9.14: Liver transplant imaging
Rashmi Badhe, Shrinivas B. Desai, Ravi Mohanka
Introduction
Liver transplantation is an established treatment for various acute or chronic end-stage liver diseases, with reported 1-year graft and patient survival rates of 80%–90% and 5-year rates of 60%–90%.
Advances in immunosuppressive therapy, surgical techniques, perioperative care and imaging have combined to improve the outcome of liver transplantation over the past few decades.
The imaging of the pretransplant and posttransplant patient is an important facet of patient management along with imaging evaluation of the donor. Proper recipient selection and complete knowledge of the vascular anatomy of donor and liver volumes is essential for successful outcome. Imaging plays an important role in all of these.
Tables 9.14.1 and 9.14.2 summarize important indications and contraindications to liver transplant.
| Chronic hepatitis | Viral hepatitis, autoimmune hepatitis, chronic drug-induced hepatitis, chronic active hepatitis |
| Alcoholic cirrhosis | |
| Neoplasia | Hepatocellular carcinomas, unresectable fibrolamellar carcinomas, hepatoblastoma, nonresectable perihilar cholangiocarcinoma (after thorough screening to exclude metastatic disease) |
| Cholestatic diseases | Primary biliary cholangitis, primary biliary cirrhosis, secondary biliary cirrhosis, biliary atresia, Caroli’s disease, Alagille syndrome, familial cholestatic syndromes |
| Acute fulminant hepatic failure | Acute drug toxicity, acute viral illness |
| Metabolic and genetic diseases | Wilson’s disease, hemochromatosis, alpha-1 antitrypsin deficiency, glycogen storage disease, nonalcoholic steatohepatitis (NASH), |
| Other conditions | Budd Chiari syndrome, noncirrhotic portal fibrosis, polycystic liver disease, trauma |
| Absolute | Relative |
|---|---|
| Active alcohol or substance use | Advanced age |
| Active extrahepatic malignancy | Psychological instability |
| Active uncontrolled infection or sepsis | Severe obesity or malnutrition |
| Severe cardiopulmonary disease | Anatomic abnormalities that preclude liver transplantation – thrombosis of the entire portal and superior mesenteric venous system |
| Brain death |
Types of liver transplant
Orthotopic liver transplantation (OLT) can be performed from a cadaver known as deceased donor liver transplant (DDLT) or from a living adult donor known as living donor liver transplantation (LDLT).
1. Cadaveric liver transplantation or DDLT
It has been the most common type of liver transplantation in adults, especially in western countries where there is more awareness of organ donation. It has the advantages of full donor liver transplantation and is usually technically more successful; however, organ availability is limited leading to longer waiting times for the transplants.
2. Living donor liver transplantation or LDLT
LDLT’s account for small but very important subset of volume of liver transplants worldwide, as it offers life-saving treatment options for patients with low MELD scores before the complications set in and also offer timely curative treatment to the patients with hepatocellular carcinoma (HCC).
One of the recent studies have shown superior survival outcomes in LDLT recipients (3-year 86% vs 80%, p = 0.03) with shortened postoperative hospital stay.
Advantages of LDLT
- 1. Reduced ischemic time and expedited and planned surgical time as it is performed on an elective basis.
- 2. Superior quality of the liver graft and absence of any other comorbidities in the healthy donors as against in DDLT where conditions like undiagnosed steatosis in the deceased can potentially compromise the liver quality.
- 3. Possibility of liver transplant (LT) for recipients who might otherwise not be eligible for standard DDLT.
Drawbacks include a potential risk of death or serious complications to the donor and a smaller graft size.
3. Split liver grafting method
A single cadaveric liver is anatomically divided and transplanted into two recipients in split liver grafting method. An extended right liver graft is harvested for an adult recipient and left lateral segment for a paediatric recipient. However, due to the technical challenges of optimizing the vascular flow and biliary drainage which depend largely on the donor anatomy and achieving adequate liver volume for the recipients, it is not very commonly performed.
Pretransplant imaging
Imaging plays vital role in evaluation of recipients and donors prior to transplantation and is an essential step along with various laboratory tests and psychological evaluation.
Recipient evaluation
Preoperative clinical and radiologic evaluation of the transplant candidate is essential for appropriate patient selection and for better outcome.
Clinical criteria for recipient
The Model for End-Stage Liver Disease (MELD) score is an excellent predictor of severity and short-term survival in patients with liver cirrhosis and hence it forms the basis of prioritizing organ allocation in DDLT. It is a multiparametric mathematical score calculated using the patient’s serum bilirubin, the international normalized ratio (INR) and serum creatinine. It ranges from 6 to 40, with higher scores indicating a sicker patient and hence more urgent need of liver transplant. MELD scores can be adjusted for patients with HCC who are transplant candidates, depending on the stage of the disease.
‘High – urgency allocation’ is generally only possible in case of acute liver failure (ALF) or for retransplantation.
Imaging evaluation in recipient
Imaging assessment in recipients should include:
- • Evaluation of the liver parenchyma and vasculature,
- • Exclusion of intrahepatic and extrahepatic malignancy,
- • Assessment of known or incidentally diagnosed HCC and exclusion of any significant tumour thrombosis or metastasis which will contraindicate the transplant,
- • Patency and evaluation of flow in PV, SMV and SV
- • Extensive perihepatic varices,
- • Celiac stenosis,
- • Hepatic arterial anatomy,
- • Splenic artery aneurysm, and
- • Location of a transjugular portosystemic shunt (TIPSS).
To be able to give all the necessary information while reporting pretransplant cross-sectional imaging, it is very important to know the transplant surgeon’s perspective and their expectations (Table 9.14.3).
Evaluation of liver parenchyma and vasculature
The typical cirrhotic liver morphology shows hypertrophy of the caudate lobe and the left lateral liver segments (segments II and III), with atrophy of the remaining liver and surface nodularity. There is also widening of the porta hepatis, enlargement of the interlobar fissure, expansion of the pericholecystic space.
The presence of splenomegaly, ascites and portosystemic collaterals allows for the documentation of portal hypertension.
Detailed description of all these features along with mention of liver size, ‘wrap around’ nature of enlarged caudate lobe (which makes hepatectomy more complicated) and vascular information including large shunts and collaterals are essential parts of the evaluation.
Acute liver failure (ALF)
In patients with ALF, usually massive hepatocyte necrosis occurs in the acute phase, and if patient survives beyond this, then liver cell regeneration in the form of regenerating nodules and fibrosis is seen. Computed tomography (CT) findings of acute hepatic failure include decreased liver volume (due to massive necrosis), diffuse or localized areas of hypoattenuation and heterogeneous hypo enhancement (corresponding to areas of necrosis) in the liver, dilatation of the portal vein, narrow or nondepicted hepatic veins. Focal or diffuse areas of hyperattenuation (corresponding to areas of regeneration) are seen in the later stages of recovery in ALF.
Vascular information in recipient
Portal vein
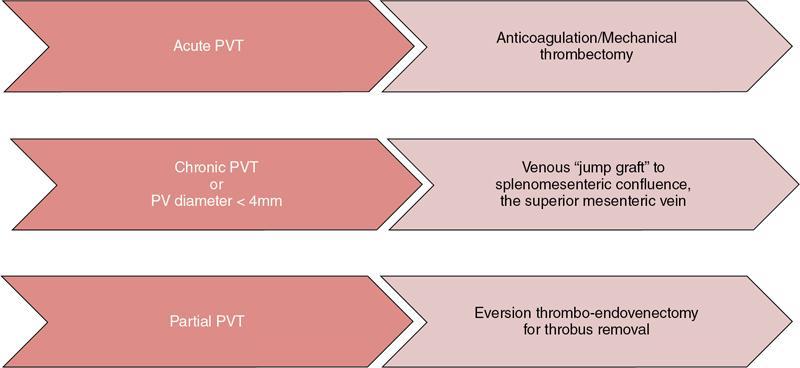
Patency of portal vein is one of the most important information required for planning of the transplant. The prevalence of portal vein thrombosis (PVT) is estimated to be approximately 10% in patients undergoing liver transplant. In cirrhotic patients who are candidates for liver transplants though presence of PV thrombosis does not preclude surgery, PVT is associated with greater operative technical difficulties and risk for rethrombosis. Fig. 9.14.1 summarizes management of various types of portal vein thrombosis in pre-transplant individuals.
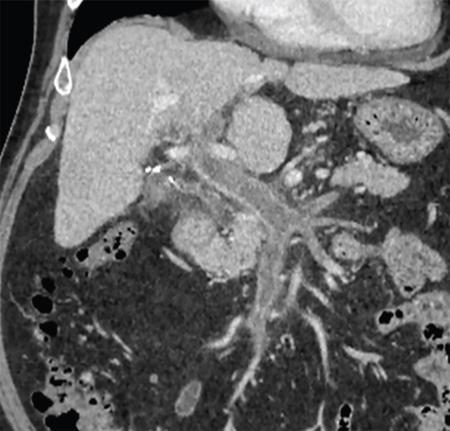
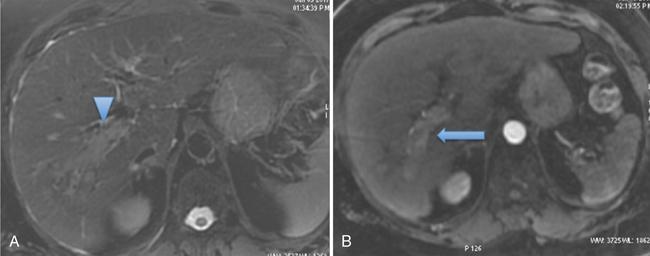
While evaluating PVT, it is essential to distinguish between bland (Fig. 9.14.2) and tumour thrombus as presence of portal vein tumour thrombus (PVTT) in major portal vein can contraindicate the liver transplant. Imaging characteristics of tumour thrombus include a contiguous thrombus with an adjacent mass showing characteristic imaging features of HCC or other malignancy, distension of the portal vein by endoluminal thrombus showing contrast enhancement on the postcontrast arterial phase (Fig. 9.14.3), and thrombosis in the setting of significantly elevated alpha-fetoprotein.
It is important to know presence and extent of portal vein thrombus before liver transplant because any unexpected thrombosis or narrowing of portal vein, can increase the operative time for vascular reconstruction, will need modifications in the surgical techniques like requirement for graft placements and also sometimes can result in graft ischemia due to prolongation of time taken for these. On the other hand, prior knowledge of PVT can help the surgeon to plan appropriate surgical strategies/procedures like intraoperative thrombectomy in case of acute or subacute thrombus or planning of venous ‘jump – grafts’ to SMV or splenic vein in case of chronic PVT (refer to Fig. 9.14.1).
It is also important to note the diameter of the portal vein as size discrepancy between the donor and recipient PV may also necessitate a venous graft to connect the SMV to the PV (Fig. 9.14.4). Flow pattern in the portal vein also needs to be assessed and in cases of centrifugal and reduced flow in the PV, ligation of any major shunt or collateral can be considered to achieve optimal PV flow for the transplant liver to avoid posttransplant PV thrombosis.
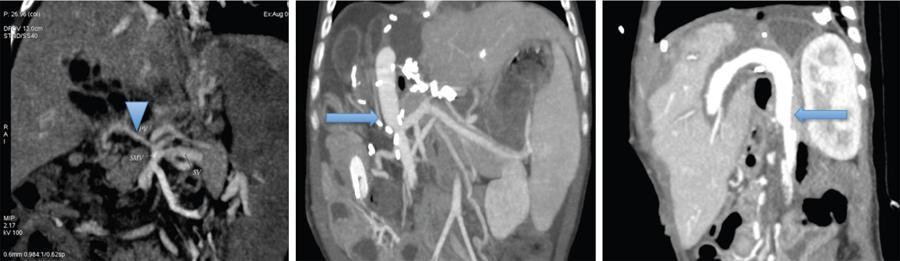
Complete chronic thrombosis of entire portosplenic axis and superior mesenteric vein system or diffuse splanchnic venous thrombosis is generally considered a contraindication for liver transplant. Though surgical techniques for modification of portal inflow like portal vein arterialization, cavo portal hemitransposition and reno portal anastomosis, and finally combined liver and small bowel transplantation are all possible alternatives to deal with this condition, it is a serious challenge to the liver transplant surgeon.
Preoperative identification of portosystemic shunts and varices
This is also very important, as these may be ligated at surgery to optimize blood flow towards the graft liver. Closure of large shunts can be planned preoperatively with percutaneous intervention (Fig. 9.14.5) if there is attenuation of portal vein calibre and reversal of flow seen in the portal vein in preoperative Doppler imaging. Preoperative identification of these may also minimize dissection time and risk of haemorrhage.

Evaluation of TIPS shunt
Patients on the waiting list for liver transplantation often require treatment of complications related to portal hypertension such as gastrointestinal bleeding, refractory ascites or vascular thrombosis. In addition to medical or endoscopic treatments, reduction of portal hypertension can be achieved with the help of Trans Jugular Intrahepatic Portosystemic shunt (TIPS); if other interventions fail to sustain their candidacy for LT. A specific indication to TIPS in patients listed for liver transplant is to maintain patency of portal vein if there is a risk of occlusion.
During TIPS placement, the superior tip of the shunt is normally in the right hepatic vein (RHV) or inferior vena cava (IVC) and the inferior tip is in the right portal vein or main portal vein (Fig. 9.14.6C). Shunt-related vessel lumen stenosis and migration of the shunt are important complications in patients with TIPS that can affect transplantation by increasing the time spent on bypass and complexity of transplant operation.

The extent of stent placement into the portal vein and hepatic vein need to be closely examined as the stent can cause inflammatory changes in the vessel wall which can lead to scarring and narrowing of the vein.
Superiorly migrated stent tip can lie in the intrahepatic or suprahepatic part of the IVC. In such instances, there could be scarring and narrowing of the cava, which may necessitate resection and supradiaphragmatic dissection or may even preclude living donor transplantation in which the recipient’s IVC is normally left intact.
In cases of low inferior shunt tips or inferior stent migration, the shunt extends into the extrahepatic part of the main portal vein, this portion of the portal vein may also need resection due to scarring of the endothelium. This results in a decreased length of portal vein available for anastomosis to the donor liver. The relationship and distance of the stent tip in the main portal vein to the splenomesenteric confluence can be documented and better shown on three-dimensional (3D) images.
Celiac artery stenosis and hepatic artery
Presence of significant stenosis of celiac artery in a potential transplant recipient is an important finding to be noted as this can cause hypoperfusion to the graft after liver transplant; which may lead to graft dysfunction.
Celiac artery narrowing could be due to median arcuate ligament which is prominent diaphragmatic crura causing compression of the proximal celiac artery or it could be atherosclerotic. It is critical to distinguish median arcuate ligament syndrome from atherosclerosis, as the management of these conditions is completely different.
Diagnosis of median arcuate ligament syndrome before liver transplantation can help in formulating an appropriate surgical plan for division of the ligament during the transplant. It is best appreciated on arterial phase in sagittal reconstructed MIP (maximum intensity projection) images of CT angiography, seen as focal narrowing of the superior aspect of the proximal celiac trunk forming a hooked or ‘J’ appearance as seen in Fig. 9.14.7 (which is accentuated during end-expiration and lessens during end-inspiration), with poststenotic dilatation or evidence of collateral formation, in absence of associated atherosclerosis.
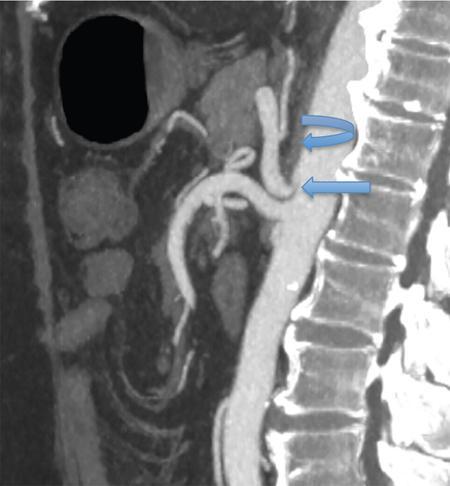
If the stenosis is due to atherosclerosis, an alternative inflow tract is created, usually by placement of an interposition conduit from a cadaveric donor between the recipient infrarenal aorta or right common iliac artery and the donor hepatic artery.
An alternate inflow tract is produced if the stenosis is due to atherosclerosis, usually by placing the interposition conduit from a cadaveric donor between the recipient infrarenal aorta or right common iliac artery and the hepatic artery donor.
Anatomy of hepatic artery, predominantly the origin of the artery and whether it is a replaced artery, is important to document, as this information can be helpful in planning posttransplant interventions in case of hepatic artery thrombosis (HAT) where sometimes the arterial stump can be difficult to identify due to complete occlusion.
In patients with prior TACE or TARE, the unused artery is preferred for anastomosis and hence knowledge of arterial anatomy is important.
Splenic artery aneurysms (SAA)
The incidence of SAA significantly higher in adult patients with cirrhosis and has been reported to be between 7% and 17% and these are almost always asymptomatic, diagnosed incidentally on pretransplant evaluation CT scan (Fig. 9.14.8). This increased incidence has been attributed to the increased flow in the splenic artery associated with portal hypertension. Preoperative identification and treatment of the SAA is critical as there is an increased risk of rupture after liver transplantation, especially in larger aneurysms.
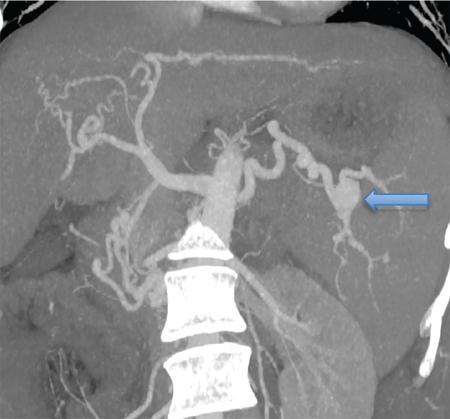
The size, number and location of the splenic artery aneurysm need to be noted.
Pretransplant intervention is usually indicated for SAA more than 20 mm in size.
IVC
In cases with IVC thrombosis or narrowing, the knowledge of extent of the involved segment is necessary as outflow reconstruction is planned preoperatively based on imaging findings.
Hepatocellular carcinoma
HCC is the commonest primary malignant tumour seen in the cirrhotic patients and hence screening for HCC is regularly performed in these patients. Presence of HCC does not preclude transplantation, and in fact, it is one of the indications for liver transplants. Liver transplantation serves as the best treatment for early-stage HCC, as it not only treats the tumour but also the underlying liver disease (the main risk factor for the development of new tumours). However, careful selection of the patients is essential to achieve best and curative results in these patients and avoid early recurrence.
All the major criteria for liver transplant take into account the number and size of the HCCs and hence is important to assess in detail. Any major vascular invasion or tumour thrombus in main and primary branches of the portal vein by tumour and extrahepatic disease are contraindications for liver transplant and hence should be carefully looked for.
Following criteria (Table 9.14.4) are most often used to decide the suitability of transplant recipient in presence of HCC, though the guidelines vary widely in different centres.
Milan’s criteria was proposed in 1996 by Mazzaferro et al. Excellent outcomes are seen after LT in patients fitting with Milan’s criteria. However, strict adherence to Milan’s criteria may deny chance of transplant to patients with slightly more advanced HCC who may still benefit and have acceptable outcomes after transplant. This was the rationale behind the development of the University of California San Francisco (UCSF) criteria (Fig. 9.14.9) which showed recurrence-free survival rate comparable to the Milan criteria at the same time expanding the opportunity of LDLT to more patients.

Many countries follow the Milan and UCSF criteria as general guidelines to cadaveric liver allocation. However, individual assessment of each case in a multidisciplinary meeting is more appropriate before being subjected to surgery.
Many of these patients undergo bridging therapies like TACE or RFA while on waitlist for liver transplant, evaluation of treatment response to these procedures during follow-up imaging to exclude disease progression is also important to assess before liver transplant.
Other than assessment of the HCC and hepatic vascular involvement, it also essential to rule out any metastatic disease in lymph nodes, lungs, skeletal system and other possible sites; as this is an absolute contraindication for liver transplantation.
Cholangiocarcinoma
Cholangiocarcinoma, the second most common primary liver tumour, also has strong association with liver cirrhosis and conditions like primary sclerosing cholangitis and is associated with poorer prognosis as compared to HCC.
Preoperative detection of cholangiocarcinoma, was initially considered a contraindication to liver transplantation due to a high recurrence rate. Though resection is the treatment of choice in case of cholangiocarcinoma, patients with underlying advanced liver disease may not be suitable to undergo major surgical resection due to the risk of hepatic failure.
Possibility of liver transplant is now being considered for such selected patients, like hilar Cholangiocarcinoma under a strict neoadjuvant chemoradiation protocol and with careful follow-up while listed, to identify tumour progression and avoid posttransplant recurrence.
Table 9.14.5 summarizes imaging evaluation in the transplant recipient.









