Devasenathipathy Kandasamy, Govind B. Chavhan Disorders of sex development (DSD) are a heterogeneous group of congenital disorders which are characterized by atypical or abnormal chromosomal, gonadal, internal or external genital structures. Ambiguous genitalia is a subset of DSD where it is not possible to clearly ascertain the sex of the baby as male or female based on the external genitalia appearance. There can be incomplete development of external genitals or can have the characteristics of both the sexes. It is also important to understand that not all DSD present with ambiguous genitalia. DSD is a stressful condition for the parents as well as for the child later on, because of the risk of inappropriate gender assignment which can have a profound effect on physical and psychological development of the child. It is a challenging condition to manage for the healthcare providers because of the fact that the underlying pathogenesis is not fully understood and it needs a multidisciplinary team comprising paediatric surgeon, endocrinologist, geneticist, radiologist and a psychiatrist to diagnose and treat the child. This is even more challenging in developing countries, where there will be delay in presentation of the child to the hospital coupled with lengthy workup and consequently delayed definitive treatment. Depending on the definition of DSD the reported prevalence is variable ranging from 0.02% to 0.2%. In spite of this relatively common occurrence, the data on DSD are limited and it is because of many factors such as confusion in the nomenclature, classification and inadequate awareness among healthcare providers, etc. One of the major steps in streamlining the nomenclature happened in 2006 when Lawson Wilkins Pediatric Endocrine Society (LWPES) and the European Society for Paediatric Endocrinology (ESPE) formed a working group and proposed the all encompassing term DSD to describe these disorders. They also proposed the classification system which is still being widely accepted. Imaging can provide critical insights to help in their proper classification, gender assignment and eventually appropriate management. Hence it is important for the radiologists to understand the basics of embryology, nomenclature and classification, and imaging features to manage these patients appropriately. Understanding the embryology behind the development of gonads and genital organs is paramount in understanding the clinical and imaging appearances in these children (Fig. 11.7.1). The process of sexual differentiation is complicated and has the following four parts to it, namely genetic or chromosomal, gonadal, ductal and genitalia differentiation. It is not possible to differentiate male from female genital structures before 6 weeks of gestation. Chromosomal sex is determined during the fertilization which is typically XX for female and XY for male. In the early stages, the embryo has the capability to differentiate either to male or female. The embryo at this stage is morphologically bisexual. Later on, genetic sex code is the one which will code for primitive testes or primitive ovaries. At around 5th week after fertilization, the gonadal ridge develops from the urogenital ridge and it remains undifferentiated till the 7th week. This stage of development is called the indifferent stage. From this point, male and female differentiation is distinct from each other. In a genetically male foetus, the differentiation is triggered by the sex determining region Y (SRY) gene in the Y chromosome which activates the testis determining factor (TDF). As a result of this, immature germ cells and somatic cells organize to form testicular cords. Some cells in the cord differentiate into Sertoli cells because of the influence by TDF and they secrete Mullerian inhibiting substance (MIS) which plays an important role in ductal differentiation (see below). Certain stromal cells differentiate into Leydig cells which are responsible for the production of testosterone from 10 weeks. Female gonadal sex differentiation is as a result of the absence of SRY gene and consequently the absence of MIS and testosterone which leads to uninhibited differentiation into ovaries. Although a single X chromosome is sufficient for female sex differentiation, two X chromosomes are required to maintain the oocytes in ovaries. This pattern can be observed in patients with 45X where the oocytes become atretic after initial development. Wolffian or mesonephric ducts and Mullerian or paramesonephric ducts are the two paired sets of ducts involved in ductal differentiation and eventually forming the internal genital organs. Paired Wolffian ducts are responsible for the development of male internal genital organs such as seminal vesicles, epididymis, vas deferens and ejaculatory ducts on either side. Similarly, the paired Mullerian ducts are responsible for the development of female internal organs such as uterus, fallopian tubes and upper vagina. The choice of the future ductal system is determined by the gonadal sex and later on maintained by hormonal influence. In males, the MIS is responsible for the degeneration of Mullerian ducts. This inhibiting influence of MIS on the paired Mullerian ducts is believed to happen only on the ipsilateral side. Hence, MIS has to be produced on both sides for the complete degeneration of Mullerian structures in males. The surviving Wolffian ducts are later on maintained by testosterone. In females, the Mullerian ducts develop into the future female internal genital structures without inhibition due to the absence of MIS and the Wolffian structures do not develop because of the absence of testosterone. The development of external genitalia begins in the 3rd week at which stage both the male and female external genital structures appear the same. The cloacal folds fuse in the cranial aspect to form the genital tubercle and caudally they form urethral and anal folds. Genital swellings develop on either side of the urethral folds. After this stage, the differentiation of genital tubercle into male or female structures happens around 9 to 12 weeks and is completely dependent on the effect of androgens. The testicular descent is also influenced by the circulating androgens. In females, the absence of androgens leads to the development of female genital structures. Even in males, if the sensitivity of the tissues towards testosterone is less, it can lead to feminization of external genitalia like in patients with androgen insensitivity syndrome (AIS). The differentiation and development of male and female sex from a morphologically bisexual stage is a complex process influenced by genetic and hormonal factors. Any anomaly in this entire process can lead to DSD of varying severity. Before 2006 the literature was flooded with a variety of terminologies which led to confusion and inconsistencies in diagnosis and reporting. Old terminologies like true hermaphroditism, pseudohermaphroditism and intersex are quite confusing and controversial. In 2006, the LWPES and the ESPE organized the International consensus conference on intersex and proposed new terminologies and a classification system to indicate atypical development of chromosomal, gonadal and anatomic sex. In this classification the DSDs with sex chromosome abnormalities are separated from those with normal chromosomes (46 XX DSD and 46 XY DSD). The older and newer terminologies of DSDs and their salient features are listed in Tables 11.7.1 and 11.7.2.
11.7: Intersex disorders: Concepts, types and diagnostic approach
Introduction
Embryology of reproductive system
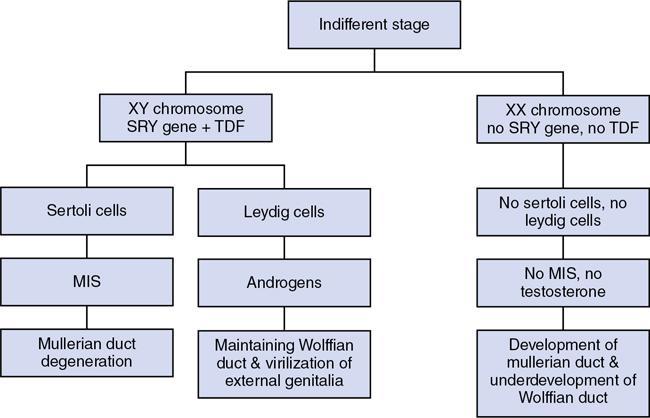
Genetic or chromosomal sex
Gonadal sex
Ductal differentiation
External genital structures
Nomenclature and classification of DSD
Old Terminology
New Terminology
Intersex
Disorders of sex development (DSD)
Male pseudohermaphrodite
46 XY DSD
Female pseudohermaphrodite
46 XX DSD
True hermaphrodite
Ovotesticular DSD
XX male or XX sex reversal
46 XX testicular DSD
XY sex reversal
46 XY complete gonadal dysgenesis
SEX CHROMOSOME DSD
Gonadal Dysgenesis
Features
46 XX DSD
46 XY DSD
Ovotesticular DSD
Mixed Gonadal DSD
Pure Gonadal DSD
Typical Karyotype
46 XX
46 XY
46 XX (most common), 46 XY, 46 XX/XY mosaic
45 XY/XO mosaic
variable
Phenotype
Ambiguous genitalia, variable extent of virilization
Ambiguous genitalia, variable extent of feminization
Variable, male with feminization to female with virilization
Variable, ambiguous genitalia or female type
Female, primary amenorrhea, no secondary sexual development
Gonads
Ovaries
Testes
Both testis and ovary or ovotestis
Testis on one side and streak gonad on the other side
Bilateral streak gonads
Common Causes
Congenital adrenal hyperplasia, antenatal androgen exposure
Defects in testosterone synthesis, androgen insensitivity syndromes (AIS)
Chromosomal abnormality
Chromosomal abnormality
Chromosomal abnormality
Typical features
Virilized external genitalia, nonpalpable gonads, Mullerian structures on imaging
Feminization of external genitalia, absence of Mullerian structures, presence of male internal genital structures, undescended testes
Presence of both ovarian and testicular tissues, presence of uterus
Testis on one side and streak gonad on the other side. High cancer risk
Streak gonads and underdeveloped Mullerian structures. High cancer risk ![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Radiology Key
Fastest Radiology Insight Engine