Fig. 1
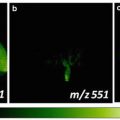
The imprinting process, shown here for the imprinting of a barley leaf. (a) The leaf is placed on the clean, porous Teflon surface and covered with tissue paper and a rubber slice. (b) The sandwich is placed in a vice a pressed for a few seconds. (c) The sandwich is opened. (d) The imprint (here visible, but not necessarily always) is ready for imaging upon a few minutes of drying in a vacuum desiccator or under ambient conditions
3.2 DESI Imaging of Imprinted Plant Material (See Note 8 )
1.
A photo is taken of the imprint, using a camera or a microscope, for subsequent comparison between the imprint and the MS images.
2.
The imprint is placed in the DESI imaging ion source like any other sample. A mixture of, e.g., acetonitrile and water may be applied as spray solvent; the exact composition of the spray solvent is chosen and optimized according to the type of analyte to be imaged (see Note 10 ).
3.
The spray geometry is optimized on an imprint not intended for imaging (see Note 11 ). The geometry optimization is relatively simple for the porous Teflon surface, and the settings are not nearly as critical as in, e.g., imaging of animal tissue section mounted on glass slides. Moreover, the success ratios with indirect DESI imaging are very high compared to other DESI imaging experiments.
4.
5.
The DESI images are generated on a computer using the appropriate software.
4 Notes
1.
Although the porous Teflon surface itself is extremely nonpolar, the method has been found not to discriminate significantly between polar and nonpolar compounds [3]. This was tested by comparing the DESI signal from an imprint of a capsule of the opium poppy (Papaver somniferum) with the electrospray ionization signal from a methanol extract of a similar capsule in a direct infusion ESI analysis. The mutual abundances between highly polar compounds such as morphine and less polar compounds such as papaverine were similar for both of the two techniques. This can most likely be ascribed to the plant juice, with all its contents, being squeezed into the pores of the Teflon surface by mechanical means. This results in a nonselective transfer of compounds, in contrast to an extraction process as known from, e.g., solid-phase extraction, which is much more chemically selective.
2.
The plant material which is to be imaged must be fresh material with some amount of plant juice present which can transfer the analytes to the imprinting surface. The method is therefore not readily applicable for imaging of dried plant material.
3.
Porous Teflon can be purchased, e.g., from Berghof (www.berghof.com, Eningen, Germany) which produces it on request with the desired specifications in terms of thickness, average pore size, and porosity (pore volume in %). For practical reasons, a thickness of 1.6 mm is preferable. The two other parameters can be combined as, e.g., 36 % porosity with 7 μm pore size or 47 % porosity with 16 μm pore size. Given the effective resolution of about 100 μm which is typically obtained in a DESI experiment, there does not seem to be much difference between images made with the two types of porous Teflon. In other DESI studies, porous Teflon with 25 μm pore size has been purchased from Small Parts Inc. (Miami Lakes, FL, USA) [15], or from Fluoro-Plastics (Philadelphia, PA, USA) [16].
4.
In practice it is often desirable to imprint at least two pieces of the plant material next to each other, so that one piece can be used for test and optimization of the DESI signal, while the imprint intended for imaging is not touched until the actual start of the image acquisition.
5.
The purpose of the tissue paper is to absorb the surplus plant juice which is not absorbed by the porous Teflon. In the absence of tissue paper, very wet and blurred imprints are typically obtained. The amount of tissue paper may be adjusted to the type of plant material which is to be imprinted.
6.




The rubber slice may in some cases be omitted. Its purpose is to distribute the pressure when samples do not have a completely even thickness. In the absence of the rubber slice, one may for example find that the regions of a leaf lying close to the midrib of the leaf are not sufficiently imprinted, since the protrusion of the midrib may “protect” them from the pressure applied. In this case, the rubber slice—being more flexible than the metal plate—serves to apply the pressure evenly throughout the sample.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree