essential for the radiologist to know the details of the individual patient’s postoperative anatomy and surgical details, since anastomoses are the locations where complications occur most frequently. Goals of ultrasound evaluation in early postoperative period: Vascular evaluation with colour Doppler: CT/MRI imaging plays a supportive role for confirmation of the findings seen on ultrasound or colour Doppler or is done as an advanced imaging in case of mismatch between clinical or laboratory parameter and ultrasound or Doppler findings. MRI is particularly important for assessment of the biliary tree. First postoperative Doppler is done within 24 hours of the surgery and it serves as the baseline for comparison in the future. The haptic artery needs to be evaluated at the anastomotic site and also in the intrahepatic portions and the parameters to be evaluated are the systolic upstroke, PSV and resistive index (RI). Table 9.14.16 gives normal ranges of these parameters. Normal hepatic artery Doppler waveform demonstrates rapid systolic upstroke, with a systolic acceleration time (SAT) of less than 0.08 second and continuous diastolic flow, displayed above the baseline. A wide variability is observed in PSV of HA in the immediate postoperative period even in the absence of any hepatic arterial complication. Normal values may vary averagely from 40 to 120 cm/s. Vascular kinks or anastomotic site oedema in postoperative period can lead to falsely increased PSV in the HA. Correction of Doppler angle must be kept in mind while measuring the PSV of HA so that the angle aligns with the direction of the artery and must be less than 60 degrees to avoid inaccurate results. The hepatic arterial RI is an important tool to assess the hepatic arterial waveform in postliver transplant setting, as it allows semiquantitative estimation of the resistance to arterial flow into the liver. The normal RI ranges from 0.55 to 0.80 in posttransplant patients. The RI increases with decrease in the diastolic flow in the hepatic artery, which suggests increased resistance to the hepatic arterial flow. Absence or reversal of diastolic flow in the hepatic artery corresponds to an RI of 1.0 It is important to note that, increased RI of the hepatic artery (greater than 0.8) is a normal findings in the first few days after liver transplantation, due to decreased diastolic flow, which is attributed to allograft oedema, increased cold ischemia time, increased portal flow or vessel spasm. These transient arterial waveform abnormalities in early postoperative period usually resolve and normalize in 7–15 days on the follow-up Doppler studies. In view of significant variability in the PSV and RI of the hepatic artery in immediate postoperative period, other than in cases of absent arterial flow, the diagnosis of a hepatic arterial complication is seldom established on the basis of a single postoperative Doppler ultrasound. Arterial waveform abnormalities on the immediate postoperative scans should be followed closely and correlated with the patient’s clinical findings including liver function tests. Persistence of the abnormalities on serial follow-up Doppler studies or further deterioration of the waveform suggest presence of arterial complications. A decrease in hepatic arterial RI (RI < 0.55) is a more ominous finding than increased RI and is usually a concern for serious arterial complications like HA stenosis or impending thrombosis. The SAT – the interval from end-diastole to the first systolic peak – is a measure of the rapidity of the upstroke and normally should not exceed 0.08 seconds (Fig. 9.14.27). Portal vein evaluation needs to be done on greyscale ultrasound as well as on the colour Doppler. The presence of echogenic thrombus or filling defect is better appreciated on the greyscale ultrasound images. The PV peak velocity (measured at the anastomotic site) is the most important parameter for Doppler evaluation along with spectral waveform analysis. The normal portal vein flow after liver transplantation is hepatopetal, monophasic with respiratory plasticity (Fig. 9.14.28). The portal venous velocity is variable and tends to decrease on serial examinations after transplantation. During the early postoperative phase, turbulent flow with high velocities may be a normal finding which tend to decrease on serial examinations. Hepatic veins reveal a triphasic waveform (Fig. 9.14.29) due to transmission of cardiac pulsations. Though loss of triphasicity is seen in HV stenosis, it is a very nonspecific finding and biphasic or monophasic waveforms are often seen in normal posttransplant patients. Therefore, presence of hepatic venous triphasicity can exclude hepatic venous stenosis; however, loss of triphasicity does not imply presence of a hepatic venous complication. The terms early posttransplant complications is variably used in literature for period of first 1–3 months after the OLT. Table 9.14.17 summarizes post transplant complications in early and late post transplant period. Vascular complications are a common cause for allograft failure after hepatic transplantation. Early detection of the vascular complications is possible with the help of imaging, which can help reduce the incidence of graft failure. Table 9.14.18 below describes approximate timeline of various post transplant complications in post transplant period. Hepatic artery stenosis First three months Portal venous thrombosis Majority within four weeks Portal vein stenosis Usually late (>6 months) Hepatic venous stenosis Usually late (>6 months) Hepatic arterial complications are particularly significant in postliver transplant patients, as unlike native liver, the biliary tree of a transplant liver is entirely dependent on arterial blood from the hepatic artery. As a consequence, any hepatic arterial complication resulting in compromised hepatic arterial flow, particularly hepatic arterial thrombosis, has a devastating effect on the biliary epithelium, inducing ischemia resulting in biliary strictures, necrosis, abscesses and allograft failure. Hepatic artery complications include HAT, stenosis, and pseudoaneurysms. HAT is a common and the most catastrophic complication after orthotopic liver transplant, and it represents more than 50% of all arterial complications. According to the onset, HAT can be defined as early (within the first month) or late (later than one month). The incidence of early HAT is approximately 4%–12% in adults and 42% in children, and it is a major cause of graft loss (53.1%) and mortality (33.3%) in the early postoperative period. Thrombosis occurring after the first month or late HAT has a more insidious clinical course and is less devastating. Other than surgical technical causes like small calibre vessels or size discrepancy in the donor and recipient vessels, stenotic anastomosis, kinking; several nonsurgical causes have been described for HAT, which include ABO blood type incompatibility, increased cold ischemic time of the donor liver, acute rejection and sluggish flow through the hepatic artery. Initially, symptoms, signs and abnormal laboratory values are absent in early HAT, whereas reduced or absence of flow within the hepatic artery can be readily picked up by bedside Ultrasound and Colour Doppler evaluation (Table 9.14.19). Ultrasound is diagnostic in 92% of cases of HAT. Timely diagnosis of HAT allows for therapeutic interventions to restore the hepatic artery patency by vascular intervention or surgical reexploration to salvage the graft; if these measures fail retransplantation is needed in most cases. On Doppler ultrasound, acute HAT is diagnosed by: Graft oedema, systemic hypotension, and high-grade hepatic artery stenosis (HAS) can lead to false-positive sonographic diagnosis of HAT; whereas, false-negative sonographic findings may occur in presence of periportal arterial collateral formation in chronic thrombosis. CT or MRI with standard angiographic protocol are usually performed to confirm the diagnosis, which will reveal a complete cut off of the hepatic artery after the anastomosis (Fig. 9.14.30). Arterial anatomical variations like replaced recipient hepatic artery or any technical modifications used during arterial anastomosis need to be noted while reporting the angiography, which can guide the interventional radiologist in a better way. In late stages of HAT, hepatic arterial collaterals can develop. This collateral vessel flow can cause a dampened (tardus parvus) hepatic arterial waveform with a prolonged acceleration time (AT), similar to that seen in significant HAS. Presence of tardus parvus waveforms in the distal hepatic arteries is an ominous sign and must not be considered evidence of patency of the hepatic artery; it rather indicates the presence of either proximal stenosis or thrombosis. HAS has reported incidence of 5%–11% in posttransplant patients and is the second most common postliver transplant vascular complication. It usually occurs within 3 months of transplantation and most commonly involves the site of anastomosis. Risk factors for HAS are similar to those for HAT with few additional causes including clamp injury and intimal trauma from a perfusion catheter. If left untreated or undiagnosed, it can progress to HAT (Fig. 9.14.32) and related complications including hepatic ischemia, biliary complications, sepsis and ultimately graft loss. Treatment options aim at reestablishing adequate blood flow at the earliest either with angiographic intervention involving balloon angioplasty or stenting or with surgery if the previous measures are unsuccessful.
Posttransplant imaging
Normal doppler findings in post liver transplant
Hepatic artery
PSV
Variable, usually between 40 and 120 cm/s
RI
0.55–0.80
SAT
<0.08 second
Portal vein
Hepatic veins
Post transplant complications
Vascular complications
Vascular Complications
Time of Presentation
Hepatic artery complications
Hepatic artery thrombosis
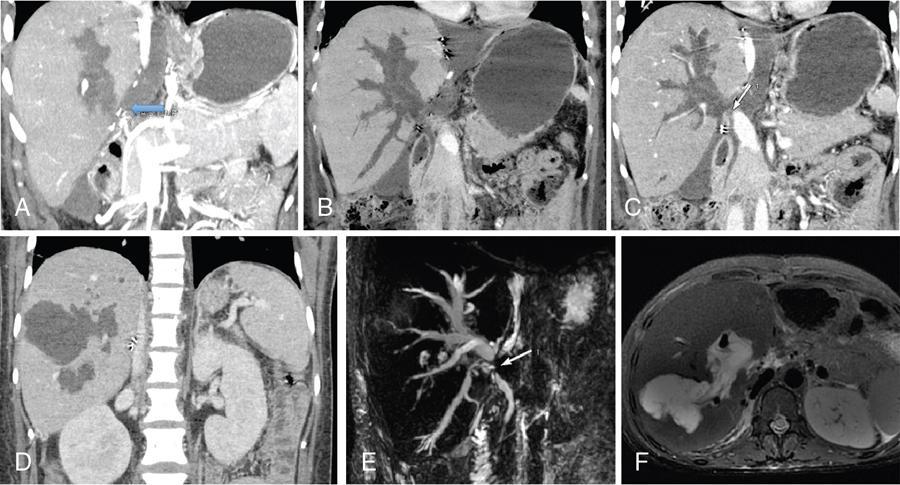
Hepatic artery stenosis
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








