Chapter Outline
Erosive Gastritis
Erosions are defined histologically as epithelial defects that do not penetrate beyond the muscularis mucosae. Although erosive gastritis is rarely diagnosed on conventional single-contrast barium studies, it has become a relatively frequent finding on double-contrast studies, with an overall prevalence of 0.5% to 20% reported in the radiology literature. However, not all patients with erosive gastritis are symptomatic. As a result, it is difficult to be certain of the clinical importance of gastric erosions demonstrated on radiologic or endoscopic examinations.
Pathogenesis
Aspirin or other nonsteroidal anti-inflammatory drugs (NSAIDs) are thought to be the most common cause of erosive gastritis, accounting for about 50% of cases. Other causes include alcohol, stress, trauma, burns, Crohn’s disease, viral or fungal infection, and endoscopic heater probe therapy or other iatrogenic trauma. Still other patients with erosive gastritis have no apparent predisposing factors for this condition. These cases presumably occur as a variant of peptic ulcer disease.
Considerable attention has been focused on the role of aspirin and other NSAIDs in the development of erosive gastritis. Clinical and laboratory investigations have shown that these agents are capable of disrupting the mucosal barrier in the stomach, causing erosive gastritis and gastric ulcers (see Chapter 29 ). In one study, 40% of patients receiving aspirin for 3 months or longer had endoscopic evidence of erosive gastritis. Other studies on healthy volunteers have shown that as few as two aspirin tablets may cause acute erosive gastritis that is recognized on endoscopy within 24 hours. Maximal damage usually occurs within 1 to 3 days, and healing may be documented on endoscopy within 1 week. Thus, gastric erosions may form rapidly after ingestion of aspirin or other NSAIDs and may heal rapidly when these drugs are withdrawn.
Clinical Findings
Patients with erosive gastritis may present with dyspepsia, epigastric pain, or signs of upper gastrointestinal (GI) bleeding. However, other patients are asymptomatic. Gastric erosions can persist for years in the absence of clinical symptoms. Because erosions may be detected as an incidental finding on barium studies or endoscopy, it is important to rule out other abnormalities in the stomach before assuming that these erosions are the cause of the patient’s symptoms.
Radiographic Findings
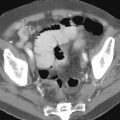
Two types of erosions may be detected on double-contrast studies. The most common type is the complete or varioliform erosion in which punctate or slitlike collections of barium representing the epithelial defects are surrounded by radiolucent halos of edematous, elevated mucosa ( Fig. 30-1 ). Varioliform erosions typically occur in the gastric antrum and are often aligned on the crests of the rugal folds. Because they are shallow lesions, erosions on the posterior (dependent) wall may be better delineated by flow technique to manipulate a thin layer of barium over the dependent mucosal surface. The surrounding mounds of edema may prevent filling of the central pits or depressions, so these erosions sometimes appear as filling defects in the thin barium pool without central collections of barium. In other patients, erosive gastritis may be manifested only by scalloped antral folds ( Fig. 30-2A ). Depending on the quality of mucosal coating, erosions may be faintly seen on the crest of the folds ( Fig. 30-2B ). These scalloped antral folds often persist after the erosions have healed. Residual epithelial nodules or polyps may occasionally be detected at the site of the healed erosions. These hyperplastic polyps are thought to represent the sequelae of chronic erosive gastritis.


Incomplete or flat erosions are epithelial defects that are not associated with elevation of the surrounding mucosa. These erosions appear as linear streaks or dots of barium ( Fig. 30-3 ). Because the surrounding mucosa is normal, incomplete erosions are much more difficult to detect than varioliform erosions, accounting for only 5% to 19% of all erosions found on double-contrast studies.

Although no causative significance is generally attributed to the shape or location of gastric erosions seen on double-contrast studies, aspirin and other NSAIDs may occasionally produce incomplete, linear or serpiginous erosions that tend to be clustered in the body of the stomach on or near the greater curvature ( Fig. 30-4 ). It has been postulated that these erosions result from localized mucosal injury as the dissolving capsules or tablets collect by gravity in the dependent portion of the stomach. Whatever the explanation, these distinctive linear or serpiginous erosions should be highly suggestive of recent aspirin or other NSAID use. Nevertheless, most patients with NSAID-induced erosive gastritis have typical varioliform erosions in the gastric antrum. Aspirin or other NSAIDs should therefore be considered the most likely cause of erosive gastritis, even when the erosions have a varioliform appearance.

Recurrent episodes of NSAID-induced erosion formation and healing may eventually lead to relatively subtle flattening and deformity of the greater curvature of the antrum, a radiologic sign of NSAID-related gastropathy ( Fig. 30-5 ). Detection of gastric erosions or greater curvature flattening should therefore lead to careful questioning of the patient about a possible history of aspirin or other NSAID use. If recent ingestion of these drugs is confirmed in symptomatic patients, withdrawal of the offending agent usually produces a marked clinical response.

Crohn’s disease is another condition that can be manifested on double-contrast studies by multiple erosions or aphthoid ulcers in the stomach. However, these patients usually have associated Crohn’s disease involving the small bowel or colon (see later, “ Crohn’s Disease ”). Shallow ulcers or erosions may also result from opportunistic infection by cytomegalovirus (CMV) in patients with AIDS (see later, “ Cytomegalovirus ”), or they may occur as a complication of endoscopic heater probe therapy or other iatrogenic trauma ( Fig. 30-6 ).

Differential Diagnosis
Gastric erosions can sometimes be mistaken on barium studies for ulcerated submucosal masses or bull’s-eye lesions in the stomach. However, the central ulcer of a bull’s-eye lesion is considerably larger than an erosion, and the surrounding mass tends to be larger than the radiolucent mound of edema surrounding an erosion (see Chapter 33 ). Bull’s-eye lesions also tend to be more sporadic than erosions and are not typically aligned on the crests of the folds. As a result, it is almost always possible to distinguish these lesions by radiographic criteria.
Antral Gastritis
Some patients have a form of gastritis that is confined to the gastric antrum, an entity also known as antral gastritis . Alcohol, tobacco, coffee and, more recently, Helicobacter pylori have been implicated in the development of antral gastritis (see later, “ Helicobacter pylori Gastritis ”). Some patients with this condition have increased secretion of peptic acid, but others have normal or even decreased acid secretion. Affected individuals may present with dyspepsia, epigastric pain, or other symptoms indistinguishable from those of peptic ulcer disease. Treatment is generally aimed at suppressing acid secretion in the stomach.
Radiographic Findings
Antral gastritis may be manifested on barium studies by thickened folds, antral erosions (see earlier, “ Erosive Gastritis ”), crenulation of the lesser curvature, mucosal nodularity, transverse antral striae, a hypertrophied antral-pyloric fold, and antral narrowing. Some patients have thickened, scalloped, or lobulated folds that are oriented longitudinally in the antrum ( Fig. 30-7A ), whereas others have thickened transverse antral folds ( Fig. 30-7B ). Thickened folds, which are detected radiographically in about 75% of cases, should be recognized as the single most common sign of antral gastritis on barium studies. The vast majority of these patients are found to have H. pylori as the cause of their gastritis (see later, “ Helicobacter pylori Gastritis ”).

Crenulation or irregularity of the lesser curvature of the distal antrum may also be recognized as a sign of antral gastritis on barium studies (see Fig. 30-7B ). Other patients may have fine transverse striations, or antral striae, as a sign of chronic antral gastritis, although this finding can also be seen as a normal variant. Still other patients may have a single lobulated fold that arises on the lesser curvature of the prepyloric antrum and extends into the pylorus or base of the duodenal bulb ( Fig. 30-8 ). This so-called hypertrophied antral-pyloric fold is thought to be a sequela of chronic antral gastritis and is often associated with other radiographic signs of gastritis. Endoscopy is not warranted when a characteristic antral-pyloric fold is seen on barium studies. If the fold is more lobulated and cannot be distinguished from a polypoid or plaquelike carcinoma on the lesser curvature, however, endoscopy and biopsy should be performed to rule out malignant tumor.

Differential Diagnosis
Severe antral gastritis associated with antral narrowing must be differentiated from gastric carcinoma. With malignant tumors, however, the narrowed antrum tends to have a more abrupt transition with the adjacent stomach and a more fixed, rigid contour. Thus, it is usually possible to differentiate these conditions by radiographic criteria. When the folds are markedly thickened and lobulated, antral gastritis can also mimic the appearance of lymphoma or even a submucosally infiltrating carcinoma. In such cases, endoscopy may be required for a more definitive diagnosis.
Helicobacter pylori Gastritis
H. pylori (formerly known as Campylobacter pylori ) is a gram-negative bacillus that was first isolated from the stomach by Warren and Marshall in 1983. Since then, H. pylori has been recognized as the most common cause of chronic active gastritis. The organism is usually found in clusters or clumps beneath the mucous layer on surface epithelial cells or, less commonly, superficial foveolar cells in the stomach. H. pylori gastritis is characterized pathologically by an acute inflammatory reaction in the mucosa with accumulation of neutrophils, plasma cells and, eventually, lymphoid nodules. The gastric antrum is the most common site of involvement, but the proximal half of the stomach or even the entire stomach may be involved by this disease. H. pylori gastritis is important not only because it may cause upper GI symptoms but also because it is associated with the development of gastric and duodenal ulcers (see Chapter 29 ), gastric carcinoma (see Chapter 32 ), and low-grade, B-cell, mucosa-associated lymphoid tissue (MALT) lymphoma (see Chapter 33 ).
Clinical Findings
H. pylori infection is acquired by oral ingestion of the bacterium and is mainly transmitted within families during early childhood. H. pylori is a worldwide pathogen, being most common in developing countries. In developed countries, H. pylori is more common in lower socioeconomic populations. The prevalence of H. pylori also increases with age; more than 50% of Americans over 60 years of age are infected by this organism. Some people with H. pylori may present with dyspepsia, epigastric pain, or other upper GI symptoms, but most are asymptomatic. Even when symptoms are present, it is often difficult to prove that the symptoms are caused by H. pylori because of the high prevalence of this infection.
H. pylori gastritis can be eradicated from the stomach by treatment with a combination of antibiotics and antisecretory agents (proton pump inhibitors). In a consensus development panel sponsored by the National Institutes of Health in 1994 and a subsequent update conference sponsored by the American Digestive Health Foundation in 1997, combination therapy with antibiotics and antisecretory agents was recommended for all H. pylori –positive patients with gastric or duodenal ulcers to accelerate ulcer healing and decrease the rate of ulcer recurrence. However, there are conflicting data about the value of H. pylori eradication therapy in patients with nonulcer dyspepsia. As a result, the panels did not recommend treatment for this subset of patients. It therefore remains unclear whether combination therapy should be reserved for patients with H. pylori who have gastric or duodenal ulcers or whether patients with H. pylori who have nonulcer dyspepsia would also benefit from treatment.
H. pylori gastritis can be accurately diagnosed at endoscopy on the basis of histologic specimens, cultures, and the rapid urease test. However, noninvasive tests for H. pylori, such as the urea breath test (using orally administered 14 C- or 13 C-labeled urea) serologic tests, and stool antigen tests have reported sensitivities and specificities of greater than 90%. Highly accurate noninvasive tests are therefore available for detecting this infection.
Radiographic Findings
H. pylori gastritis is the most common cause of thickened folds in the gastric antrum or body on double-contrast barium studies ( Fig. 30-9 ). However, other patients may have diffusely thickened folds in the stomach or thickened folds that are confined to the fundus. Still others with H. pylori gastritis may have markedly thickened, lobulated gastric folds (i.e., polypoid gastritis) in a diffuse ( Fig. 30-10 ) or localized ( Fig. 30-11 ) distribution. In such cases, it may be difficult or impossible to differentiate H. pylori gastritis from other infiltrative conditions or even malignant tumor involving the stomach (see later, “Differential Diagnosis”).



H. pylori gastritis may also be manifested on double-contrast studies by enlarged areae gastricae (≥3 mm in diameter) in the stomach (see Fig. 30-9 ). In the past, enlarged areae gastricae were associated with hypersecretory states and duodenal ulcers. In retrospect, however, this association was probably related to underlying H. pylori gastritis in many of these patients. The presence of enlarged areae gastricae should therefore suggest the possibility of H. pylori, particularly if associated with thickened gastric folds.
Patients with chronic H. pylori gastritis may gradually acquire lymphoid tissue in the gastric mucosa, resulting in the development of intramucosal aggregates of lymphocytes or lymphoid follicles containing germinal centers. This phenomenon is thought to be mediated by a specific immune response to H. pylori. In one study, more than 90% of patients with lymphoid hyperplasia of the stomach were found to have H. pylori gastritis. Lymphoid hyperplasia is therefore a potential marker for H. pylori gastritis, even in the absence of other findings. These enlarged lymphoid follicles are manifested on double-contrast barium studies by innumerable tiny (1-3 mm in diameter), round, frequently umbilicated nodules that carpet the mucosa of the gastric antrum or antrum and body ( Fig. 30-12 ). The radiographic findings are therefore similar to those of lymphoid hyperplasia in the small bowel or colon.

Differential Diagnosis
The radiographic findings of H. pylori gastritis may be indistinguishable from those of hypertrophic gastritis, Ménétrier’s disease, or lymphoma when the thickened, lobulated folds have a diffuse distribution, and the findings may be indistinguishable from malignant tumors such as lymphoma or a submucosally infiltrating carcinoma when the enlarged, polypoid folds have a focal distribution. In other patients with H. pylori gastritis, computed tomography (CT) may reveal circumferential thickening of the antrum or focal thickening of the posterior gastric wall, occasionally simulating a gastric carcinoma. Endoscopy and biopsy are required for a definitive diagnosis when malignant tumor is suspected on the basis of the radiographic findings. Nevertheless, it is important to be aware of the association between H. pylori and this polypoid form of gastritis, so careful testing for the organism is performed at the time of endoscopy.
When H. pylori gastritis is associated with lymphoid hyperplasia of the stomach, the major consideration in the differential diagnosis is low-grade gastric MALT lymphoma (see Chapter 33 ). However, gastric MALT lymphoma is manifested on double-contrast studies by multiple round, variably sized, often confluent nodules with poorly defined borders ( Fig. 30-13 ). In contrast, the nodules of gastric lymphoid hyperplasia have more discrete borders, a more uniform size, and, not infrequently, central umbilications (see Fig. 30-12 ). Lymphoid hyperplasia of the stomach should also be differentiated from enlarged areae gastricae, another finding associated with H. pylori gastritis. However, enlarged areae gastricae have a more polygonal or angulated configuration, producing a sharply marginated reticular network (see Fig. 30-9 ), and they do not contain central umbilications. Other unusual neoplastic lesions such as leukemic infiltrates or even some of the polyposis syndromes may also be manifested on double-contrast studies by multiple small nodules, but the nodules tend to have a more variable size and more sporadic distribution. Thus, it is usually possible to differentiate lymphoid hyperplasia in the stomach from other conditions on radiographic criteria. If the findings are equivocal, however, endoscopic biopsy specimens should be obtained for a more definitive diagnosis.

Hypertrophic Gastritis
Hypertrophic gastritis, also known as hypertrophic hypersecretory gastropathy, is characterized by marked glandular hyperplasia and increased secretion of acid in the stomach. Gastric folds may be thickened, not only because of glandular hyperplasia but also because of edema and inflammation. Although the pathogenesis of this condition is uncertain, glandular hyperplasia in the stomach may be caused by pituitary, hypothalamic, or vagal stimuli. These patients may present with epigastric pain, nausea and vomiting, or, less frequently, signs of upper GI bleeding. If the radiographic or endoscopic findings support the diagnosis of hypertrophic gastritis, treatment with antisecretory agents is usually recommended to suppress acid secretion in the stomach.
Radiographic Findings
Hypertrophic gastritis is manifested on barium studies by thickened folds, predominantly in the gastric fundus and body, because the acid-secreting portion of the stomach is most affected by this condition ( Fig. 30-14 ). Several studies have shown a significant correlation between the degree of fold thickening and the amount of acid secretion in the stomach. The presence of markedly thickened, lobulated folds in the stomach should therefore suggest the possibility of hypertrophic gastritis. In retrospect, however, many if not most cases of previously diagnosed hypertrophic gastritis probably resulted from infection by H. pylori, a much more common cause of thickened gastric folds (see earlier, “ Helicobacter pylori Gastritis ”).

Differential Diagnosis
H. pylori gastritis, Ménétrier’s disease, and lymphoma are the major considerations in the differential diagnosis of thickened, lobulated gastric folds. H. pylori gastritis can usually be differentiated from hypertrophic gastritis by noninvasive tests for H. pylori, such as the urea breath test and serologic tests (see earlier, “ Helicobacter pylori Gastritis ”). Ménétrier’s disease should be suspected in patients who have normal or decreased acid secretion and a protein-losing enteropathy (see later, “ Ménétrier’s Disease ”), whereas lymphoma should be suspected when associated findings such as ulcers, masses, or bull’s-eye lesions are present in the stomach (see Chapter 33 ). Gastric carcinoma is a less common cause of thickened folds and is usually associated with loss of distensibility and decreased or absent peristalsis in the involved portion of the stomach. If the radiographic findings are equivocal, endoscopy and biopsy may be required to rule out malignant tumor. Rarely, other conditions such as Zollinger-Ellison syndrome, eosinophilic gastritis, and varices may be manifested by thickened folds in the stomach, but the correct diagnosis can usually be suggested on the basis of the clinical history and presentation.
Ménétrier’s Disease
Since its original description by Ménétrier in 1898, Ménétrier’s disease has been recognized as a rare condition of unknown etiology characterized by marked foveolar hyperplasia in the stomach, enlarged gastric rugae, hypochlorhydria, and hypoproteinemia. In the past, this condition was also called cystic gastritis, giant hypertrophic gastritis, giant mucosal hypertrophy, and hyperplastic gastropathy. Ménétrier’s disease may cause chronic disabling symptoms, occasionally necessitating gastric resection. Despite its rarity, this entity has received considerable attention in the radiology literature because of its often dramatic appearance on barium studies.
Pathology
Ménétrier’s disease is characterized histologically by thickening and hyperplasia of the mucosa as a result of cystic dilation and elongation of gastric mucous glands associated with deepening of the foveolar pits. Despite these findings, gastric acid output is decreased or absent in about 75% of cases. Some patients have a protein-losing enteropathy resulting from loss of protein from the hyperplastic mucosa into the gastric lumen. Others have varying degrees of gastritis in a patchy or diffuse distribution.
Clinical Findings
Ménétrier’s disease tends to occur in older patients and is more common in men than in women. Affected individuals often present with epigastric pain, nausea and vomiting, diarrhea, anorexia, weight loss, and/or peripheral edema. Laboratory studies may reveal hypoalbuminemia resulting from a protein-losing enteropathy, hypochlorhydria resulting from decreased acid secretion, or both. Rarely, the development of gastric carcinoma has been described in patients with preexisting Ménétrier’s disease. However, it is unclear whether Ménétrier’s disease is a premalignant condition or whether this association is coincidental.
Some patients with Ménétrier’s disease have spontaneous remission of symptoms, whereas others respond to treatment with antisecretory agents, vagotomy, or antibiotics. However, most patients have a prolonged illness with intractable symptoms. A total gastrectomy may be required for patients who are unresponsive to medical therapy.
Radiographic Findings
Ménétrier’s disease is typically manifested on barium studies by considerably thickened, lobulated folds in the gastric fundus and body, with relative sparing of the antrum ( Fig. 30-15A ). In one study, however, the antrum was involved in almost 50% of patients, so diffuse thickening of gastric folds in no way precludes this diagnosis. The greatest degree of fold thickening usually occurs on or near the greater curvature. When the disease is confined to one portion of the stomach, focally enlarged folds may erroneously suggest a polypoid carcinoma ( Fig. 30-15B ).

Ménétrier’s disease is characterized on CT by a markedly thickened gastric wall, with masslike elevations representing giant, heaped-up folds protruding into the lumen ( Fig. 30-15C ). When Ménétrier’s disease is suspected on barium studies or CT, full-thickness endoscopic biopsy specimens should be obtained to confirm the diagnosis.
Differential Diagnosis
Although a variety of conditions may be manifested by thickened gastric folds, these conditions rarely produce the degree of fold thickening seen in Ménétrier’s disease. When H. pylori gastritis is associated with markedly thickened, lobulated folds, the radiographic findings may be indistinguishable from those of Ménétrier’s disease. Gastric lymphoma is sometimes associated with enlarged folds, but neoplastic infiltration should be suggested by the presence of polypoid masses, ulcers, or bull’s-eye lesions in these patients (see Chapter 33 ). Occasionally, gastric carcinoma may be manifested by thickened folds, but infiltrating cancers tend to narrow the lumen, whereas the stomach usually remains pliant and distensible in patients with Ménétrier’s disease. Zollinger-Ellison syndrome may also be characterized by thickened folds, but the presence of increased secretions in the stomach or other associated abnormalities (e.g., ulcers, thickened folds) in the duodenum and proximal jejunum should suggest the correct diagnosis (see Chapter 29 ). Gastric varices should also be included in the differential diagnosis, but varices tend to have a more serpiginous appearance and are usually confined to the region of the gastric cardia or fundus (see Chapter 34 ). Other conditions involving the stomach, such as Crohn’s disease, eosinophilic gastritis, sarcoidosis, tuberculosis, and syphilis, may also be manifested by thickened folds. In these cases, however, the correct diagnosis is usually suggested by the clinical history and presentation.
Atrophic Gastritis
Atrophic gastritis is important because of its association with pernicious anemia, a megaloblastic anemia caused by decreased synthesis of intrinsic factor and subsequent malabsorption of vitamin B 12 . Pernicious anemia is a disease of older adults; it accounts for 50 of 100,000 hospital admissions in the United States. Although the pathogenesis of this disease is uncertain, an autoimmune mechanism has been postulated because of the frequent finding of parietal cell or intrinsic factor antibodies in these individuals.
More than 90% of patients with pernicious anemia have underlying atrophic gastritis, characterized pathologically by atrophy of mucosal glands, loss of parietal and chief cells, thinning of the mucosa and, eventually, intestinal metaplasia. The finding of intestinal metaplasia is particularly worrisome because it is widely believed to be the precursor lesion of the intestinal type of gastric cancer. The literature has also implicated chronic H. pylori infection as a major cause of atrophic gastritis, intestinal metaplasia, and gastric carcinoma (see Chapter 32 ).
Pathogenesis
Atrophic gastritis may be classified into two types—type A and type B—which have different histologic, immunologic, and secretory characteristics. In type A gastritis, mucosal atrophy is confined to the gastric fundus and body with antral sparing. This type of atrophic gastritis is thought to result from immunologic injury (i.e., antiparietal cell antibodies) and is usually associated with pernicious anemia.
In contrast, type B gastritis is characterized predominantly by antral disease with limited involvement of the fundus and body. This form of atrophic gastritis is more common and usually results from mucosal injury by H. pylori or, less commonly, by other endogenous or exogenous agents such as bile acids or alcohol. In patients with H. pylori, it has been postulated that the organism progressively damages the gastric mucous layer, causing chronic atrophic gastritis and gastric atrophy. A particular strain of H. pylori known as cagA (cytotoxin-associated gene A) has been associated with an increased prevalence and degree of atrophic gastritis, predisposing these patients to the development of gastric carcinoma, so the risk of malignant degeneration may not be the same for all patients with this infection.
Clinical Findings
Although atrophic gastritis rarely causes symptoms, some patients with pernicious anemia initially present with neurologic symptoms as a result of long-standing vitamin B 12 deficiency. Early diagnosis of pernicious anemia is therefore important, so vitamin B 12 replacement therapy can be initiated before the development of irreversible neurologic sequelae. Because the average adult has a 3- to 6-year body store of vitamin B 12 , the gastric abnormalities in pernicious anemia may predate the hematologic and neurologic abnormalities in this condition by several years. The diagnosis of atrophic gastritis on upper GI studies might therefore permit these patients to be treated with vitamin B 12 supplements before the full-blown clinical entity of pernicious anemia has developed.
Relationship to Gastric Carcinoma
Patients with atrophic gastritis and pernicious anemia are at increased risk for the development of gastric carcinoma. In one study, the risk of developing gastric cancer in these patients was found to be about three times greater than that in the general population. Although some investigators advocate endoscopic or radiologic surveillance of patients with known pernicious anemia, others believe that the risk of cancer is not high enough to warrant routine screening. Nevertheless, any patient with pernicious anemia who has occult GI bleeding should be evaluated aggressively to rule out a superimposed gastric carcinoma.
The literature suggests that patients with H. pylori– associated atrophic gastritis have a substantially increased risk of developing gastric carcinoma (see Chapter 32 ). Evidence from several studies has shown that the risk of gastric cancer in H. pylori– positive patients is about four times greater than that in patients without this infection. Because of the high prevalence of H. pylori in the population, however, it remains unclear whether widespread eradication of H. pylori is justified from a societal perspective to prevent the development of cancer.
Radiographic Findings
The diagnosis of atrophic gastritis may be suggested on single-contrast studies by the presence of a narrowed, tubular stomach with decreased or absent mucosal folds, predominantly in the body and fundus, also known as a bald fundus ( Fig. 30-16 ). In one study, 80% of patients with atrophic gastritis and pernicious anemia had a fundal diameter of 8 cm or less, absent folds in the fundus and body, and small (1-2 mm in diameter) or absent areae gastricae in the stomach; however, this combination of findings was also present in about 10% of age-matched controls. The radiologic diagnosis of atrophic gastritis in patients with pernicious anemia has therefore been limited by a lack of criteria that are sensitive and specific for this condition. When atrophic gastritis is suspected on double-contrast studies, serum vitamin B 12 levels should be obtained to determine whether vitamin B 12 replacement therapy is indicated.

Many questions remain about the appearance of the areae gastricae in patients with atrophic gastritis. It was previously postulated that variations in the size of the areae gastricae depend on parietal cell mass. Thus, the small size and frequent absence of areae gastricae in patients with atrophic gastritis may be explained by the loss of parietal cells in these individuals. In contrast, focal enlargement of the areae gastricae should raise the possibility of intestinal metaplasia or even a superficial spreading carcinoma, so this finding should be evaluated by endoscopy and biopsy.
Differential Diagnosis
Scirrhous carcinoma of the stomach (i.e., linitis plastica) is the most important consideration in the differential diagnosis of atrophic gastritis. However, scirrhous tumors are usually characterized by a nodular, distorted mucosa and thickened, irregular folds, whereas atrophic gastritis is characterized by a smooth, featureless mucosa and decreased or absent folds. Thus, scirrhous carcinomas can almost always be differentiated from atrophic gastritis by radiologic criteria. Scarring from peptic ulcer disease or other conditions may also be characterized by gastric narrowing, but the antrum and body tend to be involved, rather than the fundus.
Granulomatous Conditions
Crohn’s Disease
Although Crohn’s disease primarily affects the small bowel and colon, early signs of upper GI involvement may be detected on double-contrast barium studies in more than 20% of patients with granulomatous ileocolitis. Occasionally, the onset of upper GI disease coincides with or even precedes the onset of ileal or colonic disease, so these patients do not necessarily have known Crohn’s disease when they seek medical attention. Endoscopic biopsy specimens from the stomach or duodenum may fail to reveal granulomas because of the superficial nature of the biopsy specimens and patchy distribution of the disease. Thus, the absence of definitive histologic findings should not discourage a diagnosis of gastroduodenal Crohn’s disease if the clinical and radiographic findings suggest this condition.
Clinical Findings
Patients with early gastroduodenal involvement by Crohn’s disease are often asymptomatic, but those with more advanced disease may present with pain, vomiting, weight loss, or signs of upper GI bleeding. Others may have diarrhea because of associated ileocolic Crohn’s disease. The development of a gastrocolic or duodenocolic fistula is classically manifested by feculent vomiting, diarrhea, and weight loss, but this triad of findings is present in only about 30% of patients, so gastrocolic fistulas are not often suspected on clinical grounds.
Asymptomatic patients with early gastroduodenal Crohn’s disease require no specific treatment. In patients with more advanced disease, medical treatment for Crohn’s disease may relieve epigastric pain or other upper GI complaints. In contrast, a surgical bypass procedure such as a gastrojejunostomy or duodenojejunostomy may be required to alleviate symptoms of gastric outlet obstruction.
Radiographic Findings
As in the ileum or colon, gastroduodenal Crohn’s disease is characterized by nonstenotic and stenotic phases of involvement. The initial nonstenotic phase is manifested by a spectrum of findings, including aphthoid ulcers, larger ulcers, thickened folds, and distorted, effaced or, rarely, cobblestoned mucosa. Subsequent scarring and fibrosis may cause antral, pyloric, or duodenal narrowing with progressive gastric outlet obstruction. Thus, the radiologic features of gastroduodenal Crohn’s disease are similar to those in the small bowel and colon.
Gastric Involvement.
Gastric Crohn’s disease almost always involves the antrum or antrum and body of the stomach. More proximal extension of Crohn’s disease is unusual, and isolated fundal involvement rarely occurs. When the stomach is affected by Crohn’s disease, the duodenum also tends to be involved. Most patients have associated granulomatous ileocolitis, but the diagnosis of Crohn’s disease may not be known at the time of clinical presentation. When gastric involvement is suggested by upper GI studies, a small bowel follow-through or barium enema should be performed to determine whether there is concomitant ileocolic disease.
Aphthoid ulcers, the earliest histologic lesions of Crohn’s disease, are detected in the stomach on double-contrast studies in more than 20% of patients with granulomatous ileocolitis. These aphthoid ulcers tend to be located in the gastric antrum or in the antrum and body, appearing as punctate or slitlike collections of barium surrounded by radiolucent mounds of edema ( Fig. 30-17 ). As a result, these lesions may be indistinguishable from varioliform gastric erosions (see earlier, “ Erosive Gastritis ”).

More advanced gastroduodenal Crohn’s disease may be manifested by one or more larger ulcers, thickened folds ( Fig. 30-18 ), or a nodular or cobblestoned mucosa in the gastric antrum or body. Subsequent scarring may lead to the development of a narrowed, tubular, funnel-shaped antrum that has been likened to the appearance of the sacramental ram’s horn, or shofar, used to sound the advent of the Jewish New Year ( Fig. 30-19 ). In other patients, combined gastroduodenal scarring may produce a single, continuous tubular structure involving the antrum and duodenum with obliteration of the normal anatomic landmarks at the pylorus ( Fig. 30-20 ). Because of its resemblance to a postsurgical stomach after a Billroth I partial gastrectomy, this finding has been described as the pseudo–Billroth I sign of gastroduodenal Crohn’s disease. Rarely, filiform polyps may be found in the stomach as a sequela of granulomatous gastritis ( Fig. 30-21 ).