▪
Introductory Considerations
Diagnostic imaging is used in the detection of thoracic infections, identifying complications associated with infection, and differentiating noninfectious from infectious diseases. Chest radiography and computed tomography (CT) comprise the primary modalities used in diagnosis, with ultrasound, nuclear medicine, and magnetic resonance imaging (MRI) used in select cases. Suggesting a specific organism is generally not possible in the immunocompetent patient presenting with thoracic infection. Imaging is usually able to suggest a specific pathogen in patients with known human immunodeficiency virus (HIV) infection or in patients with other forms of immunosuppression, such as bone marrow transplant (BMT) recipients. Thoracic infection is broadly categorized into three separate categories:
- 1.
Infection in the immunocompetent host
- 2.
Infection in the immunocompromised host with HIV
- 3.
Infection in the immunocompromised host without HIV
Epidemiology
Pneumonia and influenza are the eighth leading cause of death in the United States. An estimated 1.23 million hospital admissions yearly are attributable to pneumonia. Pneumonia affects all ages and genders, but tends to be more severe and deadly in the very young and very old. Pneumonia is caused by bacteria, viruses, fungi, and parasites, as well as following the inhalation of food, liquid, gas, and dust.
Symptoms
Bacterial pneumonia may have a rapid and sudden onset with symptoms of high fever, productive cough, sweats, and shaking chills. Occasionally, bacterial pneumonia may have a gradual course with less severe symptoms. The symptoms of influenza and viral pneumonia are similar and include fever, dry cough, headache, weakness, and muscle aches.
Pathogenesis
Pathogens may gain access to the respiratory system in a variety of ways, ultimately leading to infection. The most common route of spread is via the tracheobronchial tree, usually by inhalation of aerosolized organisms or, less commonly, aspiration of secretions. Less common routes are via direct spread from infected mediastinal or hilar lymph nodes or extension of an infection from an adjacent organ, such as a liver abscess or perforated esophagus. Infection may also gain access to the lungs through the pulmonary vasculature occurring with infective endocarditis, bacteremia, or infected central venous catheters.
Most hospital-acquired infections occur via aspiration of oropharyngeal pathogens or leakage of secretions containing pathogenic bacteria around the endotracheal tube cuff. Infected biofilm surrounding the endotracheal tube may also be important in the pathogenesis of ventilator-associated pneumonia. Inhalation or direct inoculation of organisms into the lungs, hematogenous dissemination, and bacterial spread from the gastrointestinal tract are less common patterns of spread in hospitalized patients.
Diagnosis
Pneumonia is determined to be present clinically when a chest radiograph shows new or increasing consolidation and clinical evidence that the consolidation is of infectious origin. Clinical features suggestive of infectious origin of consolidation include fever (≥38°C), purulent secretions, and leukocytosis or leucopenia. Pathogen-directed therapy is advised and may be obtained by Gram staining and culture of sputum, blood cultures, testing for Legionella and pneumococcal urinary antigens, and multiplex polymerase chain reaction (PCR) assays for Mycoplasma pneumoniae, Chlamydia pneumoniae, and respiratory viruses, as well as other pathogens, as indicated by the patient’s exposure history or specific risk factors. Procalcitonin is a peptide precursor produced by cells in the intestine and lungs and is a useful marker to differentiate bacterial from nonbacterial infections and other noninfectious causes of inflammation. A procalcitonin level does not generally rise in patients with viral or noninfectious causes of inflammation. The serum procalcitonin concentration is a helpful marker of bacterial sepsis, and a low concentration (<0.1 µg/L) can support a decision to withhold or discontinue antibiotics.
In one large study of 2706 adults diagnosed with pneumonia, almost one-third initially had a normal chest radiograph. In these patients, the absence of consolidation should not supersede clinical judgment and empiric therapy because many of these patients will develop bacteremia, with associated increased mortality.
Definitions
Pneumonia is broadly divided into infection that is acquired in the community and infection acquired by hospital or nursing home patients. Multidrug-resistant (MDR) organisms are more common in hospital or health care–associated pneumonia.
Community-Acquired Pneumonia
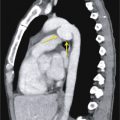
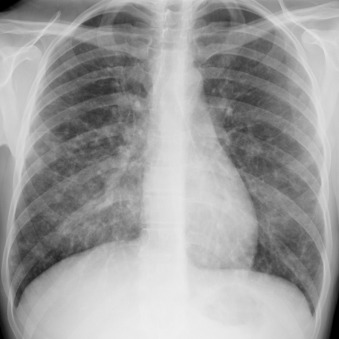
Community-acquired pneumonia (CAP) is pneumonia manifesting in a patient who has not been recently hospitalized and who has had no regular exposure to the health care system ( Fig. 16.1 ). Pneumococcus is the most common bacterial pathogen responsible for CAP, but its incidence has been declining with the use of the pneumococcal vaccination and with decreased rates of cigarette smoking. Other causes of CAP include Haemophilus influenzae , Staphylococcus aureus , Moraxella catarrhalis , Pseudomonas aeruginosa , viruses, and other gram-negative bacteria. Despite exhaustive testing, no organism is found in about 50% of cases.
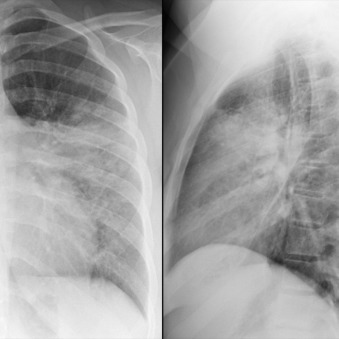
Hospital-Acquired Pneumonia
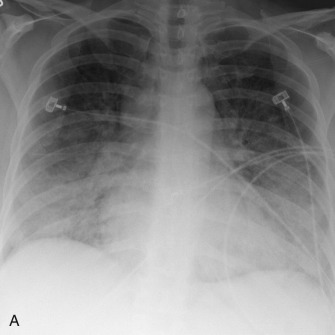
Hospital-acquired pneumonia (HAP) is pneumonia manifesting 48 hours or more after a hospital admission, usually caused by bacterial pathogens ( Fig. 16.2 ). HAP is associated with a high morbidity and mortality and is the second most common nosocomial infection in the United States, trailing only urinary tract infection. HAP increases a hospital stay by 7 to 9 days and increases the total cost by approximately $40,000/patient. Common pathogens include gram-positive cocci such as S. aureus and gram-negative organisms, including P. aeruginosa , Klebsiella pneumoniae , Escherichia coli , and Actinobacter spp. Rates of polymicrobial infection appear to be rising, especially in patients with acute respiratory distress syndrome (ARDS).
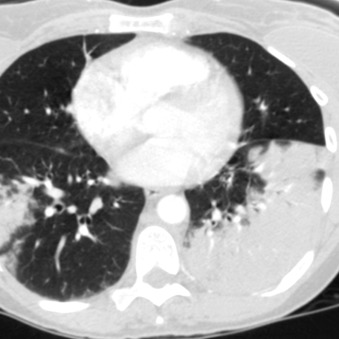
Health Care–Associated Pneumonia
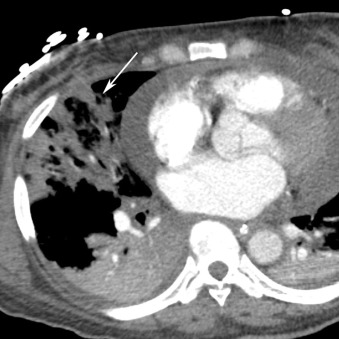
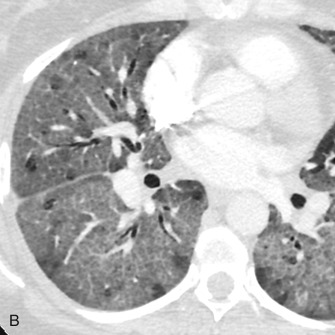
Health care–associated pneumonia (HCAP) is pneumonia manifesting in a patient who was recently hospitalized for 20 or more days in the preceding 90 days of infection, resided in a nursing home or long-term care facility, received recent intravenous antibiotic therapy, chemotherapy, or wound care within the preceding 30 days of infection, or attended a hospital or hemodialysis clinic ( Fig. 16.3 ).
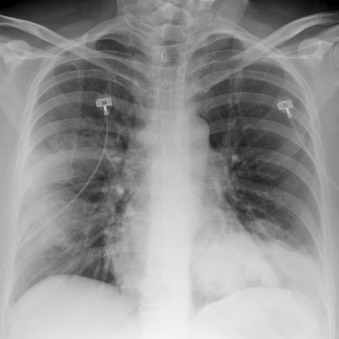
Ventilator-Associated Pneumonia
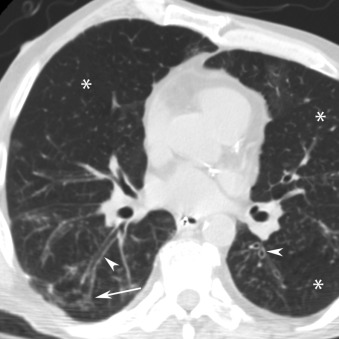
Ventilator-associated pneumonia (VAP) is pneumonia manifesting 48 to 72 hours following recent endotracheal intubation ( Fig. 16.4 ). VAP occurs in about 9% to 27% of intubated patients, with about 50% of all cases occurring within the first 4 days of ventilation.
▪
Patterns of Infection
Pulmonary infections may affect the pulmonary parenchyma (pneumonia), the small airways (bronchiolitis), and the central airways (tracheobronchitis). Pneumonia is often subdivided into lobar pneumonia, bronchopneumonia, and interstitial pneumonia, although differentiating among these different patterns is often difficult and usually unreliable in suggesting a specific pathogen.
Lobar Pneumonia
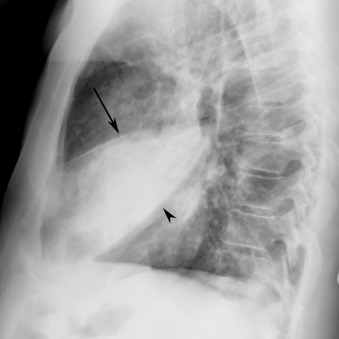
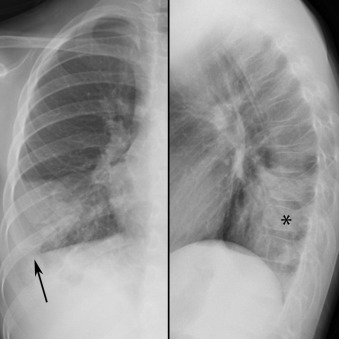
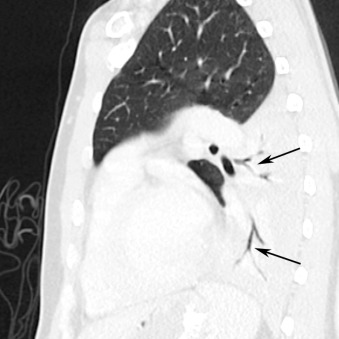
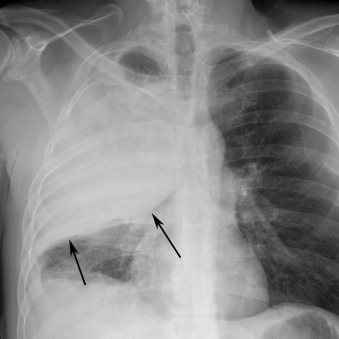
Lobar pneumonia usually starts as a unilateral peripheral opacity that progresses to homogeneous consolidation, often bounded by an interlobar fissure ( Fig. 16.5 ) or the hemidiaphragm. Lobar pneumonia is most common with infection by Streptococcus pneumoniae , K. pneumoniae , and Legionella pneumophila . Lower lobe pneumonia may manifest the spine sign on the lateral chest radiograph, which represents a paradoxic increased density of the lower thoracic spine ( Fig. 16.6 ). There are often air bronchograms ( Fig. 16.7 ) within the consolidation, and the infection tends to cross bronchopulmonary segments. It uncommonly affects the entire lobe due to the early initiation of antibiotic therapy. Occasionally, there may be copious amounts of inflammatory exudates, which cause bulging or displacement of fissures (i.e., bulging fissure sign; Fig. 16.8 ). Although typically described with K. pneumoniae , the bulging fissure sign is usually seen with S. pneumoniae owing to its greater prevalence.
Bronchopneumonia
Bronchopneumonia begins with infection of the distal airways, which eventually extends into the adjacent alveoli ( Fig. 16.9 ). Lobules are often first affected in a patchy fashion and may be initially limited to one or a few bronchopulmonary segments before progressing to multifocal bilateral consolidation. Air space nodules or acinar nodules represent infection of distal bronchioles and surrounding alveoli and manifest as 5- to 10-mm ill-defined centrilobular nodules. There are often associated branching V – and Y -shaped opacities that are referred to as the tree-in-bud sign ( Fig. 16.10 ). Bronchopneumonia is most common with infection by S. aureus , gram-negative organisms, anaerobic bacteria, and L. pneumophila .
Interstitial Pneumonia and Bronchiolitis
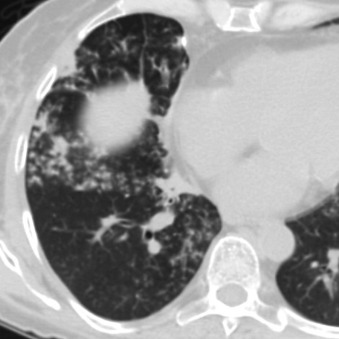
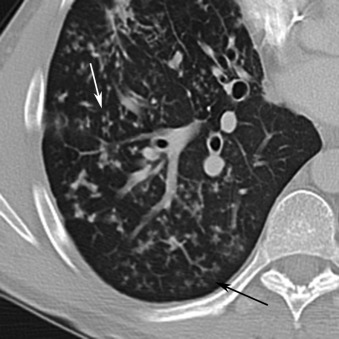
Reticulonodular and nodular opacities are the most common interstitial manifestations of pneumonia and generally are the radiographic manifestation of bronchial wall thickening ( Fig. 16.11 ) and cellular bronchiolitis on CT. The individual nodules are too small to visualize on radiography but manifest as small nodular opacities when multiple overlapping nodules summate ( Fig. 16.12 ). The radiographic appearance may mimic that of interstitial edema ( Fig. 16.13A ). On CT, interstitial pneumonia manifests as cellular bronchiolitis with tree-in-bud opacities, centrilobular nodules, bronchial wall thickening, and ground-glass opacities ( Fig. 16.14 ; see Fig.16.13B ). Consolidation is less commonly visualized. Mosaic attenuation (see Fig. 16.14 ) is common and represents air trapping caused by small airway inflammation and obstruction. Interstitial pneumonia is usually caused by atypical organisms, including viruses, mycoplasma, and Pneumocystis jirovecii .
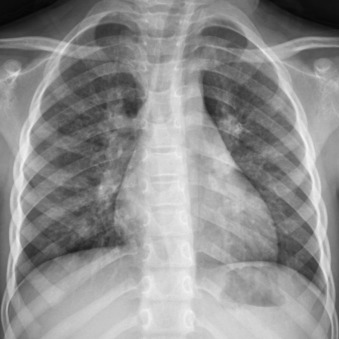
Tracheobronchitis
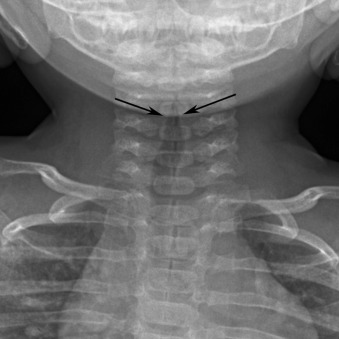
Primary infection of the trachea and central bronchi is generally caused by viruses and usually affects young children. The principle cause is croup, which manifests as the steeple sign of subglottic narrowing on anteroposterior radiography of the neck ( Fig. 16.15 ). Viral tracheobronchitis in adults is uncommonly imaged and, very rarely, will be superinfected by bacteria.
The most common manifestations of bronchitis include linear or tramlike opacities emanating from the hila on chest radiography. The primary CT manifestations include bronchial wall thickening, mosaic lung attenuation related to air trapping, and bronchial plugging. Patients are often managed conservatively in the absence of consolidation.
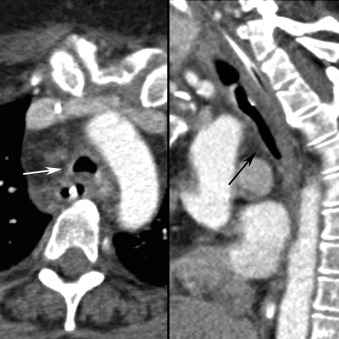
Uncommonly, Aspergillus , tuberculosis, or Candida may affect the trachea and central bronchi in an immunocompromised patient. The most common manifestations of infection include focal airway wall thickening or plaques, which may be of high attenuation. Occasionally, wall thickening is diffuse and circumferential ( Fig. 16.16 ), mimicking other diseases such as amyloidosis and polyangiitis with granulomatosis (formerly Wegener granulomatosis).
Hematogenous Spread of Infection
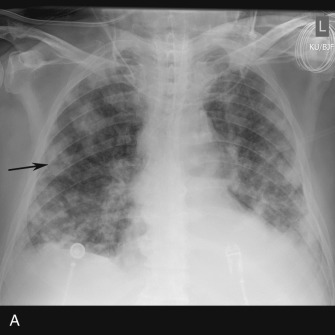
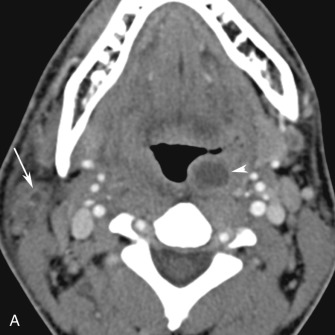
Hematogenous spread of bacterial infection usually occurs in intravenous drug users (IVDUs) or in patients with infected central catheters. The most common manifestation is multiple mid and lower lung zone poorly defined nodules and wedge-shaped consolidations ( Fig. 16.17 ). The nodules are usually located in the lung periphery and many undergo central cavitation over a period of several days. The nodules are often closely associated with pulmonary vasculature, which has been termed the feeding vessel sign (see Fig. 16.17B ). This pattern of spread is usually found with infections caused by staphylococcal species, usually S. aureus . Uncommonly, multiple septic emboli occur in patients with thrombophlebitis of the internal or external jugular vein resulting from an oropharyngeal infection from Fusobacterium necrophorum . This constellation of findings is termed Lemierre syndrome ( Fig. 16.18 ).
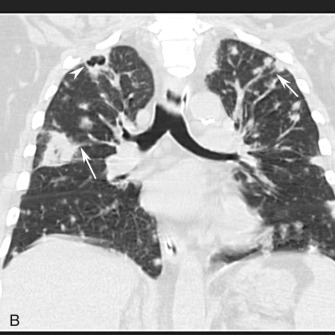

Hematogenous spread of fungal infection is uncommon and usually affects immunocompromised individuals. It manifests with a miliary pattern on radiography and CT, which represents innumerable small (<3 mm), randomly distributed pulmonary nodules throughout both lungs, often with a mid and lower lung zone predominance ( Fig. 16.19 ). Miliary spread of infection is most common with Mycobacterium tuberculosis as well as endemic fungi such as Histoplasma capsulatum and Coccidioides immitis .