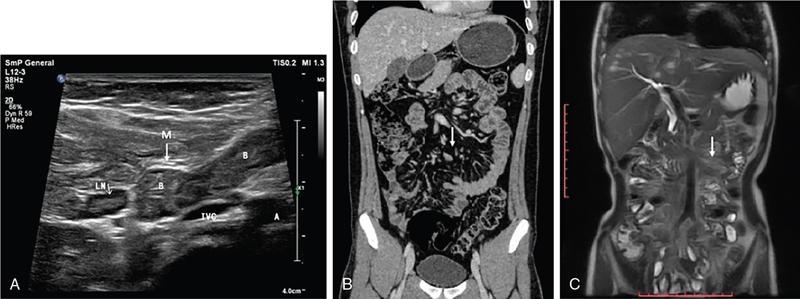
Neera Kohli, Anit Parihar Objectives: To provide the readers with a comprehensive, simplified, lucid, well-illustrated and well-presented latest account of the anatomy, functions, and childhood diseases of the mesentery. Key terms: Mesentery, anatomy, functions, infections, inflammation, neoplasia, cysts, malrotation, vasculitis, textiloma, hernias. Learning objectives: To approach a clinical condition of the mesentery in a logical and comprehensive manner so as to help the patient maximally. Increasing number of researchers, anatomists, radiologists, treating physicians and surgeons are now considering the mesentery as a separate organ. Serious attempt has been made by the authors to stick to the new theory while choosing and describing the various paediatric diseases to be included in this chapter. Likewise, each condition has been described as inclusively as possible. Mesentery develops from the splanchnic mesoderm. It has two parts: a ventral part that forms the mesentery of the abdominal part of oesophagus, the lesser omentum and mesentery of the first part and proximal half of the second part of the duodenum. The other is the dorsal part that runs below the diaphragm, from the oesophagogastric junction to the anorectal junction forming the mesentery of rest of the gastrointestinal tract (GI) including the greater omentum. It consists of fat, and has blood vessels, nerves and lymphatics running that supply the GI. Interest in the mesentery was reactivated when Heald in 1988 demonstrated that tumour recurrence following rectal cancer surgery was dramatically decreased if the mesorectum was removed. Heald’s work was the true start of mesenteric-based surgery. Hohenberger in 2009, focused on the mesocolic region, and showed that intestinal tumour recurrence was minimized, if the mesocolon was surgically excised intact. Heald and Hohenberger thus prompted a renaissance in the study of the mesentery and abdominal anatomy. Following their findings, research was done which demonstrated that the mesentery below the duodenum is continuous, and that the large and small intestine is centrally connected to the rest of the body, by the mesentery. Mesentery originates or has root posteriorly, at the level of origin of the superior mesenteric artery (SMA), as these findings clarified its shape, they marked the start of the scientific study of the mesentery and led to the proposal that it be reclassified an organ. Subsequent research showed that abdominal digestive organs, that is, liver, spleen, intestine and pancreas develop either on or in it. This relationship is retained into adulthood, so abdominal digestive organs are directly connected to the mesentery or we can say that they are ‘mesenteric’ in embryological origin and anatomical position (Fig. 7.10.1). Accordingly, all abdominal and pelvic organs belong either to a mesenteric or a non-mesenteric domain. This model explains the positional anatomy of all abdominal digestive organs and associated vasculature. Moreover, it explains the peritoneal landscape and enables differentiation of peritoneum from the mesentery. Hence, the mesentery should be subjected to the same investigatory focus that is applied to other organs and systems. One should be well versed with the normal appearances of mesentery on USG, CT and MRI (Fig. 7.10.2 A–C). Wherever mesentery is attached, a fascia (Toldt’s fascia) runs between the mesentery and the abdominal wall. This mechanism of anchorage is important. Second mechanism of anchorage involves the peritoneum which lines the abdominal wall and is reflected onto viscera as the ‘peritoneal reflection’. The mesentery, Toldt’s fascia, and peritoneal reflection are continuous. The third mechanism of anchoring is the vascular pedicle. Further mesenteries may be divided into true and specialized mesenteries. True mesenteries connect abdominal organs to the posterior wall of the abdominal cavity. These are the small bowel mesentery, the transverse mesocolon, and the sigmoid mesentery. Rest are specialized mesenteries. They do not attach organs to the posterior wall of the abdomen. The mesenteric organ is responsible for the development of systemic inflammation, such as the systemic inflammatory response syndrome (SIRS) and multiple organ dysfunction syndromes (MODS). Proinflammatory cytokines are produced in the intestine and mesentery and are then systemically distributed via mesenteric lymph. These contribute to a systemic inflammatory response, for example, post injury. The mesentery comprises a connective tissue lattice within which adipocytes are present in large numbers. Due to production of cytokines like C-reactive proteins (CRP), resistin, leptin and adiponectin by the mesentery it is considered responsible for glycemic/lipidemic disorders. Mesentery is a big source of secretion of CRP as it contains the largest part of visceral fat hence it is being implicated in diabetes and metabolic syndrome, suggesting that mesenteric adipocytes play a role in the pathobiology of these disorders. Further, visceral tissue plays an important role in immune regulation and may function as a proinflammatory agent. In mesenteric inflammation, both tissue and circulating fibrocyte levels are elevated and are associated with autoimmunity (e.g. systemic scleroderma, rheumatoid arthritis, Grave’s disease), inflammation, and fibrosis (idiopathic pulmonary fibrosis). These can differentiate into adipocytes or fibroblasts and are associated with autoimmunity in conditions such as: systemic scleroderma, rheumatoid arthritis, Grave’s disease and idiopathic pulmonary fibrosis. Fibrocytes also have the ability to act as proinflammatory cells by secreting cytokines. Although currently neutrophils, lymphocytes, adipocytes, and fibrocytes are being studied in details, it is quite possible that there are other varieties of cells involved in mesenteric inflammation. ANML, also known as primary mesenteric lymphadenitis, is a self-limiting inflammatory disease of the mesenteric lymph nodes of right lower quadrant. Clinically it closely resembles acute appendicitis (AA). It occurs in children, adolescents and young adults. CRP and leukocyte count do not help much in differentiating these two conditions. Ultrasonography is the investigation of choice for diagnosing ANML. If three or more mesenteric lymph nodes with a short-axis diameter of 8 mm or more without any associated inflammatory process is identified then ANML is a strong possibility (Fig. 7.10.5A and B). CT if needed, is usually reserved for older patients. Radiologists should try to visualize the appendix as well. If the appendix is normal then it favours diagnosis of ANML. Occasionally, very large mesenteric lymph nodes may result in vascular compromise leading to ischemic colitis. Secondary mesenteric lymphadenitis is associated with some detectable intraabdominal inflammatory process. ANML is more common than AA in the first decade of life. AA becomes more frequent in the second decade and is distinctly uncommon after the age of 20. Boys are more commonly affected. Common symptoms are: fever, vomiting, change in consistency and frequency of stools and mild pain to severe colic. The pain mimics that of AA as it is felt in the right iliac fossa as well as in the periumbilical area. Although, this condition is referred to as nonspecific yet causative micro-organisms have been identified. These are: Several viruses, Yersinia enterocolitica, Helicobacter jejuni, Campylobacter jejuni, Salmonella spp., Shigella spp. and Mycobacterium tuberculosis. Yersinia enterocolitica is considered the most common pathogen in temperate regions. It has been observed that, when ANML or appendicitis occurs in childhood or adolescence, there is a significantly reduced risk of ulcerative colitis later in life. CD is a chronic inflammatory disease that affects the GI and adjacent mesentery in the paediatric and adult populations. Its prevalence is increasing worldwide. Mesentery of patients with CD has more fat tissue. These fat cells can produce proteins such as CRP which is associated with inflammation. This may be the cause of increased inflammation and bacterial invasion. Imaging studies have very vividly shown mesenteric and intestinal involvement in CD. Following are the findings noted in CD of the mesentery: Increased vascularity, oedema, lymphadenopathy, proliferation of the fibrous and fatty tissue, and increased density of fat. Besides these, stenosis and sacculations in the mesentery are also seen. Computed tomography (CT), magnetic resonance (MR) enterography, and strain elastography findings are demonstrated equally well in the mesentery and contiguous intestine. CT enterography is a better modality for assessing mesenteric abnormalities. Sakurai et al. have demonstrated radiological evidence of mesenteric hypervascularity (‘comb sign’) and nodal enlargement correlated with endoscopic evidence of mucosal ulceration. Gale et al. compared MR and CT enterography findings in 84 children and adolescents having active CD with radiological findings in their mesentery. They concluded that mesenteric hypervascularity, oedema, fibrofatty proliferation and lymphadenopathy correlated well with active mucosal disease. Mesenteric vascular diseases like arterial emboli, venous thrombi, and venous congestion have been reported in CD. These may be complicated by bowel ischemia and perforation. Lymphoma may arise in the mesentery in CD, without involvement of other organs, so neoplasia must be considered as a differential of a mesenteric mass arising in CD. It was first reported as retractile SM in 1924. SM is a rare, benign idiopathic, chronic, fibroinflammatory disease of the small bowel mesentery. It is of three types: mesenteric lipodystrophy, mesenteric panniculitis and retractile mesenteritis. Prevalence of mesenteric panniculitis is 0.6%. All three have distinct radiographic features, however, there is considerable overlap. Mesenteric panniculitis (MP) is a localized, chronic inflammatory process, seldom advancing to become fibrosis which may be florid. SM on sonography is seen as a hyperechoic, vessel encasing mass. This mass can also cause adjacent bowel displacement. This same feature is seen as halo sign on CT but without occlusion. CT can also show significant lymphadenopathy, bowel displacement and increase in the enhancement of surrounding fat labelled as ‘misty mesentery’, which is characteristic of MP. These findings are useful in differentiating SM from lymphomas and desmoids. The soft tissue nodules and calcifications in the vicinity are encased by dense tissue and fat leading to formation of a ‘pseudocapsule’. Though ‘Halo sign’ and ‘pseudocapsule’ are dependable signs yet they do not qualify to be called pathognomonic for SM. On the other hand, MRI has a higher sensitivity than CT for detecting SM. On MRI, mesenteric panniculitis and scattered lymph nodes are hyperintense on T2WI. In contrast, retractile mesenteritis has numerous strands of soft tissue, due to fibrosis with low signal on T1WI and T2WI MRI. Tuberculosis of mesenteric lymph nodes, along with peritoneal tuberculosis is seen in 45%–80% of children with an abdominal disease. Mesenteric lymph nodes involvement is seen frequently. Lymph nodes can get involved through lymphatic drainage of infected organs, retrograde lymphatic spread from intrathoracic disease, or due to hematogenous spread (Fig. 7.10.6). Tuberculosis of intraabdominal lymph nodes may occur without involvement of the intestine and peritoneum (Fig. 7.10.7). In such cases, it is associated with enlarged extra-abdominal superficial lymph nodes. The diseased involved lymph nodes may regress over time with or without calcification, can form inflammatory masses with possibility of abscess formation later on. Fistulae and ascites may also be seen. Ultrasound is the investigation of choice. Generally, lymph nodes show a central hypoechoic area due to central liquefaction; they can form conglomerates and infiltrate the omental fat, forming omental cake. Ultrasound also detects ascites very well. Ascitic fluid may be clear or contain fibrin strands, loculations and debris. The differential diagnosis of ascitic septations includes lymphoma, other carcinomas, and pyogenic peritonitis. Echogenicity of mesentery is increased in mesenteric tuberculosis. CT has a greater sensitivity than ultrasound in detecting mesenteric lymphadenopathy. Presence of lymph nodes with calcifications or with the typical low-density centres due to liquefaction and a peripheral ring enhancement favours tuberculosis. Solid organs like liver and spleen can have calcification. CT can easily detect inflammatory masses consisting of lymph nodes, omentum and bowel (called omental cakes). In children, MRI has the usual advantage of the fact that there is no ionizing radiation. However, sedation may be required due to a long study time. MRI findings of TB lymphadenitis display a predominant peripheral rim enhancement with or without a multilocular appearance, with a central non-enhancing lesion corresponding to caseation or liquefactive necrosis. Ascitic fluid is seen very well as a T2WI hyperintensity. Radiograph of the chest is a supportive investigation as 23% patients of proven pulmonary tuberculosis also have abdominal disease.
7.10: Imaging in paediatric mesentery disorders
Introduction
Brief anatomy, parts and functions
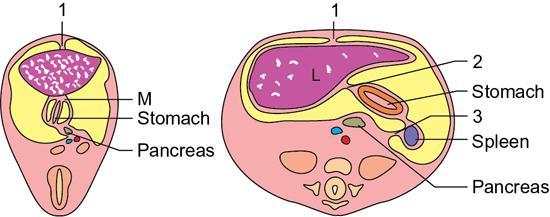
Parts of mesentery
Functions of mesentery
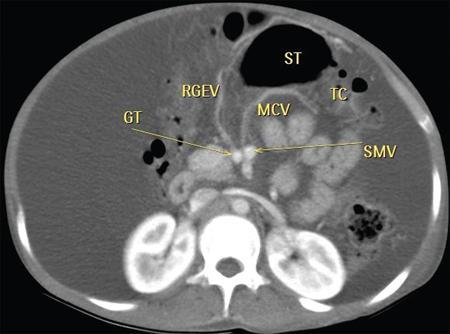
Diseases of mesentery in children
Infective and inflammatory disorders
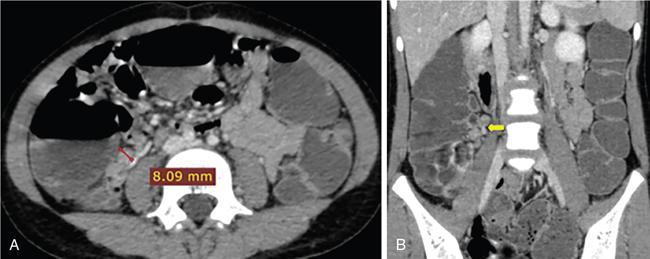
Acute nonspecific mesenteric lymphadenitis (ANML)
Crohn’s disease (CD)
Sclerosing mesenteritis (SM)
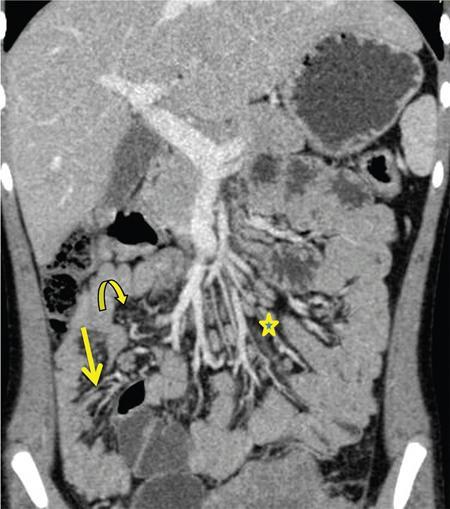
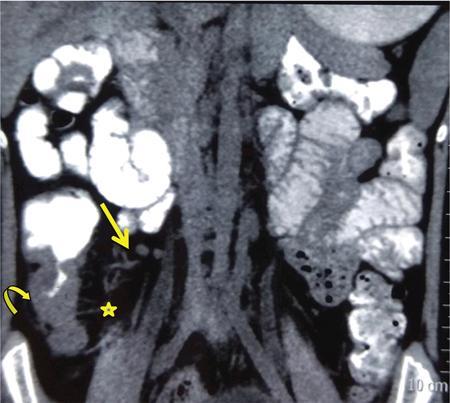
Mesenteric tuberculosis
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree