Ritu K. Kashikar, Shrinivas B. Desai, Chandresh Karnavat, Nilesh Doctor The biliary tract is subject to a variety of abnormalities. The spectrum includes benign diseases of autoimmune, infective, ischaemic, infiltrative aetiologies and malignant disorder, which most importantly represents cholangiocarcinoma. Imaging in particular magnetic resonance cholangiopancreatography (MRCP) plays a vital role in diagnosis and follow-up of these disorders. Multidetector computed tomography (MDCT) and magnetic resonance imaging (MRI) are also crucial in preoperative staging of biliary malignancies and making decisions regarding resectability and extent of resection. This chapter focuses on important benign and malignant disorders of the biliary tree and imaging features that aid in differentiation of various entities. A variety of disorders affect the biliary tree. Most biliary disorders manifest as biliary dilatation. The various causes are listed in Table 9.15.1. Imaging plays an important role in identification of aetiology based on pattern of involvement in association with clinical picture. USG is often the first investigation in a patient with jaundice. USG is excellent at showing biliary dilatation. It may be useless in assessing level of obstruction and biliary stones. Changes in liver morphology, development of cirrhosis is accurately done on USG. Early changes in conditions like primary sclerosing cholangitis (PSC), presence of intrahepatic ductal strictures, thickening of common bile duct (CBD) are, however, not confidently seen and need imaging modalities like MRCP and computed tomography (CT). Staging of hilar cancers is also best done on CT or MRI with contrast. Contrast-enhanced CT is extremely accurate in staging hilar cancers. It is a preferred modality to access radial spread of the disease and vascular involvement. Subtle changes of PSC and autoimmune cholangitis may however be missed and MRCP is preferred in these conditions. The protocol for evaluating biliary disease is standard plain scan followed by early, late arterial, portal venous and parenchymal phases similar to that obtained for focal liver lesions. Delayed phase images should be obtained for hilar malignancies, which often show delayed enhancement. MRCP with or without contrast is the modality of choice in diagnosing and characterizing biliary diseases. Owing to its noninvasive nature, it has replaced endoscopic retrograde cholangiopancreatography (ERCP) in the initial evaluation and follow-up of a variety of biliary pathologies. Absence of radiation exposure makes it suitable to obtain follow-ups in patients requiring serial scanning. The ability to diagnose abnormalities of both intra- and extrahepatic biliary tree, level of obstruction, longitudinal and radial spread of neoplastic process make it an ideal modality. MRCP protocol includes T2 weighted single-shot fast spin-echo, T1 weighted in phase and opposed phase gradient echo, diffusion-weighted imaging, T2-weighted fat-suppressed fast spin-echo in axial and coronal. A pre contrast three-dimensional T1-weighted fat-suppressed spoiled gradient-echo image is obtained in addition to 3-D MRCP. Postcontrast protocol includes dynamic three-dimensional T1-weighted fat-suppressed spoiled gradient-echo (in arterial, late arterial and portal venous, parenchymal and delayed phases). Since the advent and widespread use of MRCP, the utility of ERCP in diagnosing biliary pathologies has significantly reduced. ERCP is primarily used when stenting or other procedures need to be performed in the same setting. Although an invasive procedure with postprocedural risk of pancreatitis ERCP allows excellent depiction of biliary changes in conditions like PSC, recurrent pyogenic cholangitis (RPC) and IgG4-related disorders. Endoscopic USG (EUS) is an excellent modality in diagnosing lower bile duct pathologies and also has the added advantage of obtaining a biopsy in the same setting. EUS is less invasive than ERCP and overall safer. PSC is a premalignant cholestatic liver disorder characterized by bile duct strictures secondary to bile duct inflammation and fibrosis. Cirrhosis of liver can develop secondary to this condition and patients are at a high risk for biliary and colonic cancers. PSC is a relatively rare disease, with an incidence of less than 50 per 100,000 patients though it varies in various location. It is diagnosed in young patients aged 30–40 years and is twice as common in men than in women. Similar to other autoimmune diseases, genetic susceptibility is likely to be instrumental in the development of PSC after exposure to a trigger. There is also a strong association between PSC and human leukocyte antigens (HLAs). Environmental factors such as childhood microbial exposure also play an important role in disease pathogenesis. A strong association of PSC with inflammatory bowel disease (IBD) is seen. There is a 100-fold increased risk of developing PSC among siblings. PSC can be asymptomatic but may present with cholestatic symptoms such as jaundice, pruritus, fatigue and right upper quadrant pain, steatorrhoea and episodes of acute bacterial cholangitis. There is marked elevation of alkaline phosphatase (ALP). A twofold to threefold increase in serum alanine and aspartate aminotransferase (AST) can be seen. Elevated bilirubin is usually seen in advanced disease, malignancy or those with choledocholithiasis. Various serum antibodies can also be elevated in PSC. These have been enlisted in Table 9.15.2. On liver biopsy, classic pathologic features of periductal concentric fibrosis or ‘onion skin’, around the affected ducts can be seen. This is however not a pathognomonic finding and is seen in less than 40% of biopsy specimens. Therefore, biopsy is not routinely used as a diagnostic tool for PSC and is reserved primarily for disease staging. MRI with MRCP best depicts the biliary changes in PSC and is the modality of choice. While CT and USG may show biliary dilatation and changes of cirrhosis in advanced cases, they fail to show early changes (Table 9.15.3). USG is an effective modality for the visualization of dilatation and diffuse wall thickening of the extrahepatic bile duct. Other feature seen on USG is bright echogenic portal triad. However, the role of USG in the diagnosis of early PSC is limited owing to suboptimal assessment of the intrahepatic biliary ducts. Features of advanced disease such as heterogeneous coarse echogenicity can be readily detected with USG. The major benefit of performing US is to guide liver biopsy. Also, liver stiffness as assessed by transient elastography correlated well with the degree of liver fibrosis in PSC patients. CT can demonstrate some findings suggestive of sclerosing cholangitis such as focal, discontinuous, often peripheral intrahepatic biliary duct dilatation and thickening with enhancement of the bile ducts owing to inflammation. CT is effective in helping exclude other causes that can result in biliary stasis and dilatation such as hepatic and pancreatic lesions. CT is also excellent in diagnosing tumours, which may sometimes have similar presentation. Concomitant changes of IBD, if present, can be seen. However, CT is limited in assessment of biliary strictures and visualization of disease involving small peripheral bile duct, especially in the early stages of the disease. CT shows changes of cirrhosis in liver and assessing changes of portal hypertension. MRCP is diagnostic imaging modality of choice in the workup of patients with suspected PSC, as recommended by both the AASLD and EASL guidelines. MRCP has high diagnostic sensitivity (86%) and specificity (94%) for the detection of PSC (Table 9.15.3). Multifocal short segmental strictures in the intra- or extrahepatic biliary tree with intervening normal or dilated duct leading to beaded appearance are seen in early disease. The location in strictures is usually at the biliary bifurcation and are disproportionate to upstream dilatation. Advanced cases show pruning of peripheral biliary radicals secondary to advancing fibrosis obliterating the smaller ducts. An obtuse angle between the central and peripheral ducts is suggestive of PSC. Diverticula and webs also can be seen, though not pathognomonic (Table 9.15.4) (Figs. 9.15.1–9.15.3). Strictures of the CBD of less than 1.5 mm and of left, right or common hepatic duct (CHD) of less than 1 mm are defined as dominant stricture. A dominant stricture is associated with worse prognosis in part due to development of cholangiocarcinoma. MRCP plays an important role in raising the suspicion and guiding treatment. A dominant stricture may be confused with hilar cholangiocarcinoma on imaging and it is often impossible to distinguish the two based on imaging alone (Table 9.15.4) (Fig. 9.15.4). Changes in liver morphology are seen in the form of distortion. The classical change described in PSC includes hypertrophy of the caudate lobe and atrophy of the left lateral and right posterior segments of the liver. Hypertrophy of caudate lobe is more frequent in PSC than cirrhosis from other aetiologies. Other parenchymal changes include heterogeneity of liver, periportal cuffing, peripheral inflammation, cirrhosis and features of portal hypertension. Hyperintensity of the liver parenchyma in PSC on nonenhanced T1-weighted images has been reported. Wedge-shaped peripheral atrophic areas of confluent hepatic fibrosis are seen as high T2-weighted signal intensity. Periportal oedema is visualized as high signal intensity in periportal region on T2-weighted. Increased heterogeneous peripheral enhancement of the liver parenchyma can be seen and is likely due to the altered blood supply in those areas in response to parenchymal inflammation (Table 9.15.4) (Figs. 9.15.5 and 9.15.6). MR elastography is the most accurate noninvasive method for the diagnosis and staging of liver fibrosis and could potentially replace liver biopsy. It is predictive of progression to decompensated liver disease. However, lack of wide availability are still major limiting factors. MR elastography is not influenced by obesity or anatomical conditions (e.g. narrow intercostal spaces and ascites) and the sample size of the liver is significantly higher and hence has several advantages over ultrasound elastography (Table 9.15.5). Besides these features, enlarged reactive abdominal lymph nodes, commonly periportal and portocaval lymph nodes, are commonly diagnosed in PSC and should not be misdiagnosed as a lymphoproliferative disorder or metastatic disease. Though ERCP has higher diagnostic accuracy in detecting PSC, its role is limited to intervention required in PSC due to its invasive nature and potential complications. Also it is recommended that MRCP be performed prior to ERCP as a preprocedural MRCP can provide a road map to the endoscopist. Small duct PSC is a variant PSC syndrome with biochemical markers and histologic features suggestive of PSC with normal cholangiography. It is associated with better prognosis. Approximately one-fourth of patients’ progress to classic PSC in an average of 8 years. Bile duct calculi are a common complication of PSC. Pigmented bile duct stones are common owing to biliary stasis. Both intrahepatic and extrahepatic biliary stones can be found. However, the presence of biliary tree stones is not an essential diagnostic feature. Soft calcific foci within dilated bile ducts can be seen on US and CT images. MRI with MRCP is the imaging modality of choice to detect bile duct stones and appear as focal areas of signal-intensity-void filling defects on T2-weighted images. Usually isointense at T1-weighted imaging but frequently are hyperintense. The presence of biliary strictures puts these patients at risk of bacterial cholangitis in PSC patients. The classic Charcot triad of fever, abdominal pain and jaundice can be seen. Early enhancement of the biliary wall due to the biliary duct inflammation is a common finding with peribiliary reactive hepatic parenchymal changes. Cholangitic abscesses may sometimes be seen. PSC patients are at high risk of developing cholangiocarcinoma during the disease course with and a total risk of 10%–15%. Approximately 30% of all cholangiocarcinomas are detected within the first year of establishing a diagnosis of PSC and so it is important to be suspicious even at the time of PSC diagnosis and to survey for early detection. Rapid clinical deterioration, worsening of jaundice, pruritus, weight loss along with elevation in serum bilirubin and ALP should raise suspicion of PSC. It may be difficult to distinguish benign from malignant strictures. Progressive bile duct wall thickening, irregularity, enhancement, new onset biliary dilatation, dominant stricture or development of focal strictures with dilatation and ipsilateral atrophy are indeterminate features that may raise possibility if occult cholangiocarcinomas. Perivascular thickening, vascular involvement or occlusion are features favouring malignancy. Definite diagnosis is however often not possible on imaging alone and a brush biopsy should be taken in patients with new onset clinical symptoms, elevated tumour makers or above-mentioned indeterminate imaging features (Table 9.15.7). The most common subtype of cholangiocarcinoma in PSC patients is periductal type, characterized by long segment irregular wall thickening along the bile duct with no identifiable mass. The lesion is hypointense on T1W1 images and hyperintense on T2W1 images showing progressive enhancement on contrast study. The tumour could be at the bifurcation and can cause abrupt cut-off, commonly presenting as Klatskin tumour (Fig. 9.15.7). Combined UC and PSC is associated with a higher risk for colorectal carcinoma than UC alone. PSC is considered as an independent risk factor for development of colorectal carcinoma in patients with UC. Malignancy tends to involve the right colon or more proximal parts of the colon. Clinical presentation and history in patients with ascending cholangitis differ from those with PSC. Fever, pain and jaundice dominate the presentation in patients with ascending cholangitis while patients with PSC may be asymptomatic. The typical findings of PSC including biliary duct stenosis, beading or pruning are not typical findings in the newly diagnosed cases of acute ascending cholangitis. Wedge-shaped peripheral and/or intrahepatic peribiliary foci of increased T2 signal around the dilated radicals with arterial and/or delayed parenchymal enhancement has been described in ascending cholangitis. Patients with RPC present with recurrent episodes of abdominal pain, fever, jaundice and chills. These features differ from those in patients with PSC. Hepatolithiasis with pigmented stones in the biliary tree and upstream and downstream duct dilatation are typical imaging features in RPC (Table 9.15.8). Ischaemic cholangiopathy is a form of biliary injury resulting from decreased arterial supply. History of liver transplantation is the key in diagnosing ischaemic cholangiopathy. Biliary strictures in AIDS cholangiopathy are indistinguishable from PSC. However, the combination of papillary stenosis and intrahepatic ductal strictures appears relatively unique to AIDS cholangiopathy; this combination is not found in PSC. Clinical history may help to distinguish one from the other. Effective medical therapy for PSC is lacking. Oral ursodiol (ursodeoxycholic acid) is one of the main treatment options of cholestatic diseases though does not prevent disease progression. Azathioprine and steroids are recommended for use in patients with AIH as well as those with AIH–PSC overlap syndrome. Vedolizumab is a gut-specific monoclonal antibody that has been tried but the clinical utility in PSC–IBD patients remains under investigation. Dilatation of biliary strictures, stenting, lithotripsy and stone extraction can be done with the aid of ERCP and percutaneous transhepatic cholangiography (PTC). The only definitive cure of PSC is, however, liver transplantation. Treatment of complications like choledocholithiasis stone removal can be accomplished by using standard endoscopic techniques with or without sphincterotomy, with balloon or basket extraction for extrahepatic biliary stones. For bacterial cholangitis, immediate administration of broad-spectrum antibiotic therapy is recommended and in abscess formation percutaneous drainage with intravenous antibiotic therapy shows favourable outcome compared with surgical drainage. Incidence is 20%–25%. Diagnosis of recurrent PSC requires careful evaluation, as there are other causes of biliary changes after liver transplant with similar features. Nonanastomotic biliary strictures imply the diagnosis of recurrent PSC only if they occur more than 90 days after transplant. Characteristic multifocal strictures and segmental dilatations of biliary ducts are suggestive findings. MRCP is the initial modality. RPC is characterized by a triad of recurrent biliary sepsis, inflammatory biliary strictures and intrahepatic stones. Fifty per cent of patients may have acute pancreatitis. Oriental cholangiohepatitis, primary hepatolithiasis, Hong Kong disease and oriental infestational cholangitis. Prevalence in the third and fourth decades of life with equal frequency among men and women peak. Southeast Asia predominantly in rural population and in lower socioeconomic groups. Gut-derived organisms lead to sepsis, which initiates a cascade of events, which leads to a chronic, progressive and recurrent inflammatory process in cholangioles. Associations with Clonorchis sinensis, Opisthorchis species, Fasciola hepatica and Ascaris lumbricoides infestations have been suggested. Common organisms cultured from bile include Escherichia coli, Klebsiella, Pseudomonas and Proteus species and anaerobes. Structural biliary abnormalities may develop before stones are demonstrable. Strictures may be seen at cholangiography without stones and vice versa. Entrapped stones cause stasis, sepsis, scarring and stricturing with the increasing lithogenicity (Table 9.15.9). Typically present with abdominal pain, fever and jaundice (the Charcot triad) and commonly provide history of repeated episodes. Routine investigations may demonstrate leukocytosis, deranged liver enzymes with obstructive pattern. Elevated cholestatic markers (bilirubin, ALP and gamma GT) and deranged LFTs can be seen. Serum inflammatory markers can be elevated. Additional tests aiding in differential diagnosis include autoantibodies, ANCA, CA19-9 and serologic tests for Echinococcus. Histology is usually not required. It shows fibrous mural thickening of bile ducts and periductal tissue, as well as acute and chronic inflammatory changes. The imaging features include changes in bile ducts, development of calculi and changes in the parenchyma. The disease affects intrahepatic ducts more than the CHD and CBD. Disproportional dilatation of central intrahepatic ducts and extrahepatic bile ducts is seen with nondilated or minimally dilated peripheral ducts, leading to abrupt tapering of bile ducts. All segments of biliary tree may be involved, but the lateral segment of the left lobe is most often and extensively involved most likely because left hepatic ducts come off at a more acute angle compared with the right hepatic ducts, thus predisposing to stasis and stricture formation (Table 9.15.10). Hepatolithiasis is common in patients with RPC. Stones are composed mainly of bile pigments with variable calcification. There may be single or multiple stones scattered in the intra- or extrahepatic ducts or both. The dilatation of the extrahepatic duct is generally not related to the location of the stone. Ducts both proximal and distal to the stone are dilated. Parenchymal atrophy most commonly involves the left lateral and right posterior segments while hypertrophy of the caudate and right lobe is seen. USG shows dilatation of the central intrahepatic and extrahepatic ducts, with relative sparing of the peripheral biliary tree. Hepatolithiasis can be seen in 90% of cases. The echogenicity and acoustic shadowing of calculi may vary depending on extent of calcification. There is often associated periportal echogenicity. Ultrasound may be useful in performing image-guided percutaneous drainage of abscesses or biopsy of suspicious lesions. Limitations include inability to detect subtle intrahepatic ductal dilatation and heavy stone burden obscuring evaluation of underlying hepatic parenchyma. Contrast-enhanced CT is increasingly being used as first line of imaging. It allows for detection of characteristic disproportionate dilatation of the extrahepatic and central intrahepatic ducts. Contrast-enhanced CT also allows for detection of bile duct wall enhancement, suggestive of acute cholangitis. Ninety per cent stones are hyperdense to liver parenchyma on nonenhanced scan. Extent of calcification in calculi may vary. Parenchymal atrophy affects left lateral segment most frequently, followed by right posterior segments. Eventually, changes of cirrhosis can be seen. Heterogeneous appearance of liver parenchyma with segmental steatosis and altered enhancement can be seen particularly during acute attack. Pneumobilia is not infrequent and can usually be attributed to recent procedures or surgeries like bilioenteric anastomosis. It may, however, be seen in patients without history of prior interventions due to recent passage of stone through the ampulla or less commonly cholangitis related to gas forming organisms (Fig. 9.15.8). Subtle intrahepatic ductal strictures and noncalcified stones may be missed on CT. MRCP allows visualization of both intra- and extraductal disease and can reveal complete extent, severity and complications of the disease. Even noncalcified calculi which may be missed on CT or sonography, are seen as intraductal filling defects on heavily T2-weighted images and may appear hyperintense to the liver on T1-weighted images. MRI demonstrates central and extrahepatic duct dilatation with decreased arborization and abrupt tapering of peripheral ducts. MRI particularly MRCP sequences accurately depicts stenotic segments and delineates the entire biliary tree, including the proximal part of the stricture, without risk of aggravating biliary sepsis. Even short segment duct strictures <1 cm are easily seen on MRCP (Figs. 9.15.8 and 9.15.9). Parenchymal abnormalities like hepatic atrophy, whether diffuse or segmental, and altered signal can be accurately diagnosed. Mass lesions and hepatic abscesses, if present, are also well delineated. The characteristic finding of disproportionate dilatation of the ducts, with multiple intraductal calculi are well demonstrated. There may also be intrahepatic strictures, with abrupt tapering of the peripheral ducts as well as decreased arborization of the biliary tree (arrowhead appearance). It allows for better spatial resolution, thus permitting better evaluation of the smaller peripheral ducts. The main limitation of ERCP is its invasive nature, with complications such as ERCP-associated pancreatitis. Heterogeneous parenchymal enhancement, wall thickening and periductal enhancement are seen in acute cholangitis. Abscess formation is encountered in up to 20% of RPC patients who undergo cross-sectional imaging. The abscesses can occur in both the affected and unaffected portions of the liver but most commonly seen in the right lobe. Sonography guided aspiration can be done when diagnosis is doubtful. Intrahepatic bile lakes are not infrequent and may or may not communicate with the biliary tree. On USG bilomas appear anechoic and are usually hypodense on CT with or without calculi. Leakage of bile from a severely dilated obstructed duct may lead to formation of extrahepatic biloma. Cholangiocarcinoma is the most feared complication and may be seen in up to 5% of patients. Segments with high stone burden or those with atrophy are more prone to developing cholangiocarcinoma. Clinical and laboratory indicators for development of cholangiocarcinoma in patients with PSC include increase in jaundice without associated cholangitis, sudden weight loss, significant elevation of ALP and rising tumour markers. Peripheral cholangiocarcinoma manifests as expansion of the affected segment. Hypoattenuating mass with showing peripheral enhancement causing narrowing of the portal vein may raise suspicion of malignancy. Portal vein thrombosis has also been reported but is rare. Recurrent biliary sepsis may lead to periductal inflammation and portal thrombophlebitis. Portal thrombosis can lead to lobar atrophy with compensatory hypertrophy of the uninvolved lobe. Higher incidence of hepatocellular carcinoma (HCC) is reported in patients with RPC due to development of cirrhosis in severe cases. Management should comprise antibiotic treatment, clearance of stones, maintenance of biliary drainage and long-term follow-ups. ERCP allows for therapeutic procedures such as stone removal and stenting of strictures. Destroyed liver segments, those with multiple abscesses and patients with secondary cholangiocarcinoma are candidates or surgery. Stone extraction can also be done surgically with the aid of basket, forceps, balloon catheter or flexible choledochoscope. Transduodenal sphincteroplasty and hepaticojejunostomy are commonly performed drainage procedures. Indications for transplantation include extensive bilobar hepatolithiasis, cirrhosis and liver failure. IgG4 cholangiopathy is a cholangitis characterized by elevated serum IgG4 and infiltration of the bile duct wall with IgG4 positive plasma cells leading to storiform fibrosis and obliterative phlebitis with resultant bile duct wall thickening. This condition shows good response to steroids. Frequent association with IgG4-related autoimmune pancreatitis is seen. IgG4 sclerosing cholangitis (IgG4-SC) has a threefold to fivefold higher prevalence in men than women. It usually presents in the fifth and sixth decades of life. Incidence of this disease is approximated to be around 0.28–1.08/100,000. Autoimmunity has been considered as the most probable pathogenesis of IgG4-related disease (IgG4-RD). Several HLA association have identified as determinants of disease susceptibility. The major histologic features associated with IgG4-RD have been well described and include the steps. Chronic or recurrent cholangitis is the most common presentation and seen in up to 75% cases. Other symptoms seen are fever, pruritus, pain and weight loss. Biliary involvement may be detected incidentally in patients being scanned for symptoms related to involvement of other organs such as pancreas. One-quarter of patients with IgG4-SC may be asymptomatic. Serum liver tests show a cholestatic pattern with often marked elevation of ALP and γ-GT and only mildly increased ALT and AST. Serum bilirubin may be increased. Tumour marker CA 19-9 is not able to distinguish pancreatobiliary malignancies from IgG4-SC since levels of >1000 IU/mL (ULN = 37 IU/mL) may be observed in IgG4-SC. IgG4 levels are elevated (>140 mg/dL) in up to 75%–80% of affected patients. A cut-off level of 207 mg/dL might be useful for completely distinguishing IgG4-SC from cholangiocarcinoma. Elevated bile fluid IgG4 has a high sensitivity and specificity of 100% at a cut-off level of 113 mg/dL and seen only in cases of IgG4-RD, neither PSC nor cholangiocarcinoma. Approximately 40% of patients have peripheral blood eosinophilia, often accompanied by asthma and atopy. Patients may show hypergammaglobulinemia, elevated serum IgE. Several diagnostic criteria have been developed to aid in the diagnosis of IgG4-SC. The most widely used is the HISORt (Histology, Imaging, Serology, other Organ involvement and Response to therapy) criteria of the Mayo Clinic. This tool was initially used to diagnose AIP but can be adapted to diagnose IgG4-SC by Ghazele et al. (Tables 9.15.13 and 9.15.14). It is often the initial investigation and may be normal early in the course of the disease. Circumferential thickening of the bile ducts with biliary dilatation is seen later in the disease. US also helps in the identification of associated findings affecting other organs. However, US has low sensitivity in detecting the disease and is suboptimal for assessing the extent of the disease. Contrast-enhanced CT though not the investigation of choice shows bile duct wall thickening with proximal mild dilatation. The intrapancreatic bile duct segment is more commonly involved. The most important finding is circumferential symmetric wall thickening of the bile ducts, frequently involving the extrahepatic segments, with smooth outer and inner margins. In addition, the thickened segment shows progressive homogeneous contrast enhancement, further increasing in the delayed phase. As opposed to malignancies, wall thickening or stricturing of bile ducts in IgG4-RD is not associated with proportionate proximal biliary dilatation (Figs. 9.15.10 and 9.15.11). In addition to the above findings, imaging findings of IgG4 disease affecting other organs, that is, gallbladder wall thickening, changes of autoimmune pancreatitis, retroperitoneal fibrosis and kidney findings can be seen. MRI is the investigation of choice. MRI findings are similar to CT findings and include circular and symmetric long segmental thickening of the bile duct wall with smooth outer and inner margins. Bile duct wall thickening can be seen in nonstenotic areas as well The other findings are the visibility of patent bile duct in the strictures, disproportionate proximal dilatation, hyperenhancement during the late arterial phase, homogeneous hyperenhancement during the delayed phase, concurrent gallbladder wall thickening and no vascular invasion (Table 9.15.15).
9.15: Imaging in biliary diseases
Introduction
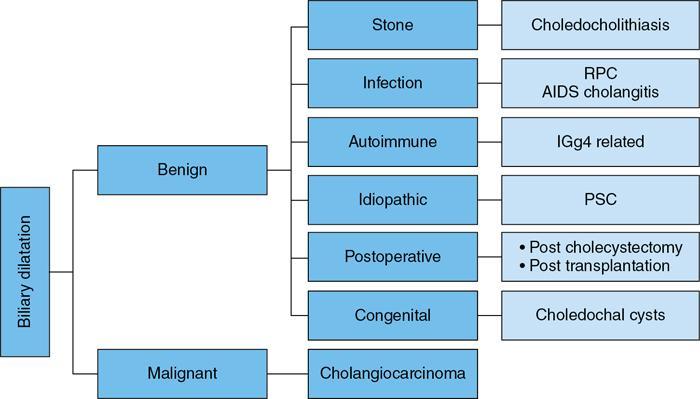
Causes of biliary dilatation
Imaging techniques
USG
CT
MRCP
ERCP
Endoscopic USG
Primary sclerosing cholangitis
Introduction
Incidence
Age group
Pathogenesis
Clinical features
Serological markers
Serum Antibodies in PSC
Approximate Incidence
Antinuclear antibodies (ANA)
75%
Antismooth muscle antibodies (ASMA)
75%
Perinuclear antineutrophil cytoplasmic antibody
30%–80%
Anti-p40 autoantibody
30%–80%
Proteinase-3 antineutrophil cytoplasmic antibody (PR3-ANCA)
38%–40%
Histology
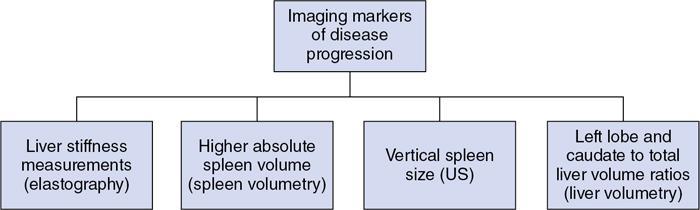
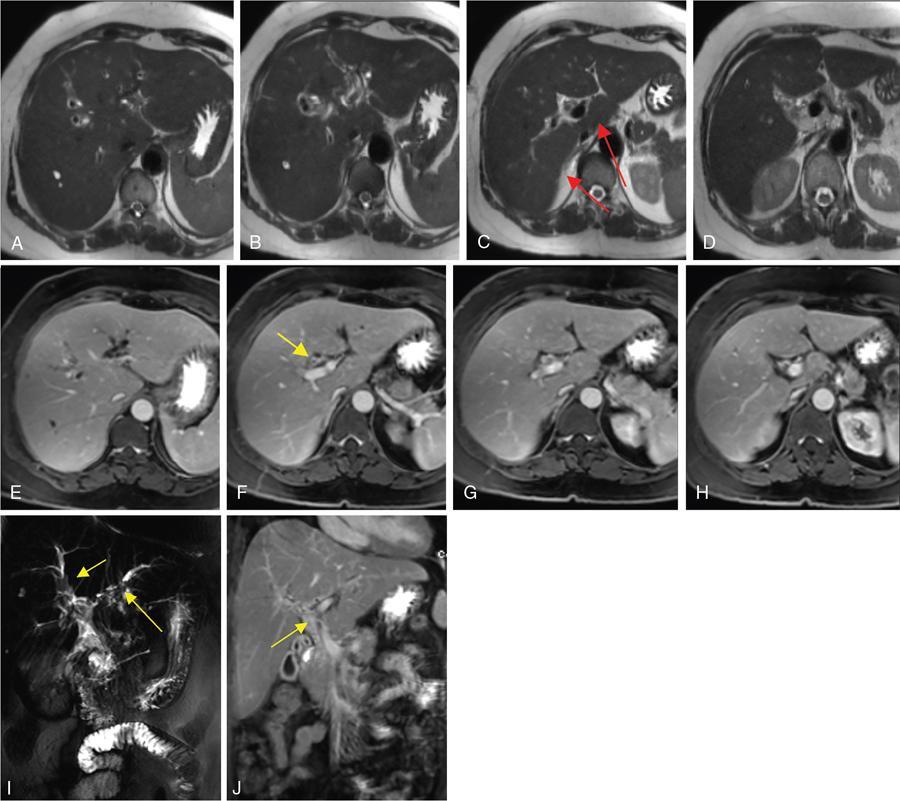
Imaging features
US
CT
MRCP and MRI
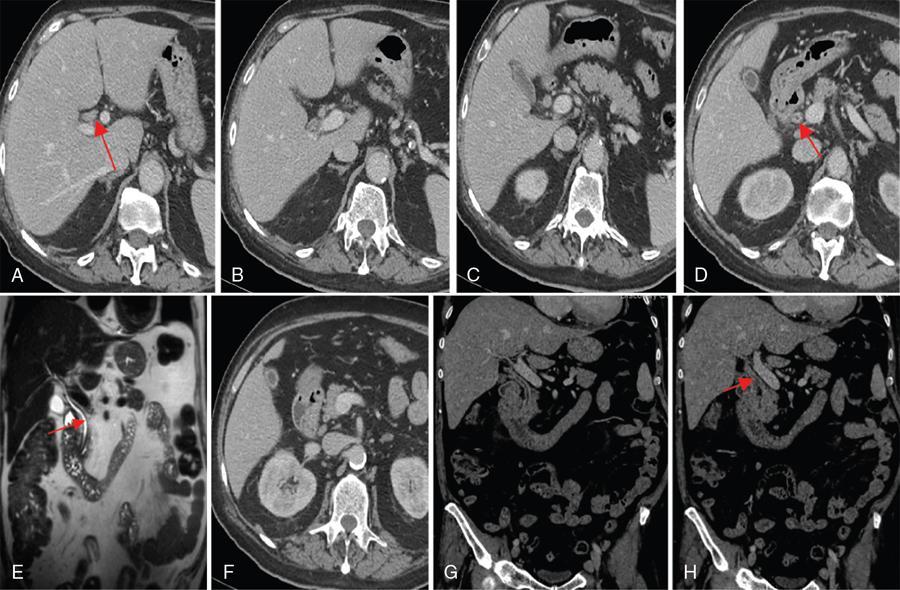
1. Biliary changes in PSC
A. Intrahepatic biliary strictures.
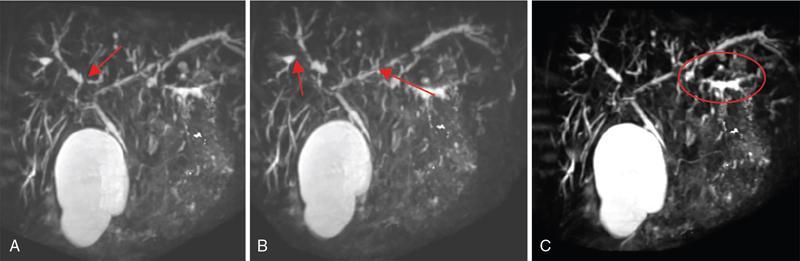
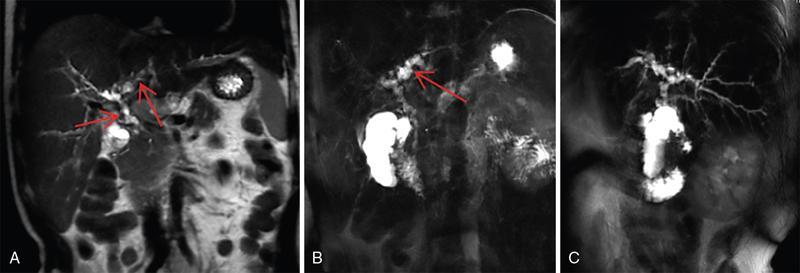
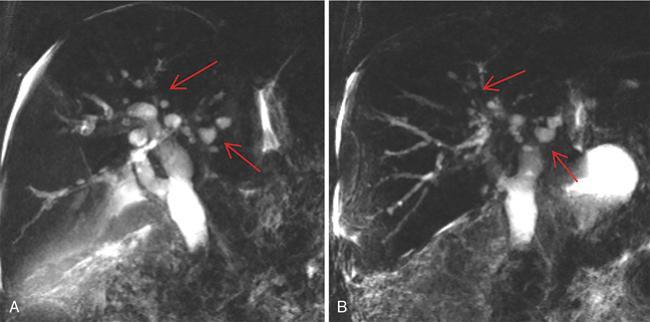
B. Dominant stricture.
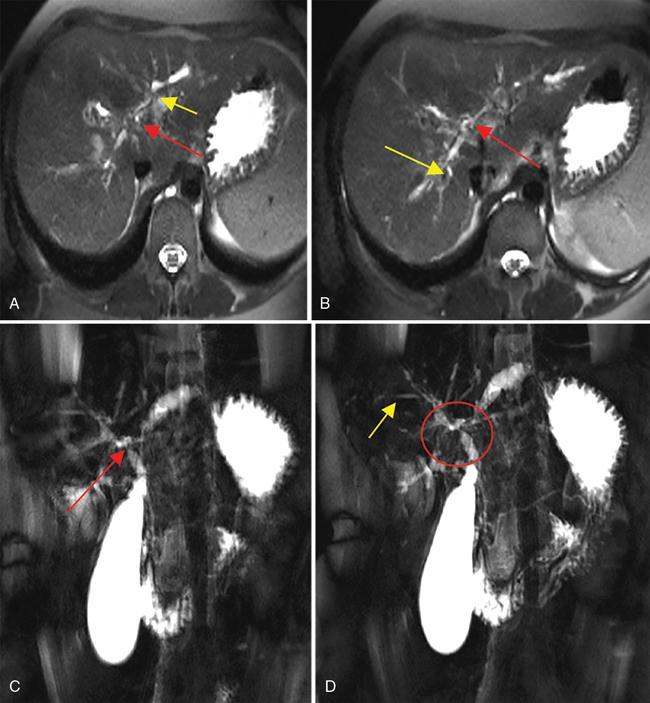
2. Liver parenchymal changes
Complications (Table 9.15.6)
Biliary stones
Bacterial cholangitis
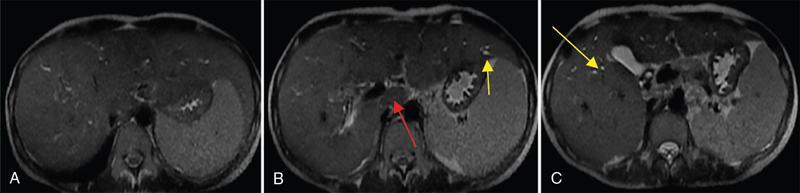
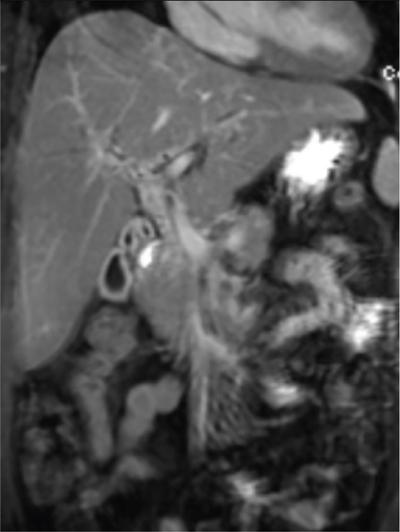
Cholangiocarcinoma
Indirect Signs
Direct Signs
Rapid clinical deterioration
Obvious mass
Elevated tumour markers
Progressive bile duct wall thickening, irregularity or enhancement
Hilar cut-off
New or progressive biliary dilation
Long segment stricture
Dominant stricture
Perivascular thickening
Obliteration of branches of the portal vein or hepatic artery
Colorectal cancer
Differential diagnosis
Acute ascending cholangitis
Recurrent pyogenic cholangitis
Ischaemic cholangiopathy
AIDS cholangitis
Management
Medical therapy
Posttransplant recurrence
Recurrent pyogenic cholangitis
Synonyms
Age and sex
Geographical location
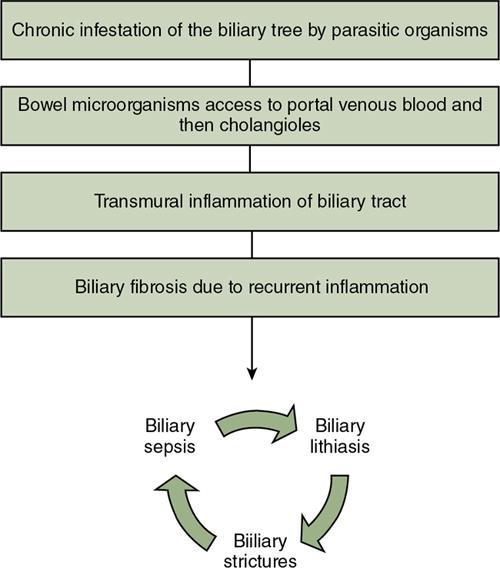
Aetiopathogenesis
Clinical features
Histology
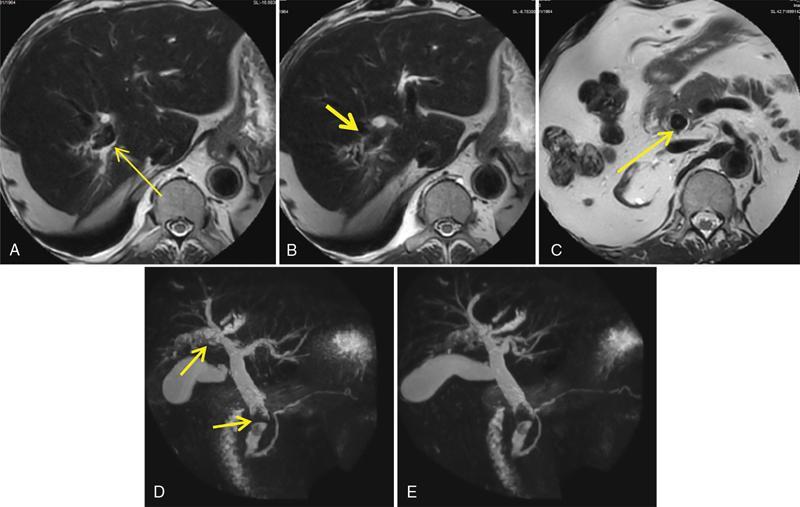
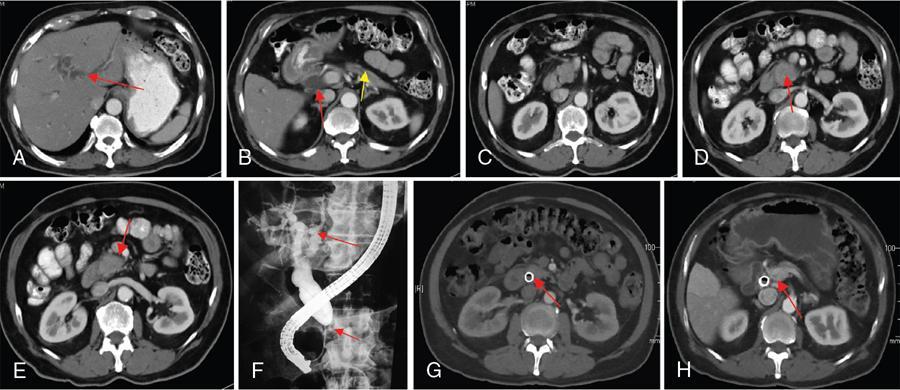
Imaging features
Biliary ductal changes
Biliary calculi
Liver parenchymal changes
US
CT
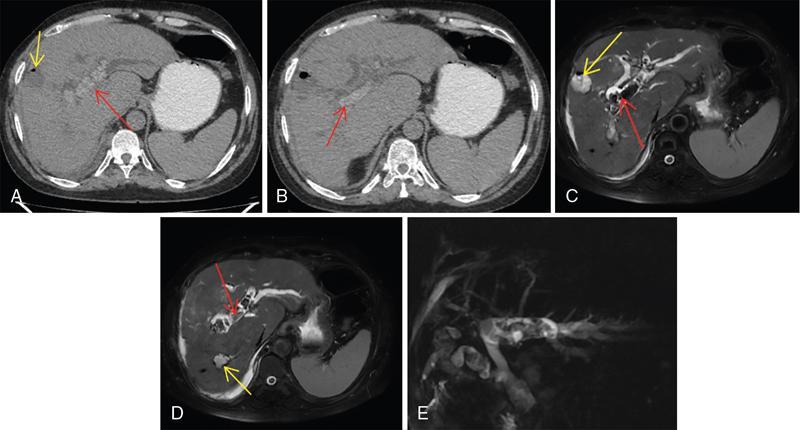
MRCP
ERCP
Complications (Table 9.15.11)
Acute cholangitis
Abscess
Biloma
Cholangiocarcinomas
Portal vein thrombosis
Hepatocellular carcinoma
Differential diagnosis
Management
Definitive surgery
Biliary drainage procedures
Liver transplantation
IgG4-related sclerosing cholangitis
Demographics
Pathogenesis
Histology
Clinical features
Laboratory findings
Diagnostic criteria
Imaging features
US
CT
MRI
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree