Do you have trees in your yard? If you see a diseased area on a branch of a dogwood tree, you will likely also look up and down that branch to see the extent of the process. Then you are probably going to look at the other branches to see if there are other areas involved. And because whatever is in the ground or air that resulted in the disease could also affect your other dogwood trees, you’ll probably look around the yard to see if they might also be having a problem.
YES! You found a highly suspicious lesion on mammography. Nice work! But wait—there’s more. Our work is not done here. After identifying a lesion on imaging that is very suggestive of breast cancer, our job shifts from detection to evaluating the extent of disease and the staging of the cancer. Let’s thoroughly evaluate the patient so that we make the surgeon’s job easier. Oh, and don’t forget about that other breast!
For women with a very suspicious (BI-RADS 4C) or highly suspicious (BI-RADS 5) finding on mammography or ultrasonography (US), evaluation of the extent of disease and the staging can occur at the initial diagnostic appointment. If biopsy of a less suspicious lesion results in a cancer diagnosis, then imaging evaluation of the extent of disease and the staging should be performed prior to surgical intervention. In women with recently diagnosed breast cancer, the incidence of contralateral cancer is 0.1% to 2% on mammography and 3% to 5% by magnetic resonance imaging (MRI), so careful review of the mammogram and consideration of breast MRI are important.
Imaging contributes to patient care by guiding surgical management and providing information about staging. Surgical management of the breast is determined by accurate assessment of the size and location of the cancer, and identification of multifocal or multicentric disease. Imaging affects staging by identifying invasive tumor for women with ductal carcinoma in situ (DCIS), accurately measuring tumor size, evaluating regional lymph nodes, and identifying skin or chest wall involvement. Imaging to identify systemic disease is also important in staging, but will not be reviewed here.
Size and Extent of the Primary Tumor
Invasive Tumor Size
In the TNM system for staging breast cancer, the T is based on the size of the largest invasive component. Although measurement of the tumor in three dimensions is helpful for monitoring change for women undergoing neoadjuvant chemotherapy, only the longest dimension is used in staging. When there is more than one invasive focus, only the largest is considered in staging. Tumors are classified as Tis (in situ), T1 (≤20 mm), T2 (21-50 mm), or T3 (>50 mm). T1mi refers to microscopic invasion of less than 1 mm.
Tumor size is measured at histologic examination. However, there are times when the size on imaging may be used, such as when the tumor is very small and mostly removed at core biopsy or in the setting of neoadjuvant chemotherapy. For invasive carcinoma, US size that includes the echogenic rim correlates best with histologic size ( Fig. 17-1 ). Measurements of invasive carcinoma on MRI correlate best with size at histologic evaluation. This is especially true for invasive lobular carcinoma (ILC) ( Fig. 17-2 ).


Associated Ductal Carcinoma in Situ
Now that you have identified a mass that is suspicious for invasive carcinoma, let’s look to see whether there is also associated DCIS. Mammography is a good place to start. Breast cancer typically affects a single ductal system. If we recall the tree branch analogy from Chapter 5 , Breast Anatomy and Physiology, we will want to focus our attention on the ductal system where the mass is located. We will especially want to look for calcifications between the mass and the nipple, as well as between the mass and the chest wall ( Fig. 17-3 ). Recall that one ductal system can extend over a wide area in the breast, so even calcifications that are not immediately adjacent to an invasive carcinoma may represent DCIS.

MRI can also be very helpful in assessing the extent of DCIS associated with a primary invasive cancer. The associated DCIS will usually present as nonmass enhancement in the same ductal distribution ( Fig. 17-4 ).

Extent of Ductal Carcinoma in Situ
The extent of calcifications on mammography typically correlates well with DCIS that is high grade, but low-grade DCIS often has more extensive noncalcified disease.
When the primary lesion presents as suspicious calcifications, other calcifications that are morphologically similar are likely to represent additional disease ( Fig. 17-5 ). Calcifications of differing morphologic appearance may also need to undergo sampling if they are also suspicious based on morphologic appearance, distribution, or change over time ( Fig. 17-6 ).


This does not mean that you must biopsy every cluster of calcifications in the breast! If the calcifications are of similar morphologic appearance, do a biopsy of the most suspicious finding first (see Fig. 17-5 ). If the biopsy shows cancer and the patient desires mastectomy, doing a second biopsy will not alter her management. On the other hand, if the patient desires breast-conserving therapy, an additional biopsy may be needed to establish the extent of disease. In our practice, a second biopsy of the ipsilateral breast is not typically performed until the results of the biopsy showing cancer have been discussed with the patient and she has seen a breast surgeon to discuss surgical options.
DCIS with Associated Invasive Disease
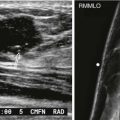
When the primary lesion is malignant-appearing calcifications and you are suspecting DCIS, finding an invasive component changes the stage from 0 to I. Providing the patient and surgeon with the higher stage of disease is helpful because the patient may then need a sentinel lymph node biopsy. An invasive component may be suspected when there is density that looks like a puff of smoke overlying the calcifications ( Fig. 17-7 ). US is useful in evaluating these patients. An invasive focus will appear as a hypoechoic mass, whereas DCIS will appear as an expanded duct with calcifications (see Fig. 17-7 ). US is also useful when malignant calcifications are present in dense breast tissue that could be masking an invasive carcinoma ( Fig. 17-8 ). US does not need to be performed for every patient with suspicious or malignant calcifications. If an associated mass is not definitely identified by US, stereotactic biopsy is preferred over ultrasound-guided biopsy because it is less likely to show only atypical ductal hyperplasia because of undersampling.


Multifocal Invasive Carcinoma
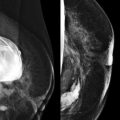
Small masses or asymmetries in the same ductal distribution as the primary lesion should be viewed with suspicion. When additional invasive disease is separated by less than 4 cm, the cancer is considered to be multifocal ( Fig. 17-9 ). Although different criteria are used to determine whether carcinomas should be classified as multifocal (vs. multi centric ), the less-than-4-cm measurement correlates well with a lower chance of local recurrence when compared with more extensive lesions ( Box 17-1 ).

- •
If there is a BI-RADS 5 lesion present, start with a biopsy there first to establish a diagnosis.
- •
If there are two different BI-RADS 4 lesions, biopsy of each may be indicated because one might be cancer and the other benign.
- •
Once a cancer diagnosis is made, give the patient a chance to digest the news and think about her surgical options.
- •
If the biopsy shows invasive carcinoma and the patient desires mastectomy, no further breast biopsies are needed.
- •
For women desiring breast-conserving therapy
- •
If a second area is present but within a centimeter or so from the primary carcinoma, the surgeon can likely remove the finding during the primary resection. Review the case with your surgeon.
- •
If the associated disease is more extensive, choose an area for biopsy that is as far away from the known cancer that is likely to yield a cancer diagnosis.
- •
US is very useful to identify multifocal invasive carcinoma. Remember our tree? With the transducer oriented in the radial plane, sweep the area between the cancer and the nipple and then the cancer and the periphery of the breast to identify additional lesions within the branches of the involved ductal system (see Fig. 17-9 ).
MRI is very sensitive for detecting additional invasive breast carcinoma ( Fig. 17-10 ).

Multicentric Carcinoma
Additional ipsilateral cancer that is more than 4 cm from the primary lesion is considered multicentric. This may or may not be identified in a different quadrant. Satisfaction of search is difficult to overcome. Ignore that really concerning finding and really look at the rest of that breast (see Fig. 17-6 ). Diagnosis of multicentric carcinoma is generally a contraindication for breast-conserving therapy, though it is not an absolute contraindication.
Skin or Chest Wall Involvement
Remember the T classification earlier? We only gave you T0 to T3, but there is also a T4 category that is defined by involvement of the skin or chest wall with tumor. Skin involvement is defined as skin nodules or ulceration ( Fig. 17-11 ), not just dermal invasion. Chest wall involvement is invasion into the intercostal muscles or ribs. Invasion of the pectoral muscles may influence surgical or medical management, but is not considered chest wall invasion. Inflammatory cancer is also considered T4.

If you are a “lumper,” then skip this paragraph. If you are a “splitter,” then you’ll want to know the T4 subcategories. T4a is invasion of the chest wall. T4b is tumor associated with skin ulceration, ipsilateral skin nodules, or skin edema that does not meet criteria for inflammatory breast carcinoma. T4c is findings of both T4a and T4b. T4d is inflammatory carcinoma.
Inflammatory carcinoma is most commonly associated with invasive ductal carcinoma—not otherwise specified (IDC-NOS). It is not associated with DCIS alone. The diagnosis of inflammatory carcinoma may be clinical (red, swollen breast in a patient with invasive carcinoma) or histologic (punch biopsy of the skin showing invasion of the dermal lymphatics).
Paget Disease Is Not T4
Although Paget disease is tumor ulceration of the skin of the nipple, it is not considered T4 disease. If Paget disease is only associated with DCIS, then it is stage Tis. If a woman with Paget disease has an associated invasive carcinoma, then the T status is based on the size of the invasive carcinoma.
Contralateral Breast Cancer
The incidence of synchronous contralateral breast cancer is 0.1% to 2% by mammography and 3% to 5% by MRI ( Fig. 17-12 ). Ignore the ipsilateral breast for a moment and really focus on the other breast. Bilateral breast cancer diagnosed at the same time is considered synchronous, whereas diagnosis that occurs at least 6 months apart is considered metachronous.

Evaluating Regional Lymph Nodes
Lymph from the breast drains mostly to the axillary lymph nodes, though some drains to the internal mammary lymph nodes. About one third of breast cancers will have metastasized to the regional lymph nodes or other surrounding tissues at the time of diagnosis.
Axillary lymph nodes are divided into levels with relationship to the pectoralis minor muscle. Lymph nodes that are lateral, posterior, or medial to the pectoralis minor muscle are level I, II, and III, respectively. Lymph nodes located in the space between the pectoralis major and minor are interpectoral (Rotter) nodes and are managed as level II lymph nodes.
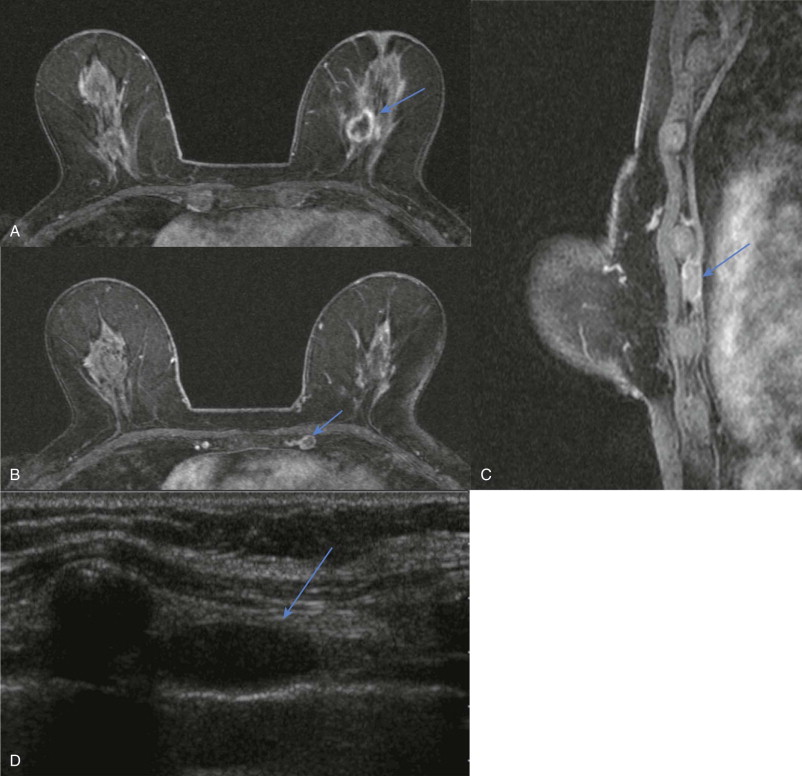
Level I axillary lymph nodes are seen on mammography, US, and breast MRI (see Fig. 17-9 ). Level II and III axillary and supraclavicular lymph nodes are seen to varying degrees on US and breast MRI. Internal mammary lymph nodes are most commonly found in the second or third intercostal spaces and can be identified on US or MRI ( Fig. 17-13 ). Small normal internal mammary lymph nodes may occasionally be seen on MRI. If seen on US, an internal mammary lymph node is usually abnormal.