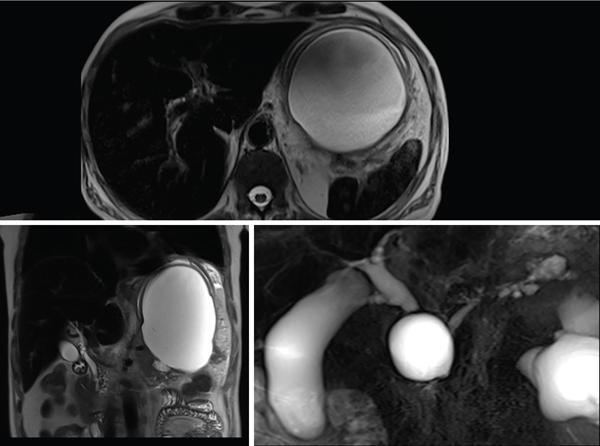
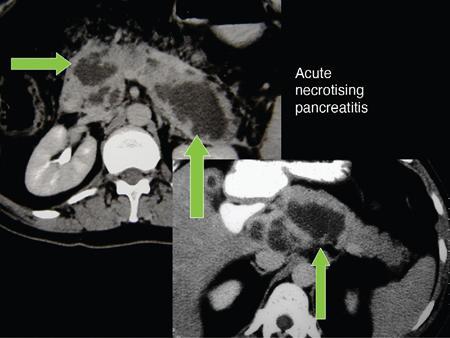
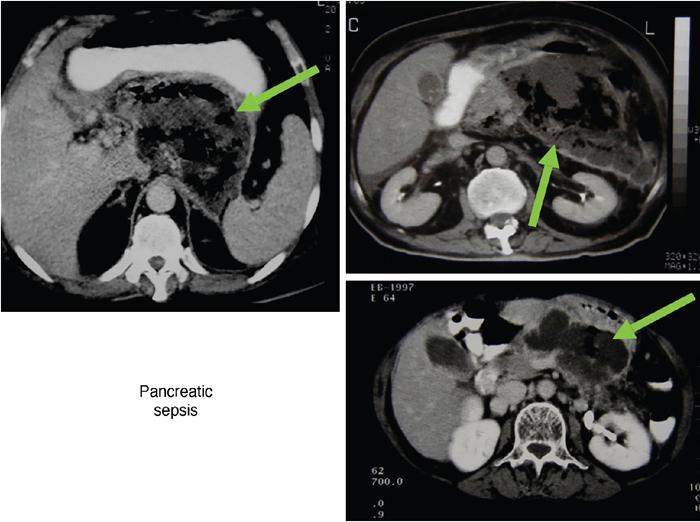
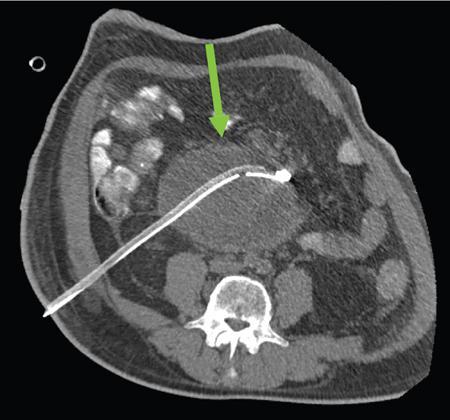
Anirudh Kohli Acute pancreatitis is one of the most dreaded diseases to afflict mankind, in view of its high morbidity and mortality. The single most important factor in reducing the morbidity and mortality of this condition over the last many years has been the role of computed tomography (CT). The incidence of acute pancreatitis is rising especially due to an increasing incidence of gallstones, obesity, as well as an ageing population. Pancreatitis occurs due to a chain of events triggered by a temporary/permanent pancreatic duct obstruction. This leads to activation and release of pancreatic enzymes into pancreatic interstitium and peripancreatic tissues leading to severe auto digestion and necrosis of pancreas and adjacent tissues. Systemically there is release of inflammatory mediators known as cytokines. Tumour necrosis factor is a cytokine which is toxic to acinar cells. These cytokines activate and intensify the inflammatory cascade that may ultimately culminate in multiorgan failure. Clinically severe acute pancreatitis is characterized by two phases, an early phase and a late phase. The early phase is dominated by the systemic effects of release of inflammatory mediators such as cytokines – termed as systemic inflammatory response syndrome (SIRS). Usually there is associated variable multiorgan dysfunction/failure. This phase lasts for a week. When the multiorgan failure lasts 2 days it is termed as mild, multiorgan failure lasting more than 48 hours is considered severe. There is no correlation between the clinical severity of pancreatitis and morphological changes in this early stage. In view of this CT scan is not of much utility during this phase, unless complications are suspected. The later phase is dominated by effects of local complications due to pancreatic/peripancreatic necrosis. Mortality follows this biphasic pattern in early weeks from the systemic effects of multiorgan failure and in later weeks due to local effects, for example infection of necrotic pancreatic/peripancreatic tissues superimposed by organ failure. The Clinical diagnosis of acute pancreatitis requires two of the following three features: If the first two findings are present without any significant SIRS then CT is not required. Acute pancreatitis is a complex disease with a wide variation in the presentation and outcome, ranging from asymptomatic with only biochemical alterations to a fatal outcome. In fact acute pancreatitis is a dynamic disease with continuously evolving appearances on imaging. There are two distinct forms of acute pancreatitis – interstitial oedematous pancreatitis and acute necrotising pancreatitis. Interstitial oedematous pancreatitis is a condition where there is only mild swelling of the pancreas with loss of normal lobulations and a diffuse decrease in attenuation of the pancreas. There may be heterogeneity of the pancreatic parenchyma due to varying degrees of interstitial oedema. This form of pancreatitis runs a mild course and rarely progresses to acute necrotising pancreatitis with its associated complications. In a third of these cases the pancreas may reveal no abnormality on a CT scan. The inflammatory changes may be restricted to the pancreas or extend into the peripancreatic regions. The inflammation in the peripancreatic regions manifests as acute pancreatic effusions. If there is peripancreatic fat necrosis with an oedematous pancreatic gland it is termed as acute necrotising pancreatitis (Figs. 9.17.1–9.17.2). Acute pancreatic fluid collections are enzyme-rich pancreatic juice collections seen in about 40% of patients with acute pancreatitis. The fluid collections occur due to exudation of pancreatic juices into the interstitium of the pancreas and subsequently leakage into the surrounding tissue spaces. These fluid collections are localized only by the anatomic space in which they collect. As the exudative process continues, the parietal peritoneum overlying the pancreas may be disrupted with the inflammatory fluid entering the lesser sac. From the lesser sac the fluid can enter the peritoneal cavity via the foramen of Winslow or by dissecting the peritoneum along the anterior surface of the lesser sac. Posterior extension of the fluid collection in the anterior pararenal space may occur into a potential space between the laminae of the posterior pararenal fascia. Rarely there may be involvement of the perirenal and posterior pararenal spaces. Other pathways of dissection are along the gastrohepatic, gastrosplenic and gastrocolic ligaments. Fluid may dissect along the root of the mesentery into the transverse mesocolon. Collections may extend around the caecum, ascending colon, descending colon and into the lumbar, pelvic and inguinal regions. Large fluid collections may dissect superiorly into the mediastinum or pericardial space. The quantity of fluid in these effusions is variable and can range from a small amount to large quantities (Fig. 9.17.3). Of these collections, 50% resolve spontaneously; the remainder may evolve after 4–6 weeks into pseudocysts. As the collections age, they tend to get localized and walled off, often developing an enhancing wall. A collection should be termed a pseudocyst only when a definite capsule develops and the collection has been static for at least 4–6 weeks. The risk of complications such as rupture, infection and haemorrhage increases with the age of the collection. Large collections of fluid are usually associated with a lesser degree of pancreatic necrosis as compared to extensive pancreatic necrosis associated with a lesser amount of fluid collection. Acute necrotising pancreatitis is a fulminant form of pancreatitis in which there is necrosis of the pancreas, that is nonviable pancreatic tissue. A contrast-enhanced CT is essential as it is the ideal means to demonstrate necrosis as well as to determine the extent of necrosis. These areas of nonviable pancreatic tissue do not enhance as compared to viable pancreatic tissue, which demonstrate significant enhancement. The extent of pancreatic necrosis has been found to correlate extremely well with the extent of necrosis found at surgery. The size, location and extent of pancreatic necrosis are very variable. These areas may be diffuse or focal, small in size to complete glandular involvement. These necrotic areas have a very important bearing on the course of pancreatitis, as they are liable to undergo secondary infection and form pancreatic sepsis. In addition, the more extensive the pancreatic necrosis the greater is the morbidity and mortality. Patients with no pancreatic necrosis have been found to have a 0% mortality and a 6% complication rate, whereas patients with pancreatic necrosis have a 23% mortality rate and an 82% complication rate. A variety of laboratory tests are available to detect pancreatic necrosis, such as a rise in the serum methemalbumin, quantitative estimation of C-reactive protein and urinary trypsinogen-activated peptide. CECT is still the most accurate modality to detect pancreatic necrosis. Pancreatic necrosis may be seen in the pancreas only, peripancreatic tissues also or only in the peripancreatic region. Isolated peripancreatic necrosis occurs due to disruption of the peripheral ductules with extravasation of activated pancreatic enzymes, may be seen in up to 20% of patients who require operative/interventional management of their necrotising pancreatitis. These patients with only peripancreatic necrosis have a better prognosis. This essentially represents retroperitoneal fatty tissue necrosis appearing as heterogeneous areas of liquid and nonliquid components. Over time necrosis evolves and liquefies as well as the liquid component resorbs. Generally if the liquid component is 2 cm or less it will resorb, while fluid collections 5 cm or more rarely resorb. If pancreatic necrosis is not resorbed it may get walled off or with time or may become infected. Encapsulation occurs between the third and fourth weeks. It is important to differentiate sterile from infected necrosis as sterile necrosis rarely needs surgery, whereas infected necrosis requires long-term antibiotic therapy and/or some kind of imaging/surgical intervention. According to the Atlanta 2012 classification, all areas of pancreatic necrosis are termed as acute necrotic collections. This represents a combination of pancreatic/peripancreatic fat necrosis with peripancreatic effusions. On CT, there is a spectrum of findings – solid, liquid containing debris (these may be nonwalled off/partially walled off). Sonography and MRI have advantages over CT scan in demonstration if the contents are pure liquid such as acute pancreatic effusions or have some nonliquefied components such as acute necrotic collections. Pseudocysts are round or oval encapsulated fluid collections containing only liquefied components. It takes 4 weeks for the granulation tissue to develop. On CT, a pseudocyst appears as a well-defined fluid collection with a thin capsule. The most common location for pseudocysts is the lesser sac, though they may be found anywhere in the mediastinum, abdomen or pelvis as they may dissect along fascial planes, along vessels and through capsules of solid organs. Pseudocysts in the bowel, though reported, are relatively rare, as the bowel wall is a strong barrier to the effect of proteolytic pancreatic enzymes. When the contents of the pseudocyst are heterogeneous or uniformly increased in attenuation, the possibilities of infection or haemorrhage should be considered. Focal areas of increased density within the fluid collection usually indicate haemorrhage. Complicated, enlarging or symptomatic pseudocysts require percutaneous catheter or surgical drainage. Infected pseudocysts are treated by percutaneous drainage. The management of a noninfected pseudocyst is controversial. Surgical treatment is only undertaken when the wall is mature after several weeks. Large pseudocysts greater than 5 cm in size can easily be drained percutaneously using intercostal drainage tube or pigtail catheter or endoscopically via the stomach. The cure rates are reported to be 85% with percutaneous drainage, the drainage period averaging about 20 days (Figs. 9.17.4–9.17.5). Walled off necrosis – as pancreatic/peripancreatic necrosis matures and evolves, an interface develops between necrosis and adjacent fatty tissue and an enhancing thickened wall is seen, resulting in a well-defined fluid collection with necrotic debris and fat necrosis. This is the end stage in the evolution of an acute necrotic collection. It is important to differentiate a pseudocyst from walled off necrosis, as a pseudocyst requires drainage and walled off necrosis requires surgical removal (Figs. 9.17.6–9.17.9). Sepsis is a major complication of pancreatitis and is accompanied by a high incidence of mortality and a prolonged hospital stay. Pancreatic sepsis may occur following secondary infection of pancreatic and/or peripancreatic necrosis, acute pancreatic fluid collections and pseudocysts. The only specific sign to demonstrate sepsis is the presence of gas in a collection. This occurs due to secondary infection by coliform/anaerobic organisms. This sign is unfortunately not common and seen in only one-third of cases. Gas is seen in the fluid collection as very dark well-defined air attenuation bubbles. Occasionally gas may be present due to a gastrointestinal fistula or previous surgery. Fat necrosis is seen in peripancreatic collections. This should not be confused with air bubbles, which are indicative of sepsis. The differentiation is easy to understand by CT, the values of fat ranging between – 20 HU and – 80 HU, and of air being >–300 HU. Also air bubbles have well-defined margins and are homogeneously jet black, whereas fat is nonhomogeneously grey with ill-defined margins. Since the presence of air is seen only in a small percentage of patients with pancreatic sepsis, the only other means to diagnose pancreatic sepsis is by CT-guided aspiration. All sites of fluid collection, parenchymal necrosis and peripancreatic necrosis are subjected to CT-guided aspiration. This is a tedious process and requires a dedicated interventional radiologist as there are often more than 5–6 sites from which it may be necessary to obtain samples. A fresh needle and syringe are used for each site, as it is important to know which site has sepsis and which is sterile. Care is taken not to transgress the large bowel as the colon has a large number of bacteria, and transgressing it could result in colonic bacteria contaminating the CT-guided aspiration sample, with falsepositive results. Further, colonic bacteria may be inoculated into a sterile acute pancreatic fluid collection or necrosis thereby converting a sterile collection into an infected one (Fig. 9.17.10). Complications of the procedure can be pneumothorax if the pleura is transgressed, haemorrhage due to trauma to a vessel, and secondary infection. Pancreatic sepsis is treated by surgical debridement, necrosectomy and drainage through thick tubes. It is crucial from the management point of view to differentiate an abscess from infected necrosis. An abscess may be treated by percutaneous drainage, whereas infected necrosis developing in relatively solid tissue can be treated by surgical debridement alone. Imaging-guided percutaneous drainage is the first step in treating pancreatic sepsis especially patients who are very ill and cannot undergo surgery. Initially pig tail catheters were used but these are notorious to get blocked as the largest bore of a pigtail catheter is 12 F. Pancreatic sepsis contains a large amount of necrotic debris, this blocks the catheters can be cleared by flushing the catheter, but this always raises the possibility of introducing secondary infection. To circumvent this issue large-bore ICD catheters have been introduced. These range from 16 F to 30 F. A safe window is necessary, not transgressing bowel or vasculature. It is quite easy to insert these tubes percutaneously under CT. A bit of initial manipulation and force is required to pierce the abdominal musculature. These large-bore drainage tubes have been very effective unless the infected necrosis is solid with minimal liquefied contents or a safe window for placing an ICD catheter is not available. CT is also invaluable in surgical planning and in the follow-up of postoperative patients to evaluate any fresh collection and also to determine whether the drains are well sited or not (Fig. 9.17.11).
9.17: Imaging in pancreatitis
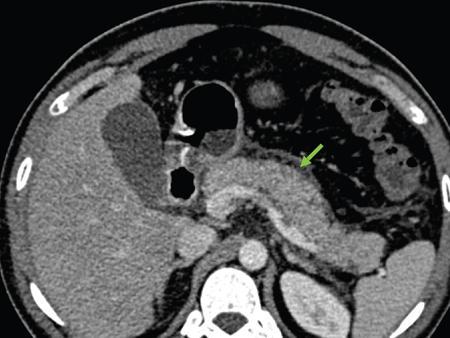
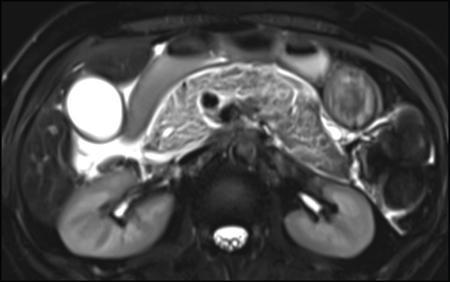
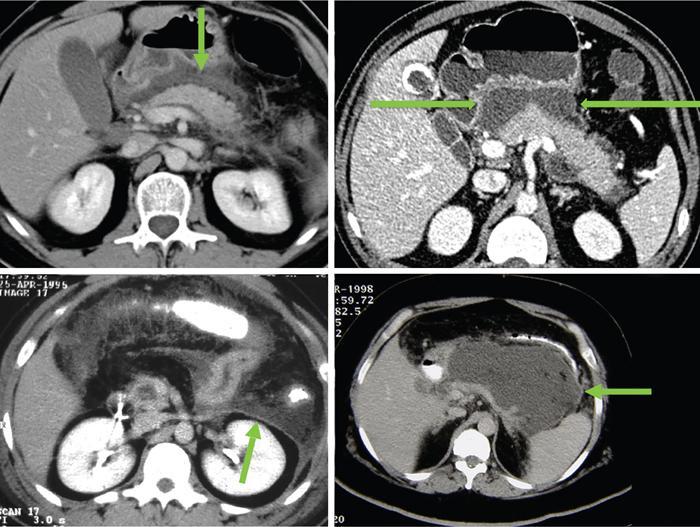
Pseudocysts
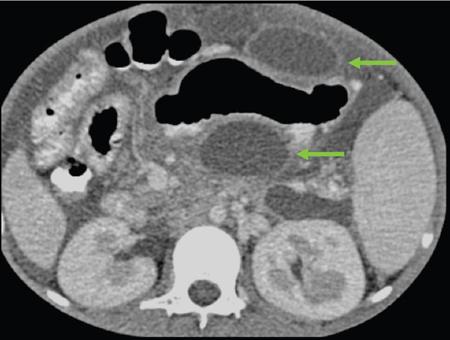
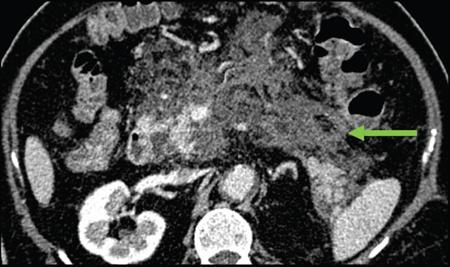
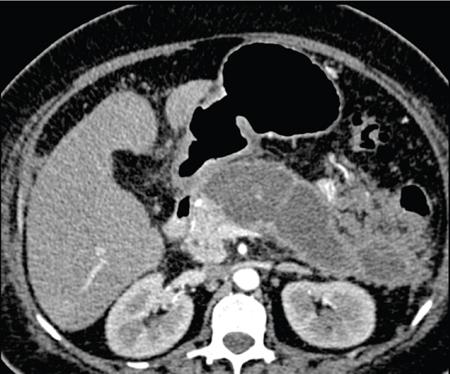
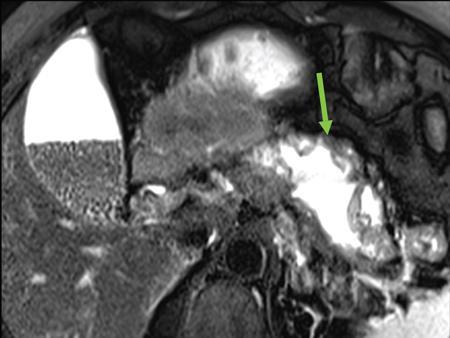
Sepsis
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








