There are various complications that can occur in the postpartum period, including pain, bleeding, and infection. These include complications related to cesarean section, postpartum hemorrhage and hematomas, bladder injury, torsion and uterine dehiscence, and rupture. It is important the radiologist is aware of these entities and the associated imaging features to help guide timely and appropriate management.
Key points
- •
There is significant overlap between normal postpartum findings and complications.
- •
Hematoma management is based on location.
- •
Endometritis is a clinical diagnosis and negative imaging does not exclude it.
- •
The immediate postpartum period has high risk for adnexal torsion when a mass is present.
- •
Enhanced myometrial vascularity can be seen with normal placental implantation site involution or associated with retained products of conception.
Introduction
The postpartum period extends from delivery of the placenta to the completion of uterine involution, which may take as long as 8 weeks. Maternal mortality in the United States rose from 12 per 100 000 births in 1990 to 28 per 100,000 in 2013. Postpartum hemorrhage (PPH) is the most frequent cause of severe maternal morbidity.
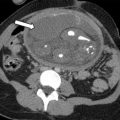
Diagnosis of postpartum complications requires a team approach with careful imaging, laboratory studies, and clinical surveillance. There is considerable overlap between the normal appearance during involution and pathologic states; gas within the uterine cavity may be seen for several weeks after delivery, increased myometrial blood flow and echogenic material in the endometrial cavity ( Fig. 1 ) are not necessarily abnormal and do not always predict the need for intervention.
The imaging findings in postpartum complication can be grouped anatomically or in relationship to mode of delivery. Cesarean delivery (CD) carries higher risk of complications, which are reviewed specifically followed by an overview of the findings associated with PPH, retained products of conception (RPOCs), placental site subinvolution, fibroid infection/infarction, and adnexal or uterine torsion.
Complications related to cesarean section
Cesarean section (CS) accounts for one-third of deliveries in the United States. The overall complication rate of CD is 14.5%, with infection (eg, endometritis, ovarian vein thrombophlebitis, and wound infection) the most common. Risk factors for complications include prolonged rupture of membranes, long duration of labor prior to surgery, anemia, and obesity. Other perioperative complications include hematoma, uterine artery pseudoaneurysm formation, bladder injury, and fistula development.
Scar endometriosis tends to present later when menses resume. Scar dehiscence and/or rupture, cesarean scar ectopic pregnancy, and placenta accreta spectrum occur in future pregnancies.
Endometritis
Endometritis is the most common cause for persistent low-grade postpartum fever; it is a clinical diagnosis and the imaging findings are nonspecific, overlapping with expected postpartum changes. It is approximately 30 times more common with CS than with vaginal birth.
Ultrasound (US) and computed tomography (CT) show nonspecific uterine enlargement and endometrial fluid and/or gas. US is superior for the detection of RPOCs; CT is superior for identification of parametrial inflammation and pelvic abscess. The treatment is antibiotic administration with appropriate management of any associated RPOC-infected hematoma or pelvic abscess.
Ovarian Vein Thrombophlebitis
Pelvic septic thrombophlebitis occurs in 0.05% to 0.18% of all deliveries and 1% to 2% of those with endometritis ; thus, it is more common after CD. The right ovarian vein is involved in 80% to 90% but both veins may be involved.
CT or MR imaging is preferred over US due to limited acoustic access. The vein is enlarged; luminal enhancement is absent or diminished due to central thrombus but the vein walls may enhance and the ovary may be enlarged.
Postoperative Hematomas
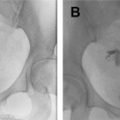
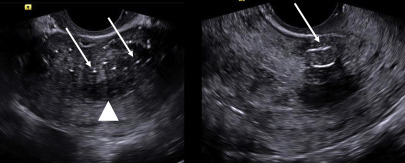
Bladder flap hematoma is extraperitoneal, between the bladder and the lower uterine segment (LUS) secondary to bleeding after low transverse CS. It is common, occurring in 50% of women and should be less than or equal to 4 cm in greatest thickness. Hematomas greater than 5 cm increase concern for scar dehiscence. Bladder flap hematomas tend to have thicker edges compared with hematomas at the incision defect, which are thickest at the center ( Fig. 2 A).
US shows a hypoechoic mass between the bladder and the intact LUS. Echogenicity decreases over time as clot liquefies. On CT, look for the typical location and lack of enhancement. Bleeding may spread to the broad ligaments and retroperitoneum with resultant hydronephrosis due to mass effect on the distal ureters. Superimposed infection may manifest as echogenic foci with dirty posterior shadowing on US or as rim-enhancing collections with or without septations and gas locules.
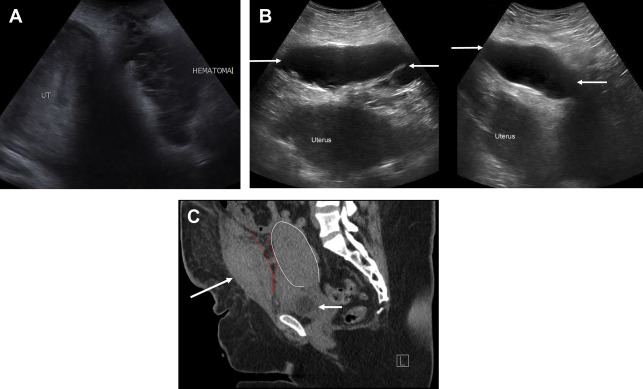
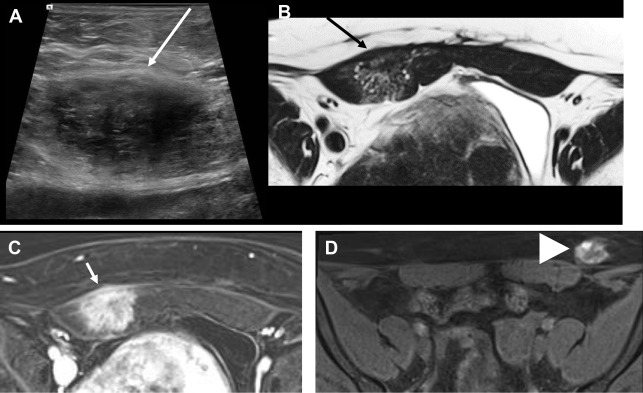
A subfascial hematoma also is extraperitoneal, arising from the inferior epigastric vessels. Hemorrhage is posterior to the rectus and transversalis muscles, anterior to the peritoneum, and contiguous with the space of Retzius ( Fig. 2 B, C), with the potential for large-volume blood loss (up to 2.5 L ) and hemodynamic instability; thus, recognition is critical. On US, there is a complex, solid, or cystic heterogeneous mass anterior to the bladder. Contrast-enhanced CT may identify active bleeding, which can be treated with embolization.
Subcutaneous hematomas are anterior to the rectus muscle.
Uterine Artery Pseudoaneurysm
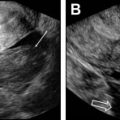
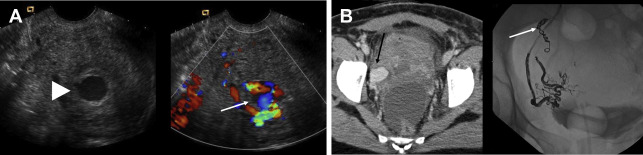
Uterine artery pseudoaneurysm is a rare but important complication of CS because rupture can lead to life-threatening hemorrhage. The sonographic diagnosis is made when color Doppler demonstrates turbulent, bidirectional flow, increased velocity, and the pathognomonic yin-yang sign in a hypoechoic lesion adjacent to the scar ( Fig. 3 A). CT and MR imaging show an enhancing mass in the myometrium or parametrium ( Fig. 3 B). Definitive diagnosis is made at angiography. Treatment by embolization has a 97% success rate.
Bladder Injury
Urogenital fistulas are more likely to follow CD than obstructed labor. The incidence of bladder injury during CS is 0.28%, with the greatest risk during creation of the bladder flap (43%), especially with adhesive disease from previous CS; 95% occur at the bladder dome, the rest at the trigone. Diagnosis usually is made intraoperatively with prompt repair. Imaging is used to confirm healing before catheter removal and to evaluate later complications, such as vesicovaginal, vesicouterine, or ureterovaginal fistula. Surgery is the definitive treatment. Post-CS fistulae repair is delayed by 3 months to wait for involution of the uterus and tissue healing.
Scar Endometriosis Implant
Endometriosis can occur in abdominal incisions with an average 3.6-year delay between surgery and the development of symptoms. The incidence is 0.03% to 1.5% with prior CD. The ectopic endometrium is functional, which causes cyclic pain due to recurrent soft tissue bleeding. Sonographic evaluation of abdominal wall pain or mass is best performed with a linear high-frequency transducer. Routine pelvic US employs 4 MHz to 6 MHz transducers for transabdominal views and 4 MHz to 9 MHz transducers for transvaginal imaging, neither of which is optimal for assessment of the anterior abdominal wall.
On US, the implants usually are solid, hypoechoic, and heterogeneous with irregular margins. Increased echogenicity adjacent to the implant reflects inflammatory/reactive changes secondary to bleeding, which creates small cystic space within the mass. Internal blood flow may be present.
On CT, abdominal wall endometriosis manifests as an enhancing mass with adjacent inflammatory change ( Fig. 4 ). On MR imaging, the key finding is areas of T1 hyperintensity due to subacute blood, which distinguishes endometriotic implant from other abdominal wall masses. Biopsy is definitive for diagnosis. Treatment is wide local excision; there is a recurrence rate of 4.3%.
Uterine Dehiscence
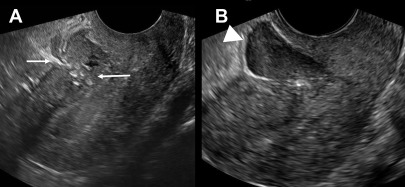
Uterine dehiscence is defined as separation of the endometrium and myometrium with intact serosa. The imaging diagnosis is difficult due to normal physiologic thinning of the LUS myometrium but should be considered in a pregnant patient with pain and LUS myometrial thickness of less than 2 mm or with a bladder flap hematoma greater than 5 cm in thickness in a postpartum patient. CT is the modality of choice postpartum, and reformatted images perpendicular to the incision scar may be useful. Treatment usually is conservative with antibiotics, with surgery reserved for complete ruptures ( Fig. 5 ).
Uterine Rupture
Uterine rupture is defined as separation of all layers of the uterine wall, including the serosa, creating a connection between the uterine cavity and the peritoneal space. Intrapartum rupture is a surgical emergency and imaging is not performed. Rupture may occur in association with prior uterine surgery or maternal trauma and should be suspected when there is focal discontinuity of the myometrium, hemoperitoneum, gas extending into the uterine cavity, or abnormal location of the fetus. ,
Cesarean Scar Ectopic Pregnancy and Placenta Accreta Spectrum
Both entities, cesarean scar ectopic pregnancy and placenta accrete spectrum, are manifestations of abnormal pregnancy implantation and placenta adherence to the cesarean scar. Early recognition and management is of the utmost importance, with some investigators suggesting that a 5-week to 7-week scan is mandatory in women with a prior CD. These topics are discussed in detail elsewhere in this issue.
Postpartum hemorrhage
PPH is the leading cause of maternal morbidity worldwide. Primary (immediate) PPH occurs within 24 hours of delivery whereas secondary (delayed or late) PPH occurs between 24 hours and 12 weeks after delivery. In the United States, PPH complicates 3% of all deliveries and 77% are due to uterine atony. Atony commonly presents in the immediate phase and is diagnosed and managed clinically with little role for imaging. Other causes of primary PPH include delivery-related trauma or coagulopathy.
Secondary PPH complicates approximately 0.2% to 1% of pregnancies, , with RPOCs accounting for at least 30% of cases. Other causes of secondary PPH include subinvolution of the placental implantation site, endometritis, uterine artery pseudoaneurysm, and benign or malignant uterine masses.
Retained Products of Conception
RPOCs are postpregnancy intrauterine remnants of placental and/or fetal tissue. The all-inclusive term of postpregnancy includes after spontaneous abortion, medical and surgical termination, and vaginal delivery or CD. RPOCs are more common after terminations, miscarriages, and second-trimester demises than term deliveries and after vaginal deliveries than CDs. Presentation is nonspecific, with bleeding, pelvic pain, fever, and/or uterine tenderness. Beta human chorionic gonadotrophin (β-hCG) may not be clinically useful because it can be positive or negative in the setting of retained products. It is beneficial, however, to obtain a baseline to exclude markedly elevated or rising levels seen with gestational trophoblastic disease.
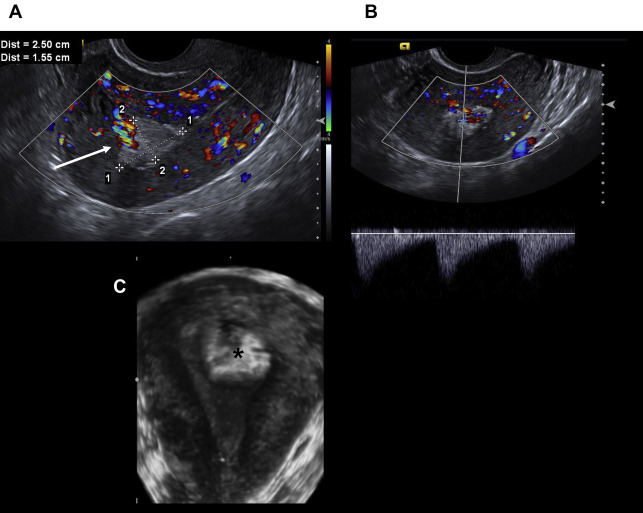
US is the primary imaging modality for evaluation of PPH. The greatest diagnostic strength of US lies in its negative predictive value. Durfee and colleagues showed no intervention was required for patients who lacked an endometrial mass or had an endometrial thickness less than 10 mm.
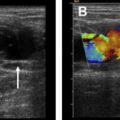
Sonographic findings of RPOCs include a focal endometrial mass and/or endometrial thickness greater than 10 mm ( Fig. 6 ). Rarely, retained placenta can present as a calcified mass. Gray-scale findings alone have a false-positive rate up to 34%, which is understandable given the significant overlap in imaging features between RPOCs and expected postpartum lochia.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree