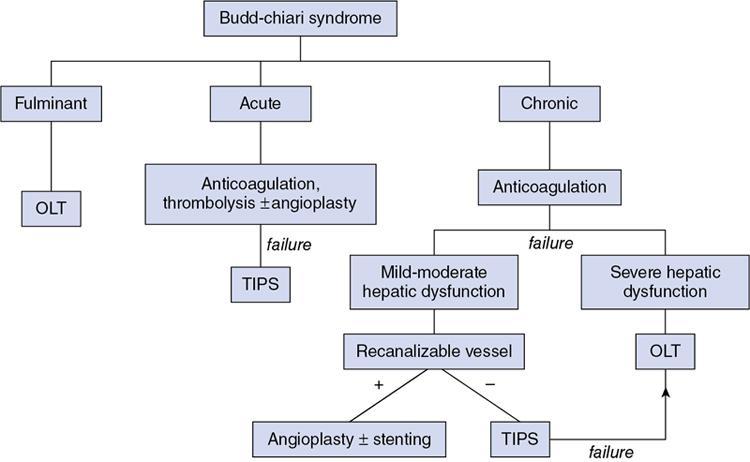
Chandan Jyoti Das, Abdul Razik Budd–Chiari syndrome (BCS) is the eponym for the clinical presentation occurring from hepatic venous outflow tract obstruction (HVOTO) anywhere between the small hepatic venules and cavoatrial junction. Obstruction at the sinusoidal level and those resulting from cardiac and pericardial diseases, do not come under BCS. Interventional radiology plays an important role in the management of BCS and has largely replaced surgical shunts. This chapter provides a brief review of some important clinical aspects of BCS, followed by detailed description of the interventional management. BCS can be primary, when obstruction occurs due to intraluminal pathology (e.g. thrombosis, webs) or secondary, when compression is caused by extrinsic pathologies such as tumours or abscesses. Based on the site of obstruction, three types exist (Table 9.27.1). The following discussion deals with only types I and II of primary BCS. There is a well-recognized demographic difference in the etiology and location of venous occlusion between the western and eastern populations. In the west, most cases occur secondary to prothrombotic conditions, with long segment or multifocal hepatic venous stenosis being the most prevalent patterns. The most common predisposing condition is usually a myeloproliferative syndrome, often with JAK2 V617F, JAK2 exon 12 or calreticulin mutations. Other prothrombotic conditions include inherited thrombophilia (factor V Leiden mutation, protein C, protein S and antithrombin deficiency), obesity, neoplasms (tumour-induced thrombophilia or direct tumour thrombus within the vein), autoimmune disorders (antiphospholipid syndrome, Behcet’s disease), paroxysmal nocturnal haemoglobinuria, pregnancy, oral contraceptive use and hyperhomocysteinemia. In 15%–20%, multiple predisposing factors may be present. In Asia, most cases possess short segment (<1 cm) fibrous web or membranous obstruction of the inferior vena cava (IVC), hepatic vein or both and demonstrate a more chronic course of the disease. Earlier considered congenital, the web has been recently attributed to organization of thrombus resulting from diarrhoea-related dehydration or postpartum sepsis. This is supported by the observation of higher prevalence of webs in patients of the low socioeconomic strata. More recently, the incidence of long-segment hepatic venous thrombosis has increased in the East, possibly from improved socioeconomic conditions. Most patients presenting with BCS in the developing countries are young adults. The presentation depends on the acuteness of the thrombus and may be fulminant, acute, subacute or chronic. Acute BCS presents with the classical triad of abdominal pain, hepatomegaly and ascites over a short duration (<1 month). In 5%, rapid progression of congestion to ischemic hepatocyte necrosis results in fulminant acute liver failure (jaundice, coagulopathy and encephalopathy). A gradually developing thrombus often permits the development of collateral vessels, which partially decompresses the congestion resulting in subacute (onset of symptoms over 1–6 months) or chronic BCS (onset over >6 months). Chronic BCS is overall the most common form and presents with manifestations of portal hypertension such as ascites and variceal bleeding. Some patients may be completely asymptomatic, with BCS being detected on evaluation for deranged liver function tests. Patients with IVC occlusion may have additional symptoms of prominent abdominal wall veins, lower limb edema and ulcers. At present, there are no randomized trials evaluating the different treatment protocols used in BCS. As such, most algorithms are based on retrospective studies or expert opinion. Asian patients with membranous/short-segment IVC or hepatic vein obstruction are excellent candidates for upfront angioplasty. On the other hand, in the West, where long-segment thrombosis is more prevalent, the American Association for Study of Liver Diseases (AASLD) advocates a ‘stepwise’ approach to escalation of invasiveness (Fig. 9.27.1). This involves initial medical management, and reserves the more invasive therapies (intervention/surgery) for nonresponders. Interventional therapies may be initiated early in patients with Rotterdam class III, high Clichy, high model for end-stage liver disease (MELD >18) and high Child–Turcotte–Pugh (CTP C) scores. Among these, the Rotterdam and Clichy scores, tailored specifically for BCS, predict early (3-month) mortality better than the CTP or MELD scores. Interventional therapies are classified as physiological (angioplasty, with or without stenting) or derivative (transjugular intrahepatic portosystemic shunt; TIPS). These therapies relieve outflow tract obstruction and the resultant stagnation, thereby improving liver perfusion. The specific treatment depends on acuteness of presentation, as well as the location and length of the involved venous segment. In acute fulminant liver failure and end-stage liver disease, once permanent damage has occurred, orthotopic liver transplantation (OLT) is indicated. After the advent of interventional procedures, surgical portosystemic shunts have been rarely used. Medical management involves anticoagulation; sodium-restricted diet and diuretics for ascites; beta-blockers for portal hypertension, as well as treatment of the predisposing factor. Anticoagulation is performed even in asymptomatic patients and those without predisposing factors as it improves long-term survival. It prevents thrombus propagation, rather than dissolution of an existing thrombus. Generally, anticoagulation is initiated with unfractionated or low-molecular-weight heparin (LMWH) till the effect of oral anticoagulants (vitamin K antagonists or direct thrombin inhibitors) takes over. Subsequently, oral anticoagulation is continued to maintain the target prothrombin time-international normalized ratio (PT-INR) in the range of 2–3. LMWH is preferred over unfractionated heparin in view of lower risk of heparin-induced thrombocytopenia. Thrombolysis restores hepatic outflow in patients with recent (acute or subacute) thrombosis who do not respond to early anticoagulation. It may be performed as systemic infusion through a peripheral vein or as direct infusion through a catheter placed in the hepatic artery or vein (catheter-directed or local thrombolysis). Best outcomes are seen in the acute phase, and in patients having a low thrombus load and some residual flow within the thrombus. Patients with higher clot load have poor outcomes since complete thrombolysis is difficult to achieve. Although no studies have compared systemic versus catheter-directed thrombolysis, the latter is preferred since it enables higher local concentration of the thrombolytic agent and better clot-to-drug contact, thereby increasing the chances of recanalization, while reducing the required dose and risk of remote bleeding. Catheter-directed thrombolysis involves the placement of a multisidehole catheter across the thrombus, followed by administration of the drug directly into the thrombus. After securing venous access through the jugular or femoral route, a catheter–guidewire combination is used to negotiate the length of the thrombus. Failure to advance the soft end of the guidewire through the thrombus (guidewire traversal test) indicates chronicity and predicts failure of lysis. Once negotiated, the infusion catheter is placed across the thrombus to bath the thrombus with the thrombolytic agent. An initial thrombus lacing is performed with a bolus dose of the drug using a ‘pulse-spray’ technique to enable pharmacomechanical disruption, followed by continuous slow infusion of the drug. Recombinant tissue plasminogen activator (rtPA, alteplase) is the most commonly used agent. The typical doses used are 5–10 mg initial bolus followed by 0.5–1 mg/hour. RtPA has a short half-life of 5 minutes, and earlier reversal is possible in case of bleeding complications. Streptokinase and urokinase are uncommonly used in view of their adverse safety profile and lower efficacy. In view of the risk of potentially fatal haemorrhagic complications, thrombolysis is contraindicated in patients with active bleeding and recent history of stroke, trauma, surgery or other invasive procedures (e.g. paracentesis within the past 24 hours). Concurrent heparin infusion (under activated partial thromboplastin time monitoring) is performed to prevent rethrombosis. During the infusion, the patient is monitored in a high-dependency unit and a repeat angiogram is obtained after 4–12 hours. The endpoints to thrombolysis are (a) complete lysis, (b) unchanged thrombus between two consecutive angiographic reviews and (c) occurrence of bleeding complications. Thrombus aspiration may be performed in conjunction with thrombolysis in acute thrombosis and when thrombolysis is contraindicated. In patients with subacute occlusive thrombus, portosystemic collaterals redirect hepatic circulation away from the hepatic veins, which results in suboptimal thrombus-drug contact and poor outcome following thrombolysis. In such cases, partial recanalization achieved using a balloon catheter improves the success of subsequent thrombolysis. In addition, partial hepatic decompression also reduces the risk of variceal bleeding during thrombolysis. In acute thrombosis, predilatation is better avoided in view of the high risk for pulmonary embolism, although a small balloon may be used cautiously. In many cases, thrombolysis wuncovers an underlying segmental venous stenosis or membrane which initially predisposed the thrombus. In such cases, postthrombolysis angioplasty would be required to achieve optimal luminal patency. Balloon angioplasty is performed in short-segment stenosis or membranous occlusion of the vein. Restoring flow in one of the three hepatic veins is sufficient to decompress the liver and relieve the symptoms. The best candidate vein is the one with a healthy proximal segment and is identified by its straight course, good calibre (7–8 mm) and echo-free lumen. Hence USG assessment is crucial for planning angioplasty. IVC is accessed through the transjugular approach using a 9/10-F, 40-cm long introducer sheath and half the bolus dose of intravenous heparin is administered. A cavogram is obtained to assess IVC patency and look for a residual hepatic venous stump. Any IVC stenosis resulting in impaired flow, collateral formation and significant pressure gradient requires angioplasty. However, in many cases, IVC may be compressed by a caudate lobe enlargement with normal flow across, in which case angioplasty is not required. A 5-F multipurpose angiographic catheter is used to cannulate the hepatic vein stump and wedge the stenosis. Subsequently, the stenosis is negotiated using the floppy end of a straight tip hydrophilic guidewire placed coaxially within a catheter, and exchanged for an Amplatz extra stiff guidewire. Once crossed, the completion dose of heparin is provided and angioplasty is performed using an 8–10 mm balloon catheter (5%–10% oversized compared to the lumen). In case there is difficulty in negotiating a tight fibrotic stenosis, the hard back end of the guidewire or a metallic needle (Colapinto needle or Rösch–Uchida trocar stylet) may be used. Ample caution must be taken to prevent inadvertent nontarget puncture and complications such as hemopericardium, haemothorax and pericaval haematoma. The procedure is demonstrated in Figs 9.27.2 and 9.27.3.
9.27: Interventional management of Budd–Chiari syndrome
Introduction
Predisposing factors
Type I
Obstruction of IVC with or without hepatic vein involvement
Type II
Major hepatic vein involvement
Type III
Obstruction of the centrilobular venules; also known as veno-occlusive disease
Clinical presentation
Management protocols
Medical management
Thrombolysis
Technique
Angioplasty and stenting
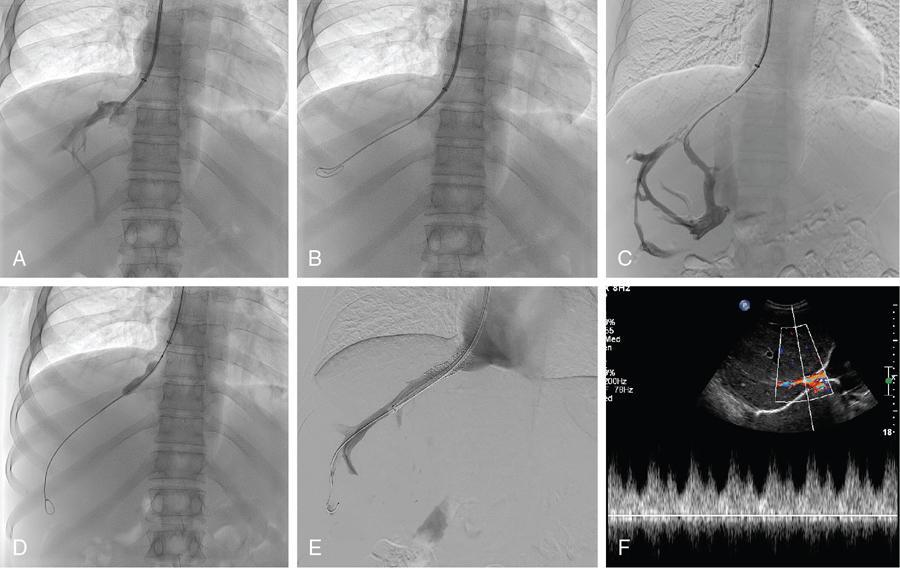
Hepatic angioplasty
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








