Key points
- •
An awareness of the preoperative imaging, the surgical technique and history, and the set of clinical problems and imaging findings most likely to occur at specific postoperative time intervals will greatly improve the accuracy and value of imaging in reports patients with lumbar fusion.
- •
Fusion surgeries can be categorized into anterior, lateral, and posterior approaches, each with advantages and disadvantages.
- •
Outcomes studies have not demonstrated a specific benefit of one type over another.
- •
Surgical exploration is the gold standard for definitive fusion assessment. Thin-section computed tomography with multiplanar reconstruction is the most sensitive and specific imaging modality to detect pseudarthrosis.
Introduction
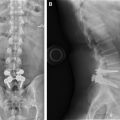
Lumbar spinal fusion, or arthrodesis, has been performed since 1911 when it was originally described by Drs Fred Albee and Russell Hibbs, who were the first to use autologous bone graft for spinal stabilization. As shown in Figs. 1 and 2 , decorticated, bleeding cortical surfaces are surgically created and osseous substrates are implanted in the disc space (interbody) or adjacent to the facet joints in a space termed the posterolateral gutter as the necessary components for successful fusion or bone healing. The process creates a physiologic environment favorable for bone formation, which is intended to limit motion of the treated spinal segment.
Fusion surgeries are performed to prevent motion of a single or multiple spinal segments, to alleviate pain or prevent neurologic compromise ( Box 1 ). Reviews and prospective randomized trials have demonstrated improved patient outcomes when successful fusion operations have been performed. Immediate stabilization improves the chance of successful arthrodesis. Before instrumentation was popularized, this was achieved with bracing or bed rest. Multilevel fusions were soon recognized as producing lower success rates for solid fusion and increased rates of pseudarthrosis. Instrumented fusion was designed to address this lower success rate with multilevel procedures. Over time, instrumented fusion was shown to provide effective immediate immobilization of the fused segment, theoretically increasing the likelihood of successful fusion for single-level procedures as had been shown for multilevel procedures. Implants, however, do not take the place of physiologic bony fusion, and are not designed to provide segmental immobility beyond the period during which true osseous fusion forms. Current practice favors the combination of decorticated, bleeding osseous surfaces and implanted substrate with spinal instrumentation, to provide early internal stabilization and thus allow for eventual complete osseous fusion, as shown in Fig. 3 .
Segmental instability: Loss of motion stiffness such that force application to the motion segment produces greater displacement than would be seen in normal structures, resulting in a painful condition that has the potential for progressive deformity and neurologic damage.
Introduction
Lumbar spinal fusion, or arthrodesis, has been performed since 1911 when it was originally described by Drs Fred Albee and Russell Hibbs, who were the first to use autologous bone graft for spinal stabilization. As shown in Figs. 1 and 2 , decorticated, bleeding cortical surfaces are surgically created and osseous substrates are implanted in the disc space (interbody) or adjacent to the facet joints in a space termed the posterolateral gutter as the necessary components for successful fusion or bone healing. The process creates a physiologic environment favorable for bone formation, which is intended to limit motion of the treated spinal segment.
Fusion surgeries are performed to prevent motion of a single or multiple spinal segments, to alleviate pain or prevent neurologic compromise ( Box 1 ). Reviews and prospective randomized trials have demonstrated improved patient outcomes when successful fusion operations have been performed. Immediate stabilization improves the chance of successful arthrodesis. Before instrumentation was popularized, this was achieved with bracing or bed rest. Multilevel fusions were soon recognized as producing lower success rates for solid fusion and increased rates of pseudarthrosis. Instrumented fusion was designed to address this lower success rate with multilevel procedures. Over time, instrumented fusion was shown to provide effective immediate immobilization of the fused segment, theoretically increasing the likelihood of successful fusion for single-level procedures as had been shown for multilevel procedures. Implants, however, do not take the place of physiologic bony fusion, and are not designed to provide segmental immobility beyond the period during which true osseous fusion forms. Current practice favors the combination of decorticated, bleeding osseous surfaces and implanted substrate with spinal instrumentation, to provide early internal stabilization and thus allow for eventual complete osseous fusion, as shown in Fig. 3 .
Segmental instability: Loss of motion stiffness such that force application to the motion segment produces greater displacement than would be seen in normal structures, resulting in a painful condition that has the potential for progressive deformity and neurologic damage.
Indications
Spine-related pain complaints are common and have several potential causes ( Box 2 ). The decision to operate on the spine to relieve pain is deferred until more conservative treatment methods fail to provide a therapeutic benefit. The broadest indications for elective spinal arthrodesis are pain or instability that threatens neurologic function.
Common
Muscle strain
Disc degeneration
Facet arthropathy
Spinal stenosis
Less Common
Trauma
Infection
Neoplasm
Instability (degenerative, traumatic, iatrogenic)
Scoliosis correction is a broad category that is not necessarily a fusion, and is beyond the scope of this article. Instability at the level of the disc and facet joints results in a cascade of degeneration leading to chronic low back pain and anatomic derangement. Disc removal and segmental arthrodesis have been used to address this problem. There are also acute clinical scenarios, such as those caused by acute spinal cord compression resulting in myelopathy, nerve root compression resulting in severe extremity pain and weakness, or cauda equina syndrome, which often necessitate immediate surgical intervention.
Primary versus secondary fusion
Primary fusion surgeries are performed on the group of patients thought to be suffering from low back pain who have segmental instability, but do not have demonstrable focal neural compression or spinal deformity. In this group of patients the rates of interbody fusion have dramatically increased, although the evidence to support which approach is best remains unclear. The general surgical approaches for fusion are described in Box 3 .
Anterior:
- •
Broad access to the anterior disc space allowing for complete anterior and central disc removal
- •
Optimization of fusion cage size and surface contact with the osseous endplates
- •
Restoration of full disc height
- •
Improved internal stabilization, which stimulates osseous fusion
- •
Decreased risk of epidural fibrosis
- •
Lateral:
- •
Wide access to the anterior half of the disc space allowing for a thorough discectomy, and placement of a large implant under adequate traction
- •
Avoids violation and direct retraction of the peritoneum, great vessels, and posterior paraspinal muscles
- •
Shorter operative time in comparison with laparoscopic anterior lumbar interbody fusion
- •
Posterior:
- •
Direct access to the spinal canal and neural elements allowing for complete decompression of the thecal sac and nerve roots, as well as some disc height restoration and a fairly complete discectomy
- •
Open access to the posterior elements facilitates optimal placement of spinal instrumentation such as pedicle screws and bridging rods
- •
Posterior dissection avoids risks of abdominal visceral injury
- •
Secondary instrumented fusions result from surgeries designed primarily to accomplish decompression of the thecal sac or nerve roots. When posterolateral fusion is not possible, or instability is likely, instrumented fusion is added to the decompression. When the decompression limits available posterior elements that serve as posterolateral fusion surface area, instrumented posterior fusion or interbody fusions are performed. Iatrogenic instability from bilateral facetectomies as part of a decompression may cause instability that would lead to kyphotic deformity or accelerated degeneration. Secondary fusions are additionally performed as part of these decompression surgeries.
Use of hardware
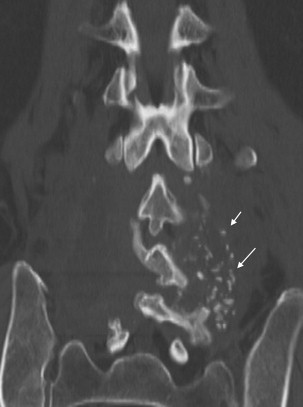
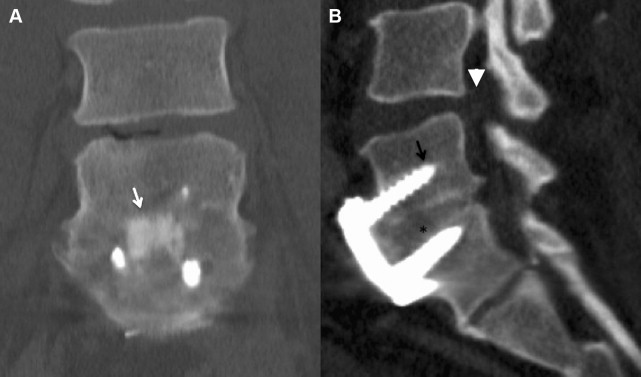
The rate of instrumented fusion increased rapidly as evidence for increased arthrodesis rates with instrumentation and the development of improved spinal hardware occurred simultaneously. Instrumentation in the current practice of lumbar fusion consists of pedicle screws, plates, rods, and intervertebral implants (hollow cages or solid ramps), which all act as internal fixation devices to immediately limit segmental motion from the time of surgery and are thought to improve the rates of subsequent osseous fusion. In addition, interbody grafts and devices increase disc-space height and can act to immediately increase foraminal and canal area by separating pedicles and putting traction on hypertrophied ligaments, as is shown in Fig. 4 .
Surgical methods
An understanding of the specific surgical approach allows an appreciation of the expected postoperative imaging appearance of the surgical bed as well as the possible complications, as shown in Figs. 5 and 6 ( Box 4 ). The surgical approach to spinal fusion can be thought of as anterior, posterior, or lateral. Each approach is used to achieve arthrodesis while avoiding injury to the thecal sac and exiting nerves. Traditional open surgical methods still predominate, although newer minimally invasive methods are growing in popularity for specific indications. Outcomes studies have not demonstrated a specific benefit of one type over another. Anterior and lateral access is preferred when disc degeneration, endplate changes, and loss of disc height predominate without the need for posterior or lateral decompression and fusion. In cases of focal posterior or lateral disc herniations and/or significant spinal stenosis, a posterior approach and decompression is necessary. Based on decreased operative time and blood loss, a convincing argument is made for stand-alone interbody fusion in those patients who do not require posterior decompression.
Posterolateral Lumbar Fusion (PLF)
Technique (Hibbs )
- •
Midline incision, retraction of the paraspinal muscles
- •
Decortication and placement of osseous substrate
- •
Instrumentation with pedicle-screw placement may be performed
- •
Laminectomy with posterior decompression may be performed depending on the specific pathologic circumstance
- •
Complications
- •
Fibrosis adjacent the exiting nerve roots or thecal sac
- •
Hardware or bone-graft material impingement of the neural elements
- •
Dural tears
- •
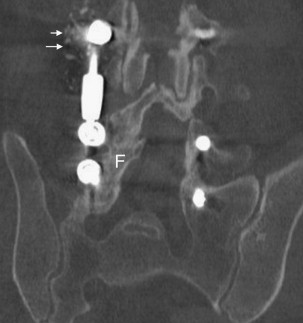
Posterolateral Lumbar Interbody Fusion (PLIF) (See Fig. 2 )
Technique (Cloward )
- •
Thecal sac and nerve roots freed and retracted to the midline
- •
Annulotomy and complete discectomy
- •
Removal of central disc material and cartilaginous endplates
- •
Implant to restore disc space height and optimize stimulus for osseous fusion
- •
Complications
- •
Retraction damage to the conus or exiting nerves
- •
Dural tears
- •
Fibrosis that can result in chronic pain
- •
Postoperative radiculopathy rates of up to 13%
- •
Postoperative instability and posterior migration of the fusion construct
- •
Transforaminal Lumbar Interbody Fusion (TLIF)
Technique (Harms and Rolinger )
- •
Placement of pedicle screws above and below the abnormal disc segment
- •
Distraction to provide space for implant
- •
Unilateral facetectomy and mobilization of the exiting nerve
- •
Preservation of the contralateral laminae and spinous processes
- •
Complete discectomy, bone-graft placement, and cage implantation
- •
Complications
- •
Cage migration rates of 1.17% (See Fig.3 )
- •
Fibrosis from retraction and passage of fusion implant through transforaminal space
- •
Anterior Lumbar Interbody Fusion (ALIF)
Technique (Burns )
- •
Parasagittal lower abdominal incision, retraction of the peritoneal contents, lumbosacral prevertebral neural elements, and great vessels to access the midline anterior lumbar spine
- •
Microsurgical modification of the ALIF developed by Mayer enters through the rectus muscles to the preperitoneal space
- •
Middle section of the annulus resected with the nucleus pulposus
- •
Cartilaginous endplates resected to level of bleeding osseous endplates
- •
Cage implant is fitted into the anterior disc space to restore disc height
- •
Complications
- •
Vascular injury, perforated viscera, and damage to the lumbar plexus
- •
Incontinence, sexual dysfunction, and retrograde ejaculation
- •
Abdominal wall hernias
- •
Deep venous thrombosis
- •
Extreme Lateral Lumbar Interbody Fusion (XLIF)
Technique (Ozgur )
- •
Lateral flank incisions used to access the retroperitoneal space
- •
Blunt dissection used to reach to the psoas muscle, which is split in anterior third
- •
Discectomy is performed similar to the ALIF technique, with preservation of the posterior annulus and creation of a wide space for an anterior fusion implant
- •
Complications
- •
Vascular damage
- •
Psoas hematoma
- •
Lumbar plexus nerve damage
- •
Abdominal wall hernias
- •
Deep venous thrombosis
- •
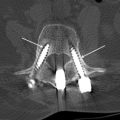
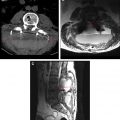
Intraoperative imaging
There is currently no standard practice for image guidance in the operating room. Imaging for instrumented lumbar fusion is performed on a spectrum from no image guidance (based on anatomic landmarks, neurophysiology, and tactile response) to full 3-dimensional real-time computer-assisted image guidance using an O-arm computed tomography (CT) and pedicle-screw tracking systems as shown in Figs. 7 and 8 . Use of intraoperative fluoroscopy is the mainstay of clinical practice, along with a growing use of an O-arm CT guidance system. Several studies have demonstrated improved accuracy of pedicle-screw placement using image guidance to verify placement of screw trajectories. Newer systems using preoperative CT, computer registration, and modeling provide real-time tracking of implant placement during the operations.