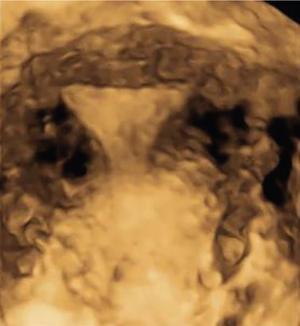
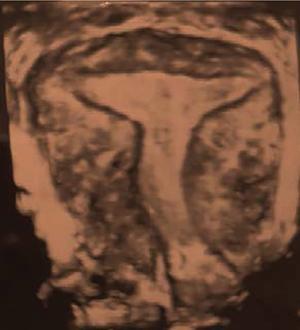
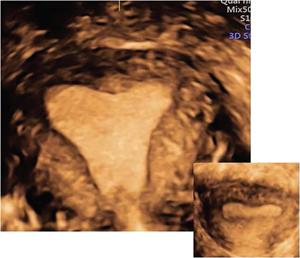
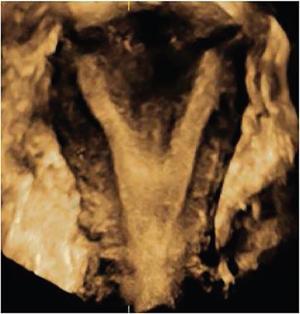
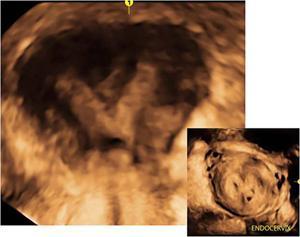
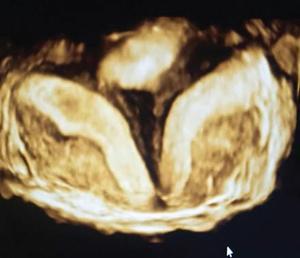
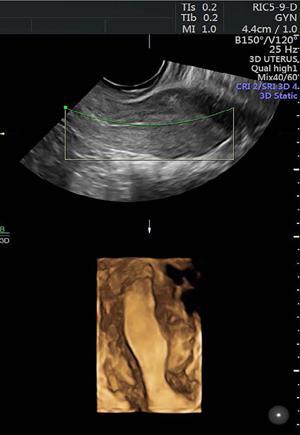
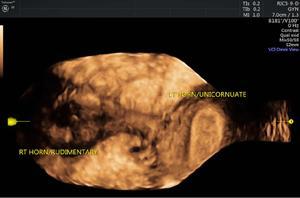
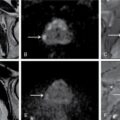
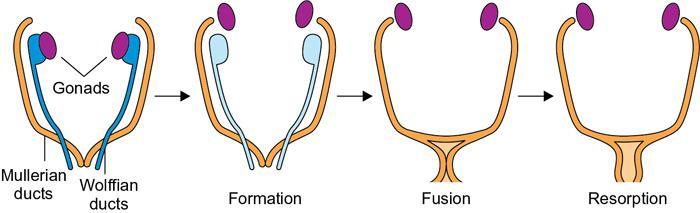
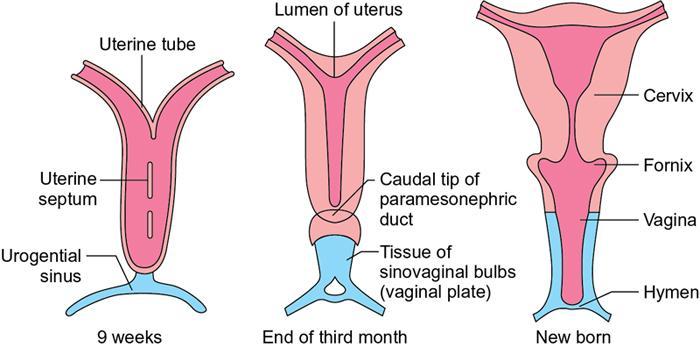
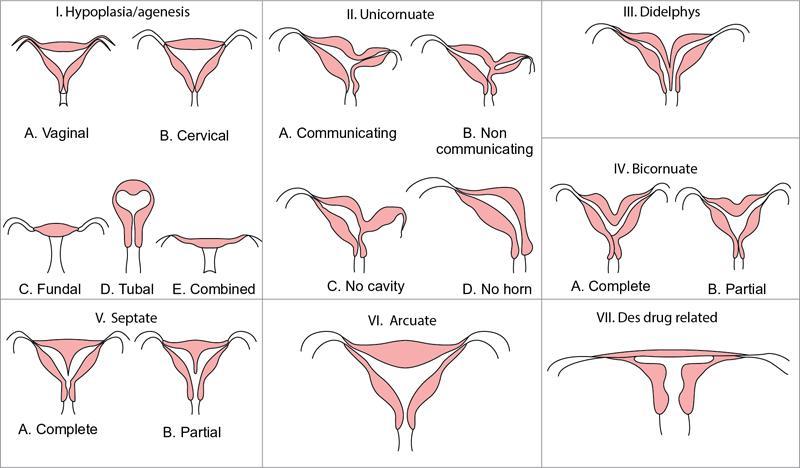
MÜLLERIAN DUCT ANOMALIES Rajani Gorantla Müllerian ducts are a pair of tubular structures that give rise to uterus, cervix, fallopian tubes and upper two-thirds of vagina. Disruption or failure of normal development can result in occurrence of Müllerian duct anomalies (MDAs). These are commonly associated with other congenital anomalies of urinary tract and ovaries due to the close relationship between the development of mesonephric and paramesonephric ducts; few patients also have associated skeletal system anomalies. The prevalence of MDAs in an unselected population was 5.5%; 8% among the infertile women, 12.3% in women with a history of recurrent pregnancy loss and 24.5% in women with miscarriage and infertility. Most of the patients with MDAs are asymptomatic and found incidentally on imaging for any other gynaecological problems, in evaluation of a patient with renal, skeletal or abdominal wall abnormalities. In symptomatic women, complains are related to the type, severity, obstructive anomaly or nonobstructive anomaly. The symptoms can be obstetric or gynaecological related, includes recurrent spontaneous abortions, infertility, preterm labour, intrauterine growth restriction and abnormal foetal lie. Few adolescent girls may present with primary amenorrhoea, hypomenorrhoea, abnormal vaginal bleeding, cyclical pain and mass. Rarely they can present with pelvic inflammatory disease with abnormal vaginal discharge, dyspareunia and urinary tract infections. The female reproductive system develops from a pair of Müllerian or paramesonephric ducts, urogenital sinus and vaginal plate. The ovaries develop separately from the primordial ridge. It is of great importance to understand the normal stages of development, as failure or interruption of any of these stages can lead to a simple to complex spectrum of anomalies. The three stages of Müllerian duct development include: At 6–10 weeks of gestation age, two paired Müllerian ducts and Wolffian ducts formation occurs. When Y chromosome factor (testicular determining factor) is absent, the Wolffian ducts undergo degeneration. The Müllerian ducts further elongate caudally and cross the Wolffian ducts to fuse in the midline. At 10–13 weeks of gestation age, caudal and lateral fusion of the two Müllerian ducts forms primitive uterovaginal canal, which is possessed of solid tissue initially located side by side and further internal canalization leads to the formation of two channels/canals divided by a septum. At this stage, there is reabsorption of the caudal septum and forms single cervical canal and vagina lumen. In around 15–20 weeks of gestation age, complete resorption of the septum takes place in a caudocranial direction from the isthmus to fundus and development of single endometrial cavity results. The fused caudal part of Müllerian ducts gives rise to the uterus, cervix and upper two-thirds of vagina; the unfused cranial part forms the fallopian tubes (Fig. 11.14.1.1). The lower vagina develops from the urogenital sinus, which is separated from the rectum by urorectal septum around 7 weeks of gestation age. The primitive uterovaginal canal embeds into the dorsal wall of urogenital sinus and forms Muller’s tubercle. Around 13 weeks of gestation, two solid masses known as sinovaginal bulbs originate from the upper part of the Muller’s tubercle, further proliferate into the caudal end of the uterovaginal canal to become a solid vaginal plate. Later canalization or degeneration of the central cells of the vaginal plate forms the lower vagina, which is usually completed by 20 weeks’ gestation. The vertical fusion of the upper and lower vagina occurs with resorption of tissue in between and forms single vaginal cavity (Fig. 11.14.1.2). The vaginal lumen is separated from the urogenital sinus by the hymenal membrane. Just before birth, the hymen normally ruptures due to retrogression of the central epithelial cells. However, a thin fold of mucous membrane persists around the vaginal introitus. The ovaries develop separately from migration of primordial germ cells to the genital ridge. Ureteric buds develop separately and concurrently. So renal anomalies are most commonly associated with MDAs with an association of 30%–50%. Various classification systems have been proposed over the past several decades to describe MDAs. Ideally, same classification system needs to be followed by a gynaecologist, surgeon and radiologist for better communication. Buttram and Gibbons classification was suggested in 1979, and it was based upon the level of failure in normal development and segregate the anomalies into groups with similar clinical manifestations, treatment, and prognosis for foetal salvage. According to Buttram and Gibbons, the uterine anomalies were classified into six classes as described in Table 11.14.1.1. The drawback is the lack of classification of vaginal and other anomalies separately. The ASRM (previously the American Fertility Society – AFS) classification system is the most widely accepted classification worldwide over the past years and was introduced in 1988. According to this classification, MDAs are classified into seven classes (class I to class VII) as mentioned in Table 11.14.1.2. However, several limitations have been described by Grimbizis and Campo in 2010 (Fig. 11.14.1.3). The drawbacks of the ASRM classification system are as follows: Hence in 2016, an updated classification of uterine septum, that is ASRM-2016 was proposed and officially approved morphometric criteria are given for distinguishing between septate, normal/arcuate and bicornuate uteri (Table 11.14.1.3). The VCUAM classification was proposed in the year 2005. The main concern is to provide a simple, systematic, clinical classification in addition to providing a precise reflection of the entire malformation. The female genital organs were divided into the following subgroups in accordance with the anatomy: vagina (V), cervix (C), uterus (U) and adnexa (A). Associated malformations were assigned to a subgroup (M) relative to each specific organ. The disadvantage is due to its inherent complexity, and more than 56,700 individual combinations of anomalies are possible. ESHRE and ESGE established a common working group named CONgenital UTerine Anomalies (CONUTA) in order to devise an improved classification system (Table 11.14.1.4). It was published in 2013. Anatomy is the basis for systemic categorization of the MDAs. The subclasses are divided by the different degrees of uterine deformity and their clinical significance (Fig. 11.14.1.4). Cervical and vaginal anomalies are classified into independent supplementary subclasses (Figs. 11.14.1.5–11.14.1.6). For most of the clinicians, it helped as starting point for the development of guidelines for their diagnosis and treatment. The malformations are graded according to severity, U0–U5, C0–C4 and V0–V4, with U5, C4 and V4 being more severe. Class U3 incorporates bicorporeal fusion defects (didelphys and bicornuate) as this was considered as a more functional mode of classification. Arcuate uterus was not included separately, but this is categorized under normal variant into class U1c. Recent studies have demonstrated that the ESHRE/ESGE system provides an effective and comprehensive classification for almost all the currently known MDAs and overcomes the limits of previous classifications. However, there is a relative overdiagnosis of septate uterus with the application of ESHRE–ESGE criteria has been reported and which potentially might lead to unnecessary surgical overtreatment. C0 C1 C2 Normal cervix Septate cervix Double ‘normal’ cervix C3 C4 Unilateral cervical aplasia Cervical aplasia U4 V0 V1 V2 V3 Normal vagina Longitudinal nonobstructing vaginal septum Longitudinal obstructing vaginal septum Transverse vaginal septum and/or imperforate hymen V4 Vaginal aplasia C MDAs’ characterization and classification is of great significance, as the treatment is determined by the type and severity of abnormality. The various imaging modalities available include: HSG was the most recognized imaging modality, earlier to the development of the ultrasonography (USG) and MRI. It is an invasive fluoroscopic-guided procedure for uterine and tubal assessment, and is performed during the midproliferative phase of the cycle, ideally between days 7 and 10 of the cycle when endometrium is thin. Fluoroscopic spot images obtained to evaluate uterine configuration, uterine filling defects and fallopian tube patency. HSG allows evaluation of only the component of the uterine cavity that communicates with the cervix. The anatomic information about myometrium and external fundal contour will not be provided by HSG. The diagnostic criteria used to diagnose MDAs on HSG include: HSG findings of different MDAs are described in Table 11.14.1.5 and Fig. 11.14.1.10. Virtual HSG is a noninvasive technique performed by using a computed tomography (CT) scanner, done 45 s after the contrast material instillation begins. Contraindications are similar to those for HSG and include pregnancy and active pelvic infection. There is no necessity for retraction of the uterus or manipulation of the cervix. The procedure is quick, easy and prophylactic administration of antibiotics is not required. The use of a power injector helps to ensure a steady low pressure of instillation. The procedure is less painful, more comfortable and easily tolerated by patients than conventional HSG. Radiation exposure sometimes requires cervical clamping, which may result in complications such as bleeding and infection. Transabdominal pelvic ultrasound can diagnose uterine anomalies with accuracy rate of 47%. Two-dimensional transvaginal ultrasound (TVUS) has high sensitivity and specificity than transabdominal study and provided some information about external and internal fundal contours. The detection rate is high if the scan is performed in secretory phase due to better visualization of endometrium. Three-dimensional USG shows great accuracy than 2D USG in evaluation of the uterine morphology. The technique of 3D USG varies with different vendors. It displays both the external and internal fundal contours and lower uterine segment by acquisition of single coronal view of uterus (c-view) (Fig. 11.14.1.11). The only disadvantage is that it is transvaginal study and shall not be done in paediatric age group and sexually inactive women. Three-dimensional TVUS has become the first line of screening tool in most of the infertility clinics as it is noninvasive, faster, repeatable, allows storage of volume data and has multiplanar capability for systematic evaluation of the uterine and cervical cavities. The salient features of various Müllerian anomalies on 3D USG are described in Table 11.14.1.6 with images. Three-dimensional ultrasound is combined with sonosalpingography in this technique and provides better delineation of the uterine morphology. It is a less invasive procedure done in proliferative phase with instillation of saline into the uterine cavity and assessment of the uterus will be done. MRI is a universally accepted imaging modality in the documentation of MDAs and accuracy rate of 100% have been reported. MRI provides excellent delineation of both the internal and external uterine anatomies. T2-weighted (T2W) images are the mainstay of pelvic imaging and are performed without fat suppression. T1-weighted (T1W) images are mainly for the haemorrhagic content. The disadvantages of MRI include time-consuming procedure, not cost-effective, large body habitus, pacemakers, recent surgical history and claustrophobia. The current and proposed MRI protocol given by the European Society of Urology (ESUR-MRI protocol) intends a dedicated evaluation of MDAs as mentioned below (Fig. 11.14.1.12): Vaginal anomalies can be accurately diagnosed with the prior administration of the ultrasound gel, to distend vagina (Fig. 11.14.1.13). It will help in better diagnosis of complex vaginal anomalies, like vaginal septations or vaginal duplication. The normal MRI appearance of uterocervical canal and vagina are shown in Fig. 11.14.1.14. Uterine aplasia/hypoplasia/agenesis is class 1 MDA according to ASRM classification and U5 uterine anomaly according to ESHRE classification. It is a formation defect of the paramesonephric ducts with complete or segmental agenesis of uterus and vagina. The incidence rate is around 10%–15% of all MDAs and considered as the most severe form of uterine anomaly. Most of them have complete uterovaginal agenesis with no single completely developed uterine cavity and are associated with Mayer–Rokitansky–Küster–Hauser syndrome (MRKHS). Two types of this syndrome are depicted. The typical form or type A represents the absence or remnants of the uterus, cervix, upper two-thirds of vagina with normal ovaries and fallopian tube (Fig. 11.14.1.15). The atypical form or type B is associated with the abnormalities of the ovaries, fallopian tubes and genitourinary system (Fig. 11.14.1.16). In a few cases (approximately 10%), unilateral or bilateral uterine remnants and with or without endometrial cavity will be seen (Fig. 11.14.1.17). Most of them present with primary amenorrhoea due to complete uterovaginal agenesis and manifest as MRKHS. These patients have normal secondary sexual characteristics due to the preserved normal ovarian function and phenotype. If uterine remnants with functional cavity is present, will present with cyclical pain along with amenorrhoea due to cryptomenorrhoea and haematometra. USG is the first modality for identification of these anomalies, absence of uterus with normal ovaries is diagnostic. But, it is difficult to locate the uterine remnants and cavity due to small acoustic window. MRI is the gold standard and it classifies into uterovaginal agenesis and hypoplasia. If hypoplasia is identified, sagittal and axial sections are taken. These remnants are seen as T2 hypointense tubular structures, located in close relation to ovaries in the adnexa. Once we identify the uterine remnants, it is important to look for the functional endometrial cavity. These will have reduced endometrial and myometrial width. MRI can easily delineate the zonal anatomy due to its high soft tissue contrast resolution. The management of Müllerian agenesis consists of counselling for the patient and her parents. Some patients with MRKHS opt for creation of neovagina for normal sexual life. Various techniques are available. In the presence of a functional Müllerian remnant, regardless of whether it is communicating or not, medical suppression of menses can be initiated and should be followed by laparoscopic removal of the hypoplastic remnant. Unicornuate uterus or hemiuterus is defined as the unilateral uterine development and the contralateral Müllerian duct could be either partially formed or absent. It is a formation defect and the necessity to classify it in a different class than that of uterine agenesis, which is also a formation defect, is due to the existence of a fully developed functional uterine hemicavity. It is considered as class II anomaly according to ASRM classification and class U4 according to ESHRE classification. The frequency rate is around 20% of MDAs. Renal anomalies most often occur in association with unicornuate uterus and usually on the same side of uterine agenesis. Further unicornuate uterus is divided into two subclasses depending on the presence or absence of a functional rudimentary cavity in ESHRE classification: In ASRM classification, unicornuate uterus is divided into four subcategories: Mostly asymptomatic and presence of the noncommunicating uterine remnants will be identified at the time of infertility work up or caesarean section. If a functional cavity is present, the presentation is dysmenorrhoea or haematometra in an adolescent. The common obstetrics-related complications include abnormal foetal lie, intrauterine growth retardation, preterm delivery, placental abnormalities and uterine rupture. Gynaecological complications are ectopic pregnancy and endometriosis due to retrograde menstruation. On imaging, unicornuate uterus is seen as tubular and fusiform or banana-shaped structure at paramedian location, the endometrium is narrow and tapers to the apex with normal myometrial anatomy and reduced uterine volume. The rudimentary cavity or remnants are better depicted on 3D USG (Fig. 11.14.1.18) and MRI. However, MRI is superior to USG due to its high soft tissue resolution. The rudimentary cavity can communicate with main cavity or connected by fibrous band. The nonfunctioning cavity will be seen as T2 hypointense structure with loss of zonal anatomy (Figs 11.14.1.19 and 11.14.1.20), whereas the functioning cavity will show the deformed zonal anatomy (Fig. 11.14.1.21), and its complications like haematometra as T1/T2 hyperintense endometrial collection. Unicornuate uterus without rudimentary cavity does not require any procedure. But in the presence of communicating or noncommunicating rudimentary horn, surgical resection must be considered to prevent complications. Uterine didelphys is a class III MDA based on the ASRM classification and class U3b bicorporeal uterus according to ESHRE classification with an incidence rate of 5% among the uterine anomalies. It is a lateral fusion defect; results from complete failure of the Müllerian duct fusion. Two noncommunicating endometrial cavities with preserved zonal anatomy are seen with separate cervix. According to ESHRE classification, it is defined as external fundal indentation completely dividing the uterine corpus up to the level of external cervical os. It is most commonly associated with longitudinal vaginal septum in around 75% of cases. Some patients may show transverse vaginal septum and these will be obstructive or nonobstructive type. Renal anomalies are also commonly associated with uterine didelphys. Uterine didelphys is usually asymptomatic and diagnosed incidentally on pelvic examination or caesarean section. The uterine didelphys with obstructed vagina (Fig. 11.14.1.22) will present at adolescence as dysmenorrhoea, haematometrocolpos and haematosalpinx. Sometimes retrograde menstrual flow can cause endometriosis and pelvic adhesions. Obstetric-related complications include abortion/foetal growth restriction and poor pregnancy outcome. The uterine didelphys with obstructed hemivagina due to transverse vaginal septum and associated ipsilateral renal agenesis are the manifestations of the syndrome called obstructed hemivagina and ipsilateral renal agenesis anomaly/Herlyn–Werner–Wunderlich (HWW) syndrome (Fig. 11.14.1.23).
11.14: Imaging of paediatric female reproductive tract
Introduction
Incidence
Clinical features
Embryogenesis
Key points to remember
Classification
Buttram and Gibbons classification
CLASSIFICATION OF MÜLLERIAN ANOMALIES BY BUTTRAM AND GIBBONS (1979)
American society for reproductive medicine (ASRM) system for Müllerian duct anomalies
Class
Classification
Class I
Agenesis or hypoplasia – (a–e) (vaginal or cervical or fundal or tubal or combined)
Class II
Unicornuate – (a–d) (communicating horn or noncommunicating horn or no cavity or no horn)
Class III
Uterine didelphys
Class IV
Bicornuate uterus (a and b – complete or partial)
Class V
Septate uterus (a and b – complete or partial)
Class VI
Arcuate uterus
Class VII
DES-related
Uterine Morphology
Inner Fundal Contour
External Fundal Contour
Normal
Straight or convex
Uniformly convex or indentation <10 mm
Arcuate
Concave fundal indentation with central point of indentation at obtuse angle (>90 degrees)
Uniformly convex or with indentation <10 mm
Subseptate
Presence of septum, which does not extend to cervix, with central pt. of septum at acute angle (<90 degrees)
Uniformly convex or indentation <10 mm
Septate
Uterine septum completely dividing cavity from fundus to cervix
Uniformly convex or with indentation <10 mm
Bicornuate
Two well-formed uterine cornua
Fundal indentation >10 mm dividing the two cornua
Unicornuate (rudimentary horn)
Single well-formed uterine cavity with single interstitial portion of fallopian tube and concave fundal contour
Fundal indentation >10 mm dividing the two cornua, if rudimentary horn present
The vagina cervix uterus adnexa–associated malformation (VCUAM) classification
European society of human reproduction and embryology (ESHRE) and European society of gynaecological endoscopy (ESGE) classification
UTERINE ANOMALY
CERVICAL/VAGINAL ANOMALY
Main Class
Subclass
Coexistent Class
UO
Normal uterus
U1
Dysmorphic uterus
U2
Septate uterus
U3
Bicorporeal uterus
Hemiuterus
U5
Aplastic
U6
Unclassified malformations
U
V
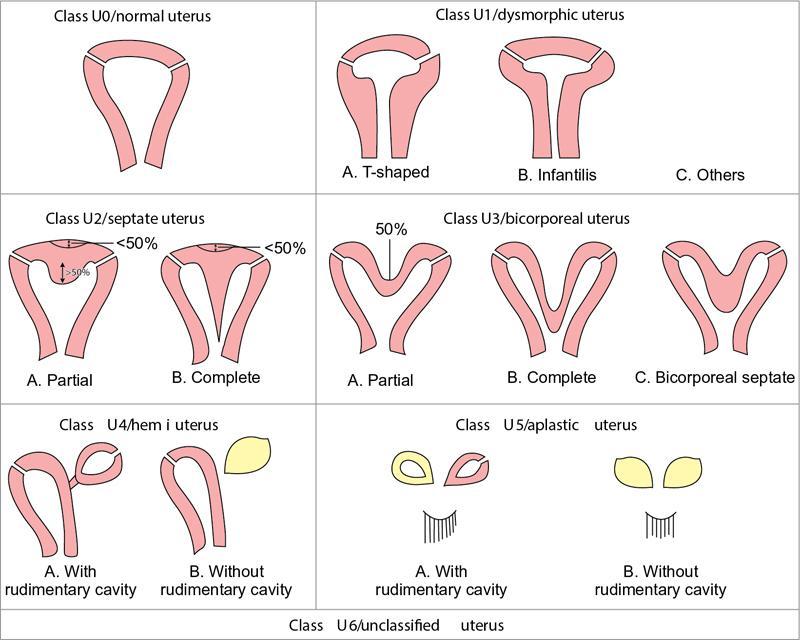
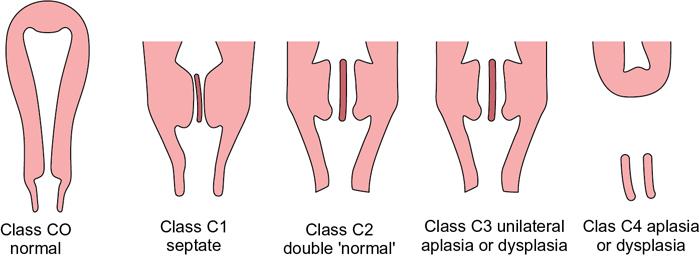
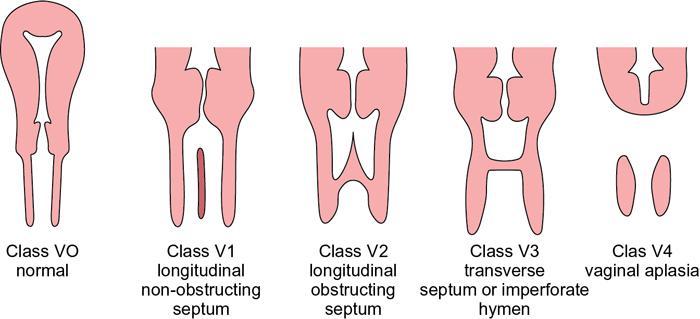
ESHRE guidelines for classification of Müllerian duct anomalies


Imaging modalities
Hysterosalpingography
Arcuate Uterus
Septate Uterus
Bicornuate Uterus
Didelphys Uterus
Unicornuate Uterus
DES Exposure
Single uterine cavity with a broad saddle-shaped indentation at the uterine fundus
Two cavities with intercornual angle of less than 75 degrees
Two cavities with intercornual angle of more than 105 degrees
Two separate, oblong endometrial cavities with contrast opacification of fallopian tubes
Off-midline fusiform cavity deviated to one side with opacification of the single fallopian tube
T-shaped appearance with shortened upper uterine segment
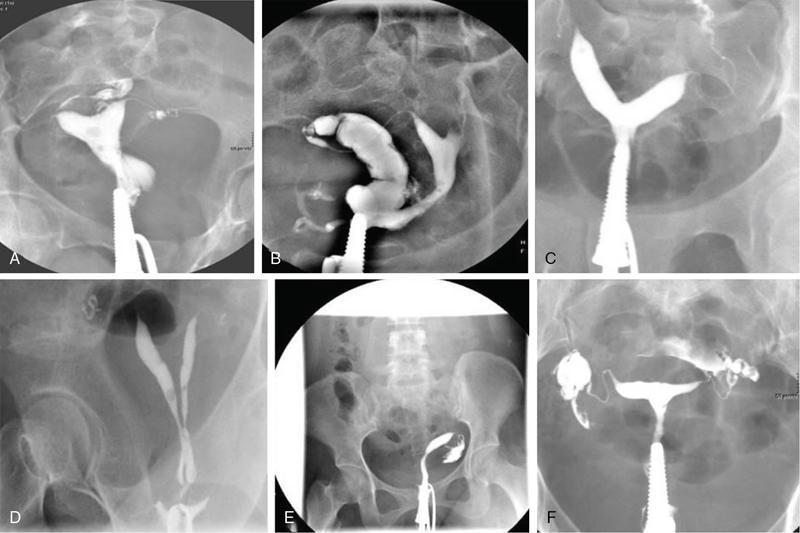
Virtual hysterosalpingography
Uses
Advantages
Disadvantages
Ultrasonography (2D and 3D)
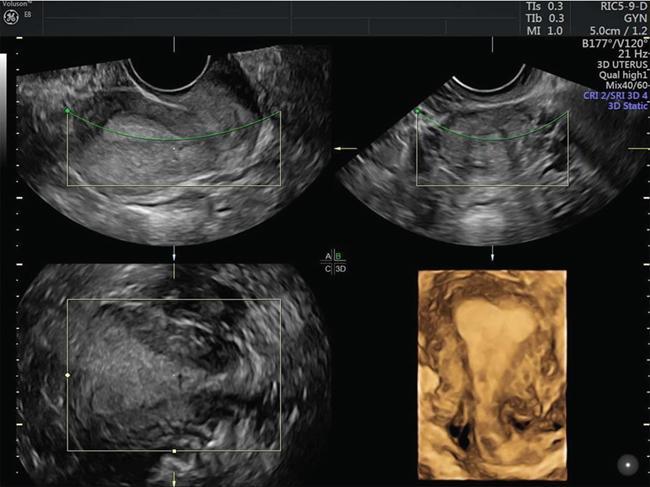
3D sonohysterography
MRI
MRI protocol
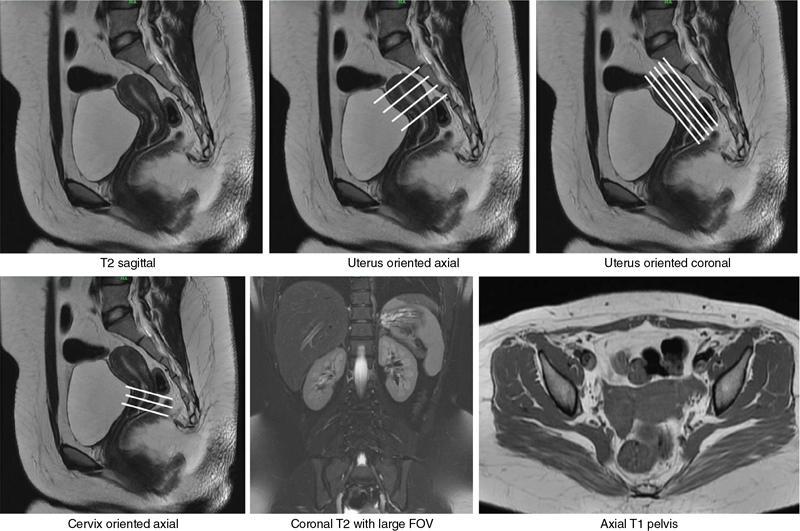
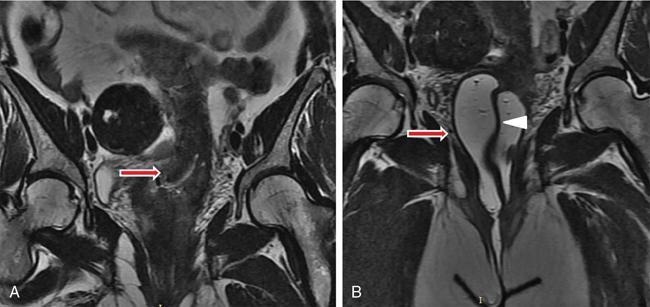
Imaging features of Müllerian duct anomalies
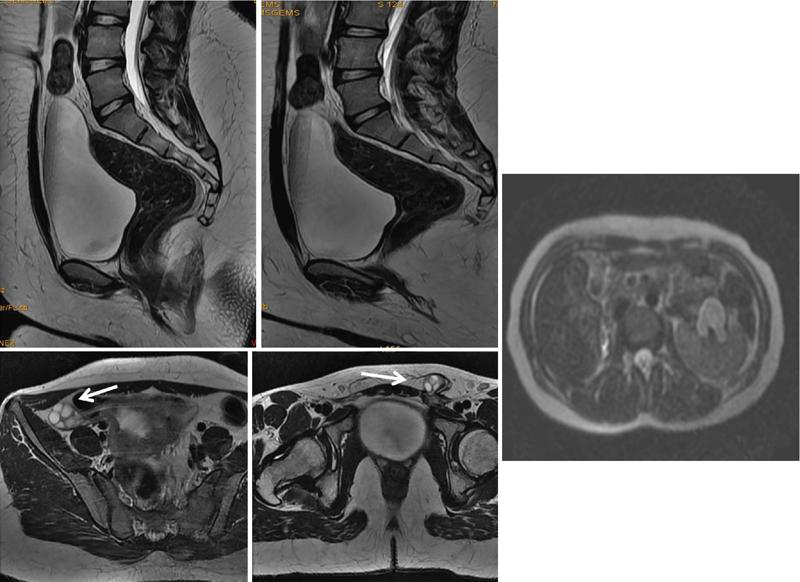
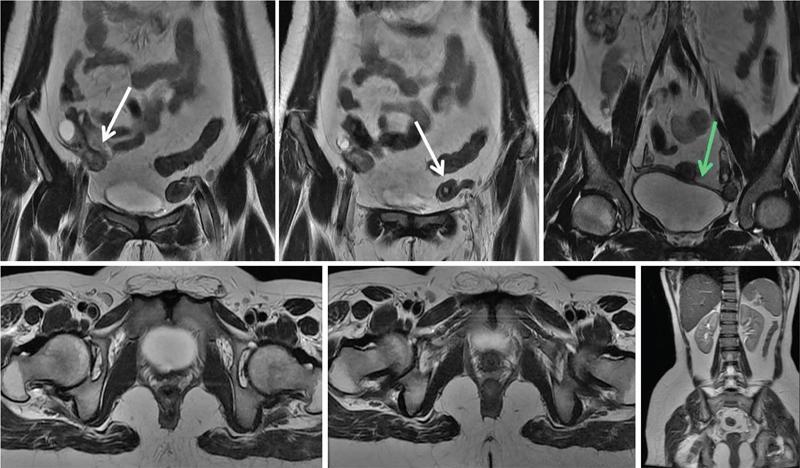
Uterine aplasia/dysplasia/agenesis

Clinical features
Imaging
Management
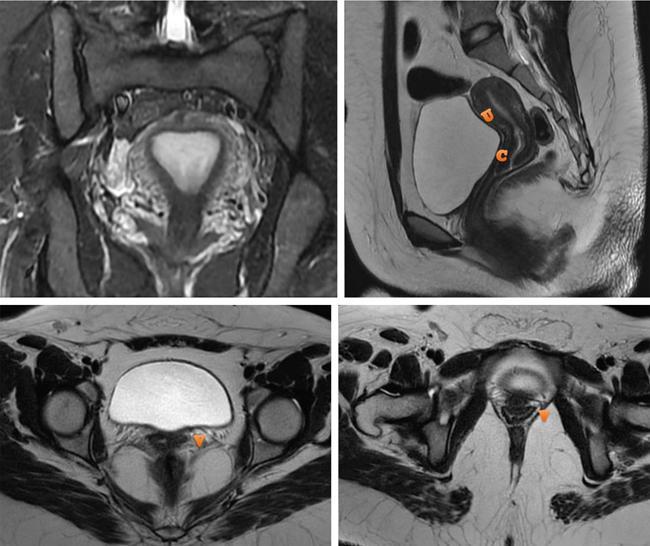
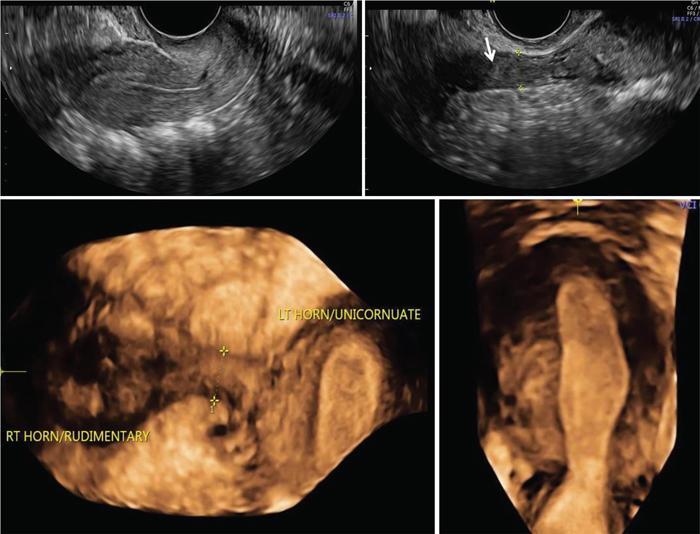
Unicornuate uterus
Clinical features
Imaging appearances
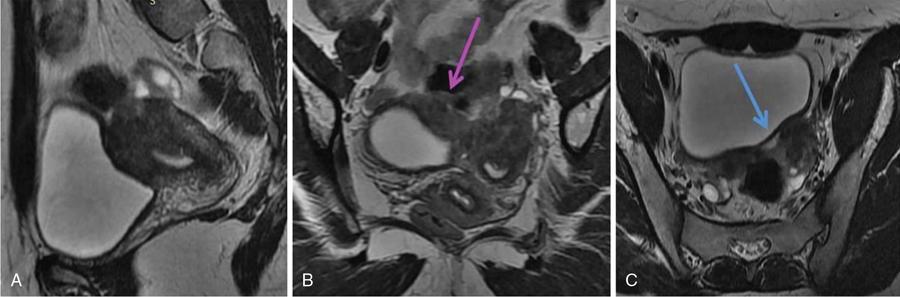
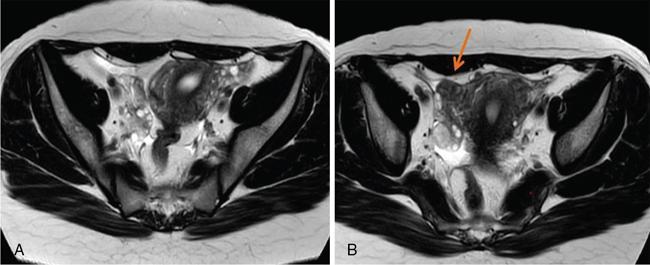
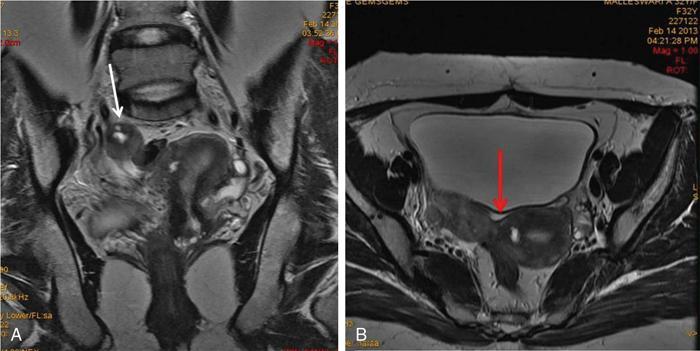
Management
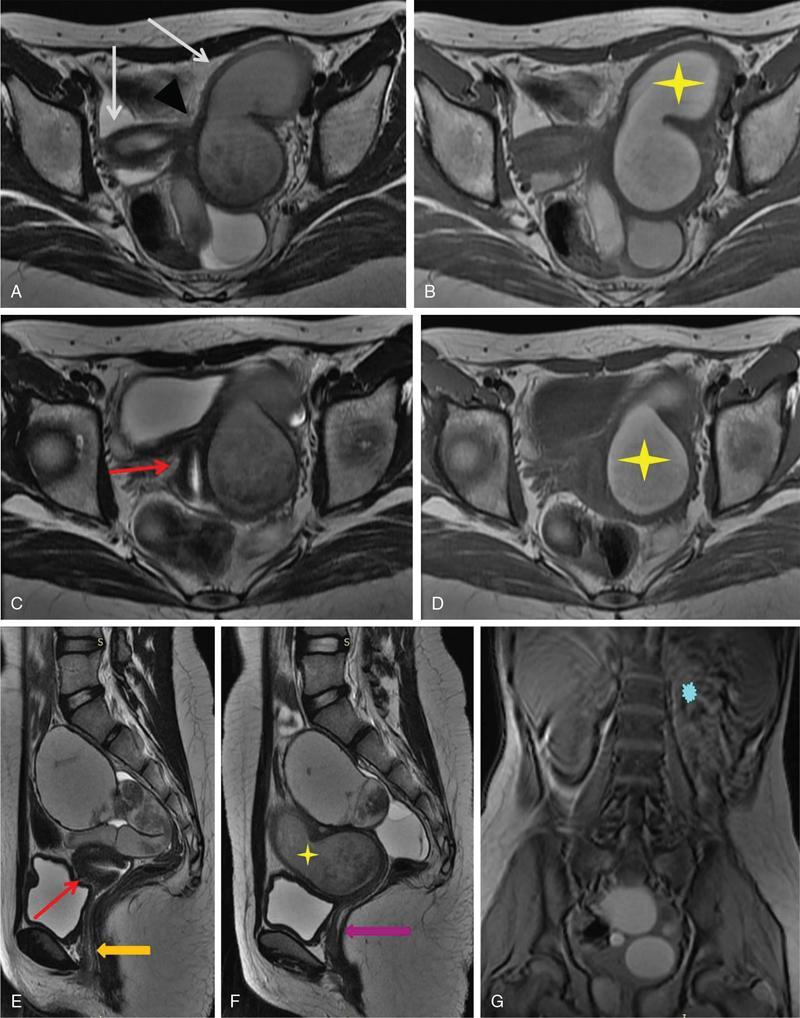
Uterine didelphys
Clinical symptoms
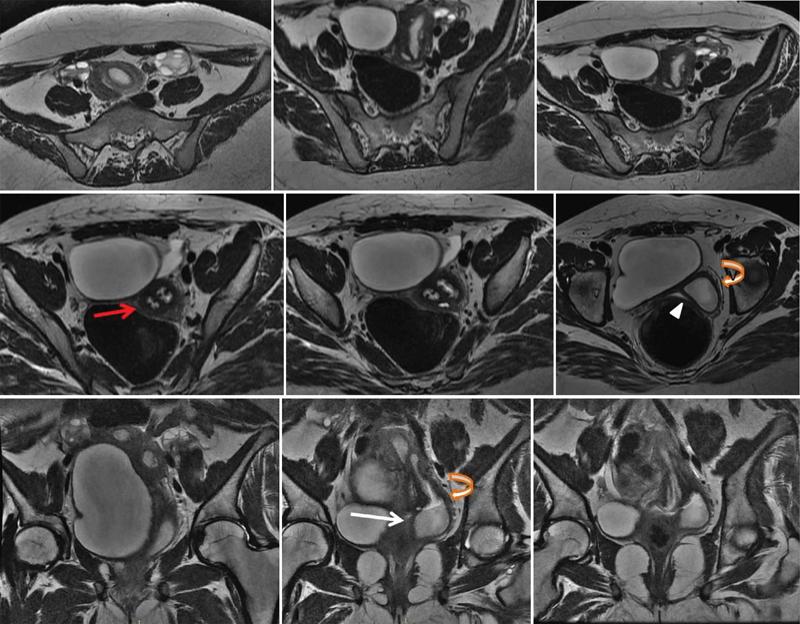
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree