Vijay Kumar K.R. CONGENITAL SPLENIC ABNORMALITIES A multitude of variants and pathologies affect the spleen and it is pertinent for a radiologist to be aware of the common imaging appearances of the same. The spleen begins to develop in the fifth week of gestation from a mass of mesenchymal cells that condense between the two leaves of the dorsal mesogastrium. Several such aggregates fuse to form the lobulated embryonic spleen. The spleen is translocated to the upper left side of the abdominal cavity due to rotation of the stomach and differential growth of the dorsal mesogastrium. The mesogastrium fuses with the peritoneum over the left kidney and forms the splenorenal ligament. The lienorenal ligament fuses dorsally and the splenic artery courses behind the peritoneum as it enters the splenorenal ligament. The portion of dorsal mesentery between the stomach and spleen forms gastrosplenic ligament. Differentiated mesenchymal cells form the parenchyma, connective tissue and capsule of the spleen. The foetal spleen shows lobulations, which normally disappear before birth. However, they may persist along the medial part of the spleen. Sometimes a persisting portion of the spleen may extend medially and anteriorly over the upper pole of the left kidney. It may be mistaken for a space-occupying lesion arising from the kidney. Rarely it may extend posterior to the upper pole of the left kidney, displacing it anteriorly (Fig. 9.22.1.1). The notches and clefts along the superior borders are vestiges of the grooves that initially separated the foetal lobules. They may be 2–3 cm deep. They may mimic a splenic laceration in a patient with abdominal trauma, and the embedded peritoneum at the waist may simulate splenic infarction or haematoma. Hence caution should be exercised. A normal spleen which does not have a fixed ligamentous attachment can change position within the abdomen and is hence known as a wandering spleen. The spleen is normally anchored by the gastrosplenic and lienorenal ligament. Absence or weakness of one or more ligaments that affix the spleen. The spleen with its vascular pedicle as the sole attachment can become a hypermobile intraperitoneal organ. Rarely, the spleen may be normal in position with a wandering accessory spleen. Pregnancy (due to hormonal effects), prune belly syndrome, splenic cysts, malaria, Hodgkin’s disease and lymphangioma. Some of the affected patients may be asymptomatic and diagnosis may be incidental. Clinically it presents as a mobile, palpable, notched mass. Pressure on the vascular pedicle or torsion causes symptoms such as acute abdominal pain, nausea and vomiting. Torsion involving the tail of the pancreas may present as pancreatitis. Confirms the absence of spleen in the left upper quadrant of abdomen with identification of splenic tissue at an ectopic site. Power Doppler, colour Doppler and duplex sonography allow for the identification of blood flow in the splenic vessels and parenchyma. Intermittent torsion may be responsible for an enlarged spleen. Pathognomonic feature of wandering spleen is absence of the spleen in the upper abdominal cavity and presence of a well-demarcated, homogeneously enhancing mass of soft tissue in the pelvis or abdomen. Sometimes a wandering accessory spleen can be visualized as a long vascular pedicle connecting a small mass to the splenic vessels (‘jokari sign’). It can locate ectopic splenic tissue and can also indicate the viability of the tissue. Splenic vessel anatomy can be well demonstrated. 99mTc-sulphur colloid scintigraphy and heat denatured 99mTc-labelled red blood cell imaging provide useful information regarding size and location of splenic tissue. Torsion is indicated by lack of tracer activity in a previously demonstrated wandering spleen. Twisting and torsion of splenic artery can be demonstrated by celiac arteriography. The venous phase may demonstrate splenic vein obstruction with collateral circulation and varices. A congenital focus of normal splenic tissue that is separate from the main body of the spleen is known as an accessory spleen. It results from the failure of fusion of some embryonic splenic buds within the dorsal mesogastrium. Its incidence varies from 10% to 30% on autopsy examinations. They vary from a few millimetres to several centimetres in diameter. They most commonly are found around the splenic hilum. The second most common location is the pancreatic tail. They can also be found in the stomach wall, omentum, mesentery, large bowel and scrotum. Reveals a round or oval mass of tissue around the splenic hilum that is isoechoic with the main spleen. On noncontrast images they are isodense to the main spleen. CECT reveals serpiginous enhancement on arterial phase followed by homogeneous enhancement on venous phase-like spleen (Fig. 9.22.1.2). Accessory spleen is mildly hypointense to the spleen on T1 images and isointense on T2 images. Contrast administration reveals enhancement similar to that of spleen on the arterial, portal and late phases. It is isointense to spleen on DWI images with similar ADC values. 99mTc-sulphur colloid scintigraphy and heat denatured 99mTc-labelled red blood cell imaging show functional uptake in splenic tissue and differentiate splenule from other masses. PET CT-FDG avid mass can mimic tumour. Surgical resection is the preferred mode of treatment in the background of hypersplenism or lymphoma. It is characterized by an ectopic splenic tissue or an abnormal connection between the gonad and the spleen. The left side is more commonly involved than the right side. The accepted theory is that splenic gonadal fusion occurs due to abnormal attachment of the splenic anlage in the left dorsal mesogastrium with the left urogenital fold, during the fifth to eighth weeks of gestation. It has a higher male preponderance and can interfere with left testicular descent and closure of processus vaginalis. It commonly presents as inguinal hernia and cryptorchidism. The spleen and gonad are attached by a fibrous or splenic cord across the peritoneal cavity. It is associated with limb defect syndrome, cleft palate, micrognathia, anal anomalies, craniosynostosis, spina bifida, cardiac malformations, thoracic malformations and diaphragmatic hernias. This is not associated with congenital anomalies. Shows a well-defined scrotal mass with echotexture similar to that of spleen. Confirms splenic tissue in inguinal or scrotal area. It is characterized by the absence of spleen with duplication of right-sided structures. (Right isomerism). It has a slight male preponderance with an incidence of 1 in 40,000 live births. It has a poor prognosis due to its high association with congenital cardiac abnormalities (Fig. 9.22.1.3). It is of uncertain aetiology, and maybe associated with the mutations of CFC1 gene, SHROOM3 gene etc. Delayed development of embryonic curvature can be another cause. It is also theorized that it can be caused due to vascular compromise by pressure on splenic blood supply from adjacent structures. Splenectomy – Usually has a surgical history and is not accompanied by associated anomalies. It is a heterotaxy syndrome associated with multiple spleens and duplication of left-sided structures. (Left isomerism). It has an equal gender predilection with a slightly higher female preponderance. It has a fairly better prognosis than asplenia and is associated with a lesser incidence of cardiovascular anomalies (Fig. 9.22.1.4). It is of uncertain aetiology, and maybe associated with the mutations of CFC1 gene, SHROOM3 gene etc. Accelerated development of embryonic curvature can be another cause. SPLENIC INFECTION AND ABSCESS The infection of the spleen can show three patterns. Fungal and granulomatous infections usually present with multiple micronodules. A pyogenic abscess usually manifest as a solitary lesion. Imaging of diseases causing splenic nodules is never the sole basis for diagnosis. The clinical presentation and laboratory testing (tumour markers, tuberculosis testing and bone marrow biopsy) help in establishing an accurate diagnosis. The imaging findings are nonspecific and the nodules may vary in size from a few millimetres up to 2 cm in diameter. They maybe hypoechoic on ultrasound and hypoattenuating on CT. They are of low signal intensity on T1-weighted sequences and vary from hypointense to hyperintense on the T2-weighted sequences. The nodules show no enhancement or show ring enhancement after contrast study. Healed granulomas are seen as scattered, discrete, small calcifications in an otherwise normal spleen. With increasing number of immunocompromised patients, such as oncology patients undergoing aggressive chemotherapy, critically ill patients in intensive care units, injection drug abusers, and HIV-infected patients, there is rise in the incidence of splenic abscess. For splenic abscess to develop, it may require the presence of bacteremia (sepsis and septic emboli) and altered splenic architecture, which is caused secondary to a haematoma, infarct, or sickle cell disease. Abscesses can also develop from the extension of infection from adjacent organs, like in cases of infected pancreatitis and perinephric abscess, which are less common. In about 20% of cases, cause or source could not be discovered. Abscesses can be unilocular or multilocular, solitary or multiple, depending on the causative organisms and patient’s immunity. Mortality ranges from 15% in otherwise healthy patients with unilocular splenic abscess to 80% in immunocompromised patients with multiple abscesses. Abscesses which are multilocular, gas-containing or due to gram-negative bacillus show poor prognosis. A pyogenic abscess is usually solitary, unilocular/multilocular, but it can also be multifocal. These are uncommon and are reported in 0.14%–0.70% of large autopsy series. Most common presentation is fever (92%), left upper abdominal pain (77%) and leucocytosis (66%). Other symptoms are left pleural effusion and splenomegaly. It can be solitary or multiple, unilocular or multilocular. The appearance of splenic abscess depends on the stage of development. In the early stages of the disease, an abscess appears as an ill-defined mass later developing into a complex collection with septations, debris and sometimes gas. In later stages, after a capsule is developed, the lesion becomes well defined. Left pleural effusion is seen in around 42% of cases. Left lung base atelectasis in 20% of cases and splenomegaly is seen. Abscess with air can be identified based on the presence of extraluminal gas or air–fluid levels in the left upper quadrant. Ultrasonography has a sensitivity of 75%–98% in detecting splenic abscess. This is a very useful modality in patients who are bedridden, with renal impairment and for assessment of small splenic lesions (Fig. 9.22.2.1). Ill-defined hypoechoic mass mimicking a mass is seen in the early phase of the infection and a complex mass with internal septations, debris, rarely acoustic shadowing caused by gas is seen in later stages. The capsule appears as a thin hyperechoic rim. It is noninvasive, speedy, and a modality with high sensitivity (92%–98%). It helps in differentiating unilocular from multilocular lesions and, gives exact anatomical localization and information about perisplenic area, which later helps in planning-guided management procedures. An abscess is seen as a hypodense collection or complex cystic lesion with enhancing rim on the postcontrast study (Fig. 9.22.2.2). CT may help in identifying concurrent areas of infection and, thus, the source of infection. On CT, an abscess may look similar to an infarct, haematoma, or neoplasm. An infarct lacks mass effect, unlike abscess or tumour (Fig. 9.22.2.3). The presence of gas within the lesion is diagnostic of abscess; however, this finding is rare. Splenic abscesses show low signal on T1-weighted images and high signal on T2-weighted images compared with the normal spleen. Minimal to intense peripheral enhancement is seen after IV administration of gadolinium. But MRI is not routinely done, as many patients are clinically unstable. This plays a limited role in the detection and localization of splenic abscess. A nonspecific filling defect is seen on a 99mTc-sulphur colloid scan within spleen if the size is more than 2 cm in diameter. Normal inherent splenic activity on 67Ga scans and 111In-labelled leukocytes scans obscures any kind of inflammatory focus in the spleen, giving rise to false-negative examination. 67Ga scans are nonspecific as tracer uptake can be seen in neoplastic conditions such as lymphoma. Detection and characterization of the abscess are better done if 99mTc-sulphur colloid scan is done prior to 111In-labelled leukocytes scan. FDG PET/CT can also detect the site of infection but not used in the diagnosis of splenic abscess. Early diagnosis of an abscess is prompted for a better outcome. Based on laboratory findings, appropriate antibiotic therapy initiated. USG or CT-guided percutaneous aspiration is tried if the abscess is less than 5 cm in size, and for larger abscesses, percutaneous drainage procedures are done preserving the spleen. Splenectomy is reserved for complicated infections. Fungal abscesses are usually seen in immunocompromised patients with neutropenia such as patients with acquired immunodeficiency syndrome (AIDS), on chemotherapy and immune suppressive agents, and with lymphoproliferative disorders. With the advent of aggressive chemotherapy, the incidence of splenic fungal infections has increased. Approximately 7% of patients with acute leukaemia have hepatosplenic fungal infections. Multiple microabscesses are common. Hepatosplenic candidiasis lesions are very small, and before the advent of CT and MRI, their detection was too difficult. They measure in the range of 5–10 mm in diameter; however, they may be even miliary (<5 mm). Infections are mostly caused by candida species. Other organisms are aspergillus, Histoplasma and Cryptococcus. Imaging characteristics depend on the stage and severity of neutropenia. In cases of chronic disseminated infections with poor neutrophil counts, sometimes lesions are not visible as the host fails to produce an immune response. Lesions become apparent once neutrophil counts improve. Patients presenting with fever and splenomegaly are not improving on conventional antibiotic therapy, and with associated immunocompromised status, fungal infections are to be considered. No single imaging is specific or sensitive in establishing the diagnosis. Serial imaging is needed to detect hepatosplenic involvement; however, tissue diagnosis is the gold standard. Four different patterns are seen in hepatic candidiasis. Multiple, small, homogeneous hypoechoic nodules are the most common finding. Less commonly wheel-within-a-wheel appearance (i.e. the target sign) is seen in the early stage of infection. The first wheel is the peripheral hypoechoic zone of fibrosis and an echogenic second wheel of inflammatory cells around a central hypoechoic nidus containing necrosis and fungal elements. Later these lesions evolve into Bull’s eye lesion where there is a central echogenic focus surrounded by a hypoechoic halo. Lesions become small and hyperechoic with various degrees of posterior acoustic shadowing, with or without calcification, or they may even disappear later in the course of the infection. Splenic nodules are hypodense lesions measuring in the range of 5–10 mm. Only 30% of cases are detected in noncontrast studies. They often have low attenuation, although there can be a focus of high attenuation or a ‘wheel-within-a-wheel’ pattern. Peripheral ring of enhancement is noted in around 70% of cases in the arterial phase of CT, and it is said to disappear on the portal venous phase. There is no significant difference in the detection of the lesions between arterial and portal venous phases of contrast studies. MRI is said to be the investigation of choice for diagnosis of hepatosplenic candidiasis and is superior to CT and USG, especially when short-time inversion recovery sequence is used. MR imaging is also used to monitor the response to antifungal therapy. Based on MRI findings different phases of hepatosplenic candidiasis are described. Lesions are small, measuring less than 1 cm in diameter. They show mildly hypointense on the T1-weighted images and markedly hyperintense on the T2-weighted images. In 2 weeks to 3 months, acute lesions are transformed into subacute lesions. The lesions reveal mildly increased signal on both T1- and T2-weighted sequences. On all sequences, a peripheral ring of very low signal intensity is seen. Following the gadolinium administration, the central region of the lesions show enhancement; however, the peripheral ring continues to have low signal intensity, making them more obvious. It takes from 3 months to more than 1 year for the healed fungal foci composed of fibrous tissue of differing densities and degrees of vascularization to appear on MRI. The lesions measure 1–3 cm, become irregular, and the central area disappears. Chronic healed fungal abscesses have low signal intensity on T1-weighted images and are isointense to mildly hyperintense on T2-weighted images. No perilesional dark ring is noted. These lesions are moderately hypointense on early postgadolinium images and minimally hypointense on delayed postgadolinium images. Mycobacterial tuberculosis infections are mainly seen in immunocompromised patients. Spleen involvement is seen in disseminated, miliary infection in about 80%–100% of autopsy cases; however, it is documented less on imaging. Splenic tuberculosis is seen in the form of nodules measuring ∼ 0.2 and 1 cm in diameter. The macronodular form is very rare (Fig. 9.22.2.4). Hypoechoic pattern is seen. Hypoattenuating nodules with lymphadenopathy noted. Depending on the different stages of the tubercular process, intensities on T1- and T2-weighted images vary. On contrast-enhanced T1-weighted images, lesions most often show peripheral enhancement less commonly gradual peripheral enhancement with complete filling is noted. It is also called as Mycobacterium intracellulare infections. These are also typically seen in immunocompromised patients. Splenomegaly with multiple, low attenuation nodules is seen. Associated findings such as hepatosplenomegaly, diffuse jejunal wall thickening and enlarged lymph nodes are seen. Lymph nodal involvement is homogeneous as opposed to Mycobacterium tuberculosis infections. Focal splenic or hepatic lesions are noted in about 30% of patients with HIV who are infected with M. tuberculosis but are rarely noted in those infected with M. avium intracellulare. Marked splenomegaly is more common in M. avium intracellulare than in M. tuberculosis infection. It is a common opportunistic infection in patients with HIV. Extra pulmonary involvement manifested in the form of necrotizing granulomas. Spleen involvement is an incidental finding. When treated the nodules may enlarge and become progressively calcified in a rim like or punctate fashion. Disseminated infection can show punctate calcification in adrenal glands, pleural or peritoneal effusions, that subsequently calcify. Splenomegaly with small hypoechoic lesions with cystic components or tiny, highly reflective, nonshadowing foci or calcified granulomas. The nodules are hypodense. Calcified granulomas are seen in later stages. This is a self-limited disease caused by bartonellae henselae. Hepatosplenic involvement is rare. This disease is to be considered when a patient presents with abdominal pain, fever of unknown origin, and multiple hypodense lesions in the liver and spleen. It is also called as echinococcosis, a disease seen in endemic regions and in patients emigrated from endemic areas. The causative organism is a zoonotic tapeworm echinococcus granulosus. The most commonly involved organs are the liver, lung, peritoneum, kidney, brain, mediastinum and heart. Spleen is rarely primarily involved. Spleen is involved in approximately 2% of cases of abdominal hydatidosis. Spleen involvement is seen in case of an extensive abdominal hydatidosis, like after the rupture of liver hydatid cyst or in cases of systemic dissemination of infection. Larvae of the tapeworm incite an inflammatory response after its entry into the target tissue in the host where it forms a hydatid cysts consisting of three layers. Peri cyst is an outer layer of modified host cells forming a dense and fibrous layer. The middle layer laminated layer and an inner germinal layer made up of daughter cysts, also called as brood capsules. Scolices develop in the brood capsules. The laminated and germinal layer forms the true cyst wall. After the parasites die, the cysts become inactive, may calcify, and undergo fibrosis. Patients present with abdominal pain, splenomegaly and fever. The diagnosis is to be suspected in all patients from endemic areas presenting with a splenic cyst. A World Health Organization Working Group on echinococcosis has a standardized sonographic classification of echinococcal cysts (Table 9.22.2.1). CE, cysticechinococcosis; CL, undifferentiated simple cystic lesions. Radiological appearances depend on the natural aging process of the cyst. Classic imaging characteristics are lost when the cyst becomes secondarily infected. A ring-like or curvilinear calcification noted in the left hypochondriac region. This may represent the calcification of pericyst in later stages of the disease. Ultrasound is an easy and most commonly used for evaluation of a suspected case of abdominal echinococcal disease. It is used for the diagnosis, planning the management and follow-up. Shifting of brood capsules within the cyst causes fine internal echoes, that is ‘The snowflake sign’ (hydatid sand). Detached laminated endocysts may be seen within the cysts, that is ‘The. Waterlily sign’. Mother cysts may be partially /completely filled by daughter cysts. Calcifications with posterior acoustic shadowing are seen in later stages of the disease. CT and MRI are indicated for widespread disease and complications and for planning percutaneous therapy and surgery. Complications of a hydatid cyst include compression of the adjacent structures, cyst rupture, secondary infections, anaphylaxis and fistulization to surrounding structures. A hydatid cyst may be seen as a well-defined, hypoattenuating cystic lesion with fluid-attenuation values and a distinguishable wall. CT is more sensitive for subtle wall calcification. May appear hyperdense because of debris, hydatid sand and inflammatory cells (Fig. 9.22.2.5). In CE 2 cysts, peripherally arranged daughter cysts, completely fill them other cyst. The daughter cysts show lower attenuation than the mother cyst. Type CE 3 lesions are round or oval with occasional calcification and daughter cysts. They usually show high attenuation. Type CE 4 cysts are complex masses. Intravenous contrast administration reveals enhancement of septa and cyst wall. Type CE 5 cysts are complex cystic or solid-looking lesions, and they can be partially or completely calcified. The simple cysts show low signal on the T1-weighted images and markedly hyperintense on the T2-weighted images. The rim sign which is a low signal intensity rim well seen in T2-weighted MRI images is characteristic of hydatid disease. Enhancement of septa and cyst wall is noted after the intravenous administration of gadolinium. On T1- and T2-weighted images, the maternal matrix appears hyperintense relative to the daughter cysts. The collapsed parasitic membranes may show serpent sign or snake sign, which are hypointense on all sequences. Hypointensity on T1- and T2-weighted images are seen in type CE 4 and CE 5 cysts. The imaging findings combined with the clinical, immunologic testing and epidemiologic results, usually provide the correct diagnosis. Seen in cases of tuberculosis, histoplasmosis, less commonly in Pneumocystis jirovecii pneumonia and brucellosis. These are incidental findings usually preceded by hypoattenuating microabscesses. Microabscesses heal with or without preceding regression. More than five calcifications usually represent histoplasmosis, whereas less than five represent tuberculosis. A ring-like calcification is seen in Pneumocystis carinii pneumonia. Any granulomatous infection is more likely to involve other organs such as the liver, kidneys and lymph nodes. Hydatid cysts can show ring-like or curvilinear calcification (Fig. 9.22.2.6). SPLENOMEGALY AND HYPERSPLENISM Splenomegaly is often a nonspecific finding, and there are many causes for splenomegaly. The spleen can grow into large sizes towards right iliac fossa, crossing midline. On average adult spleen measures <11 cm in length,<7 cm wide,<5 cm thick, and weighs approximately 150 g (80–300 g). The size and weight of the spleen decrease with advancing age. Spleen size is significantly influenced by the sex and body height of an individual. Chow et al. assessed 1230 healthy volunteers and found that spleen length and volume were significantly and independently associated with sex, body height and weight. Taller and heavier male individuals have larger and longer spleens. In one study the spleen length in 6% of women, and 26% of men exceeded a strict upper limit of normal of 12 cm. Rosenberg et al. established an upper limit of standard splenic length of 12 cm for girls and 13 cm for boys (≥15 years). Massive splenomegaly is defined as a spleen size greater than 18 cm, is less common and includes haematological disorders and infections (Table 9.22.3.1).
9.22: Imaging of spleen and splenic pathologies
Introduction
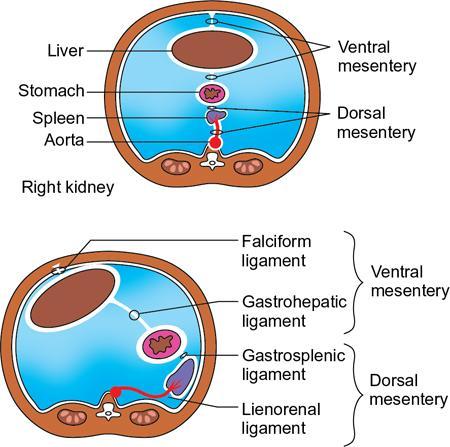
Embryology
Wandering spleen
Aetiology
Congenital
Acquired
Clinical features
Imaging features
USG
CT
Features of torsion
MRI
Nuclear imaging
Angiography
Therapy
Accessory spleen
Clinical features
Imaging features
USG
CT
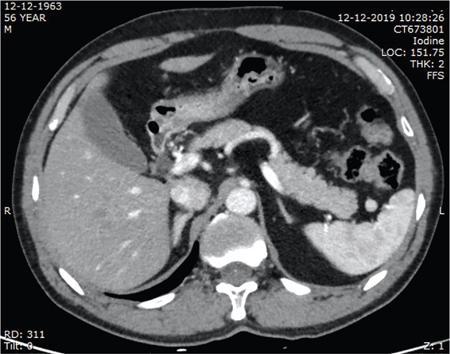
MRI
Nuclear imaging
Differential diagnosis
Treatment
Splenic gonadal fusion
Continuous form (55%)
Discontinuous form (45%)
Imaging features
USG
Scintigraphy
Heterotaxy syndromes
Asplenia syndrome (ivemark syndrome)
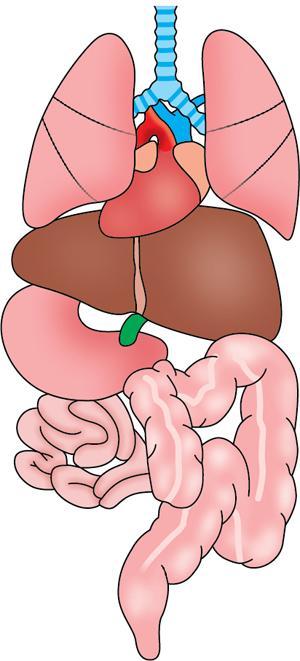
Aetiology
Clinical features
Imaging features
Conventional radiography
CT
Differential diagnosis
Polysplenia syndrome
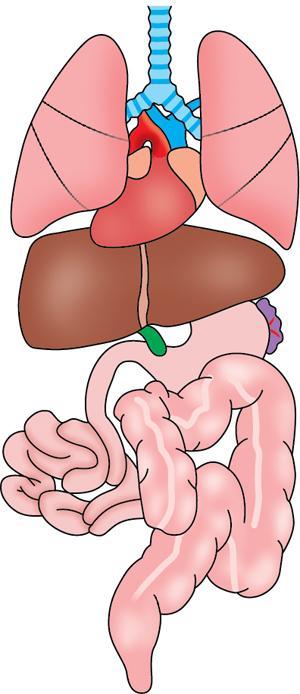
Aetiology
Clinical features
Imaging features
Conventional radiography
CT
Treatment
Splenic infections
Causes of splenic nodular lesions (the lesions less than 2 cm)
Splenic nodular disease
Abscess
Common causative organisms
Pyogenic abscess
Clinical features
Radiological findings
Plain radiograph
USG
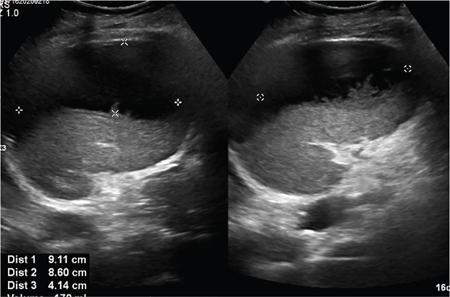
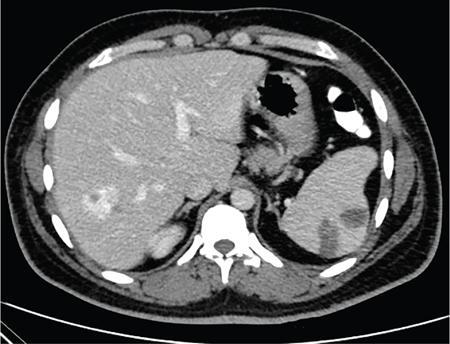
CT
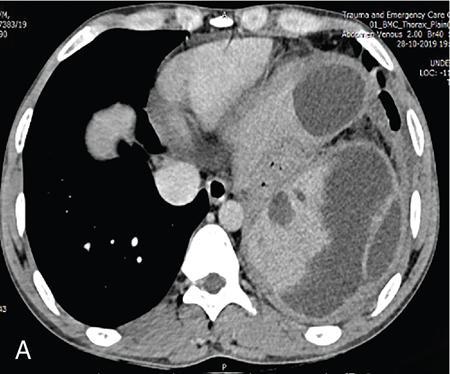
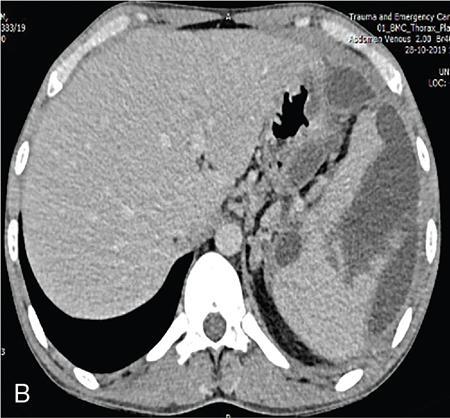
MRI
Nuclear medicine
Treatment
Fungal abscess and microabscess
Clinical features
Radiological features
USG
CT
MRI
Acute
Subacute
Chronic
Tuberculosis
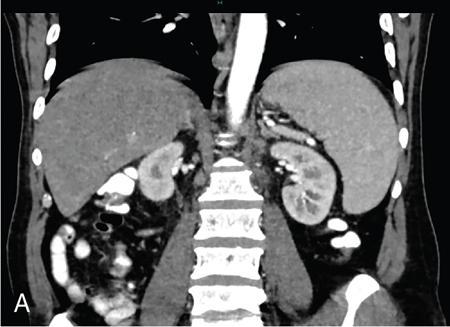
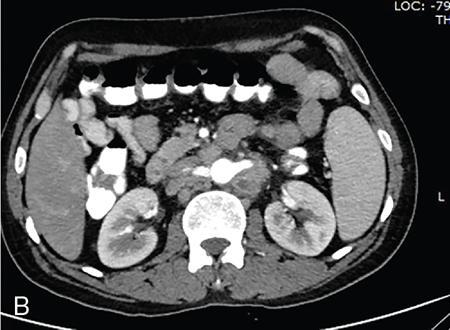
USG
CT
MRI
Mycobacterium avium complex infection
Imaging
Pneumocystis jirovecii pneumonia
USG
CT
Cat scratch disease
Hydatid disease
Pathogenesis
Clinical features
Radiological features
Classification
Description
CL
Simple cystic lesion; cyst wall not visible.
CE 1
Unilocular simple cyst with visible cyst wall; may show ‘hydatid sand’, fine internal echoes due to shifting of the brood capsules (i.e. snowflake sign).
CE 2
Multiseptate, multivesicular cyst; daughter cysts may partially or completely fill the mother cyst; septations may produce wheel-like structures; contained daughter cysts may produce a rosette-like or honeycomb-like structure.
CE 3
Unilocular, anechoic content with the detachment of the laminated membrane from the cyst wall, visible as a floating membrane (i.e. water lily sign), or complex masses caused by unilocular cysts containing daughter cysts and echoic areas.
CE 4
Heterogeneous hypoechoic or inhomogeneous degenerative contents; no daughter cysts; degenerating membranes may produce a ball of wool sign.
CE 5
Cysts with a thick, calcified, arch-shaped wall; lesions are partially or completely calcified.
Plain radiographs
USG
CT
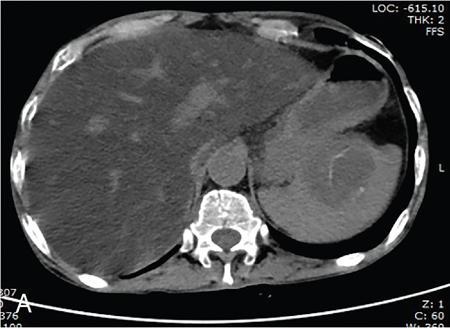
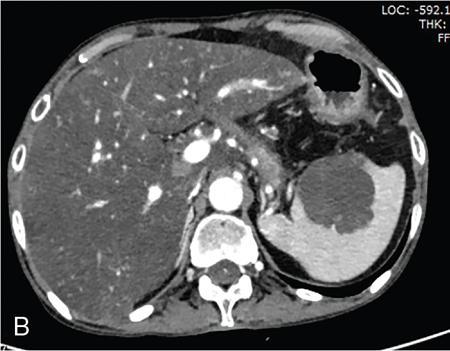
MRI
Differential diagnosis for splenic abscess
Infections causing splenic calcifications
Calcified granulomas
Splenomegaly
Splenomegaly (<18 cm)
Massive Splenomegaly (>18 cm)
HAEMATOLOGIC
RHEUMATOLOGIC
INFECTIOUS
CONGESTIVE
INFILTRATIVE
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








