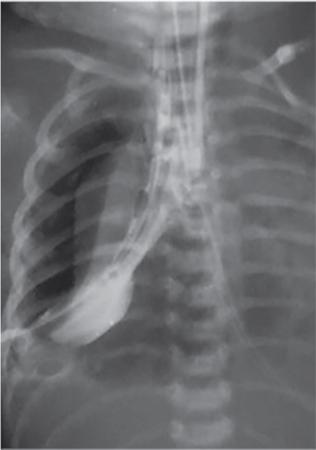
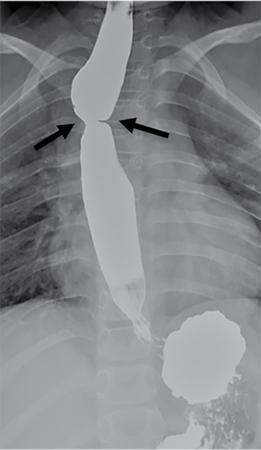
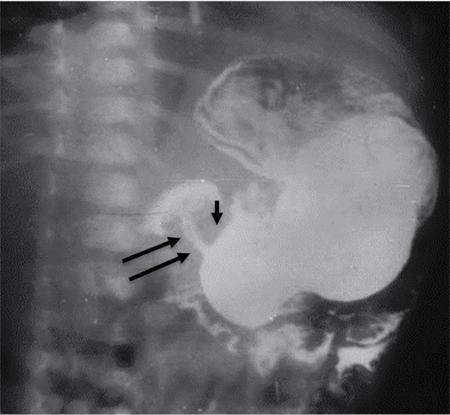
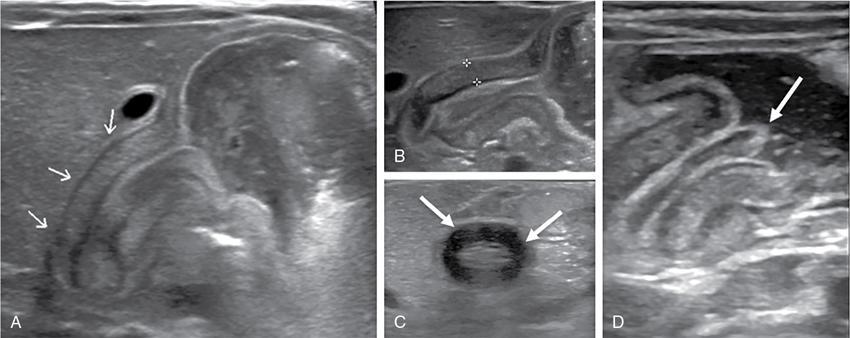
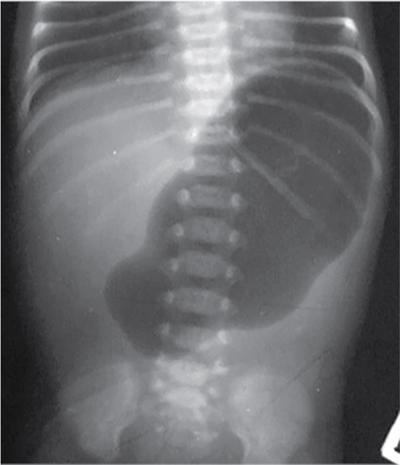
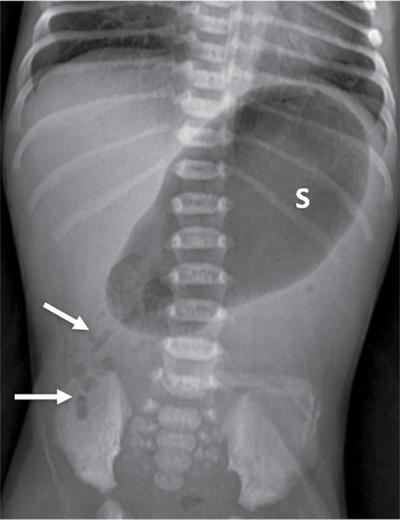
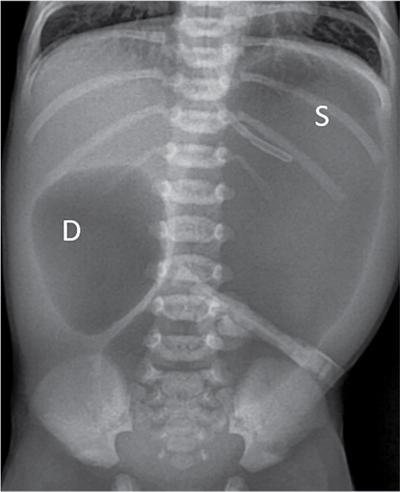
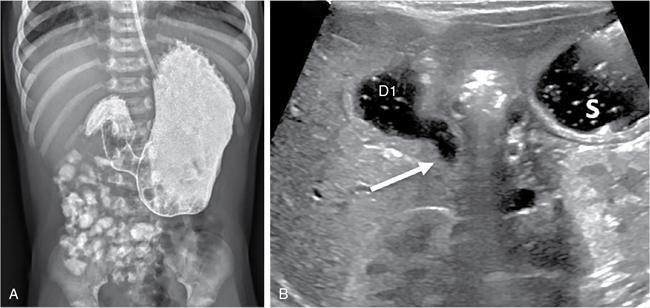
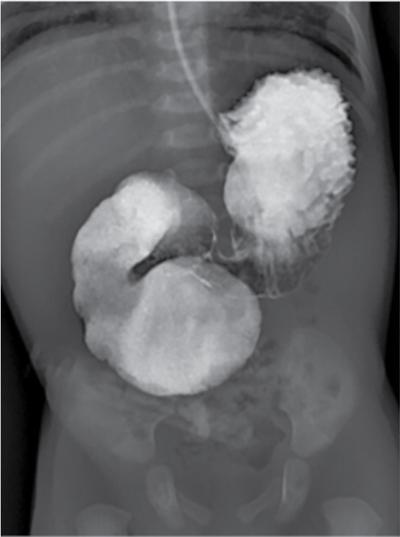
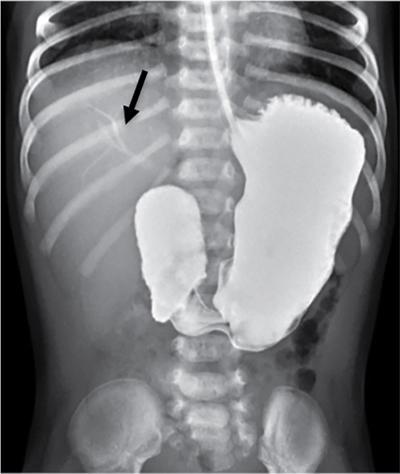
C.V. Kanimozhi, S. Muralinath, Raveendran J. Many of the important congenital gastrointestinal abnormalities present acutely in the newborn; some present much later. These include obstructive gastrointestinal lesions that present with vomiting, abdominal distension and not having passed meconium. The imaging modalities that are most commonly used in the evaluation of neonatal gastrointestinal abnormalities are plain films, contrast studies and ultrasound. Plain films remain a valuable tool in the assessment of abdominal abnormalities in the neonate. They are also an excellent guide to determining the next proper imaging study to perform for a particular problem. Certain neonatal bowel disorders have a pathognomonic appearance on radiographs obviating the need for further imaging. The essential and basic view to be done at the bedside is the supine view of the abdomen. In the evaluation of abnormal intra-abdominal gas patterns, alternative decubitus views, prone and supine cross-table lateral views may be taken. Erect view is not utilized in neonatal practice at the bedside as it is impossible to restrain a neonate in that position and unnecessarily subjects the neonate to stress, which may worsen the condition of an already sick baby. Evaluation of the bowel gas pattern and the anatomic localization of the intra-abdominal gas is the key to diagnosis in the evaluation of diseases of the GI tract. The bowel gas in the neonate is essentially swallowed air (Box 7.8.1). Movement of Air Through the GIT in a Term Neonate Most infants pass meconium by 24 hours of life. In order to interpret the bowel gas pattern and determine if an abnormality is present, it is crucial to know the age of the neonate (hours since birth) at the time the radiograph was taken. When there is impaired swallowing, such as due to CNS depression or in prematurity, radiographs show diminished bowel gas. In cases of bowel atresia, radiographs show absence of bowel gas in distal segments. Thus, the distribution of bowel gas and its pattern serve as pointers to the diagnosis. After the radiograph has been done, depending on the findings, an ultrasound or fluoroscopic contrast study may be performed next. The radiologist should tailor the contrast study to suit the particular patient and the clinical question that needs to be answered. In cases of suspected high GI obstruction, an upper GI contrast series is study of choice; in cases of suspected low GI obstruction a contrast enema should be performed to delineate the anatomy. When performing fluoroscopic studies, it is important to bear in mind the radiation dose and to adopt the ALARA principle. Minimizing radiation exposure in children is important because they are more sensitive to the effects of radiation and have a longer life expectancy than adults. The technical parameters must be adapted to paediatric imaging. Ultrasound is a useful bedside tool particularly in the sick neonate. In neonates, the sonographic image resolution is excellent due to the lack of significant body fat. In neonates, exquisite images of the abdomen can be obtained with the high-resolution ultrasound probe, which permits imaging the bowel wall in great detail. In addition, it can also reveal other causes for abdominal distension or vomiting. In this chapter, we will discuss the aetiology, clinical presentation and imaging appearances of congenital neonatal gastrointestinal abnormalities. We will also discuss common acquired abnormalities that present in the neonatal period, namely necrotizing enterocolitis. Congenital oesophageal malformations of the newborn include the various types of atresia (with and without fistula) and oesophageal duplications. Oesophageal atresia with or without tracheoesophageal fistula (TEF) is a common congenital anomaly that manifests in the neonatal period. It is also the most important congenital malformation of the oesophagus. Oesophageal atresia is commonly associated with other anomalies of the gastrointestinal tract such as imperforate anus, pyloric atresia, duodenal atresia and annular pancreas and less frequently with anomalies of the cardiac and genitourinary systems. About 20% of patients with oesophageal atresia have the VACTERL syndrome (vertebral anomaly, anorectal atresia, cardiac lesion, tracheoesophageal fistula, renal anomaly, limb defect). Oesophageal stenosis and webs may be associated with oesophageal atresia. The aetiology of oesophageal atresia and TEF is not completely understood. Faulty separation of the primitive trachea and oesophagus is the most widely accepted theory. The way the primitive foregut cleaves into a ventral (tracheal) passage and a dorsal (oesophageal) passage is a complex process and is poorly understood. Folds developing from the longitudinal ridge is said to separate the two. When the separation has an extreme tracheal bias, an oesophageal atresia occurs and if the bias is towards the oesophagus, then a tracheal atresia ensues. Incomplete separation may result in fistula of various types. Oesophageal atresia and TEF are classified depending upon the absence or presence and location of the fistula between the oesophagus and the trachea (Fig. 7.8.1). Neonates present within a few hours of birth with excessive salivation, drooling, cyanotic spells and regurgitation of feeds. The inability to successfully pass a nasogastric tube strongly suggests oesophageal atresia. Newborns with H-type fistula may have feeding difficulties and choking, but often the presentation and diagnosis is only later in infancy when the patient presents with cough, choking episodes and recurrent pneumonia. The role of imaging is to diagnose, define the anatomy to the best extent possible and evaluate for other anomalies to provide a clear picture for the paediatric surgeon. For example, it is important for the surgeon to know the side of the aortic arch in order to lateralize the surgical approach. Chest X-ray and Echo can localize the arch and determine if an anomalous right-sided aortic arch is present. Plain films of the chest including abdomen need to be obtained. The radiograph will demonstrate the air-filled distended proximal oesophageal pouch. If a nasogastric tube has been inserted, the coiled tube will be seen within the pouch. Absence of bowel gas in the abdomen indicates oesophageal atresia without a distal fistula – types A and B have this radiographic appearance (Fig. 7.8.2). The presence of gas in the gastrointestinal tract indicates the presence of a distal fistula – types C and D (Fig. 7.8.3). Contrast studies to delineate the proximal pouch are generally not indicated. Coiling of the nasogastric tube in the proximal pouch confirms the diagnosis. Chest radiographs frequently show changes of consolidation due to aspiration pneumonia, most often involving the right upper lobe (Fig. 7.8.4). In cases of H-type fistula, plain films are nonspecific and the diagnosis depends on contrast studies. Radiological diagnosis is made by means of a tube oesophagogram. This study has the potential risk of aspiration and the neonate may desaturate quite rapidly. Hence, it is important to have neonatal emergency resuscitation at hand. The tube oesophagogram is performed under fluoroscopy with the baby in lateral- or steep-prone oblique position. The NG tube is placed at the oesophagogastric junction and is gradually withdrawn while simultaneously injecting contrast. Nonionic low-osmolar contrast agent should be used. Most of the H-type fistulas are located in the lower cervical and upper thoracic regions with the fistula typically coursing upward and forward from the oesophagus. In the presence of a fistula, the contrast will be seen passing into the trachea in an upwardly oblique configuration. Aspiration during the study can also cause the contrast to appear in the tracheal tree and the lungs. It needs to be established whether the contrast has passed through the fistula or whether aspiration has occurred. Recording of the fluoroscopic run and reviewing the images may be required to make this distinction. Repeat examination may be required. Demonstration of H-type fistula can be difficult radiologically. In such patients, bronchoscopy with transfistula guidewire placement has been found to be of use. MRI – High resolution structural MRI is a new technique that is being used to visualize tracheobronchial anatomy. The disadvantage with radiographs is that one cannot determine the anatomic subtype of TEF, the site of the TEF or the length of the gap between the atretic segments. Using ultrashort echotime (UTE) MR imaging, it is possible to visualize the exact site of the fistula and assess the size of the oesophageal gap. This can aid presurgical planning and help identify infants at risk for complications, such as those with a long oesophageal gap. Imaging plays an important role in the evaluation of early and late complications of atresia repair. Early complications include anastomotic leaks and are seen in about 10%–20% of cases. Oesophageal contrast study, using a nonionic low-osmolar contrast agent, is performed to demonstrate the leak at the anastomotic site (Fig. 7.8.5). Leaks are associated with a greater incidence of subsequent stricture development. Anastomotic stricture is a common complication seen in about 30%–40% of cases. Often there is a slight narrowing at the site of repair without the patient having any difficulty in swallowing (Fig. 7.8.6). The anastomotic narrowing is clinically relevant only if the patient has dysphagia; such cases respond to dilatations. Recurrent TEF occurs in 5%–10% of cases after oesophageal atresia repair. Late complications include dysphagia, gastroesophageal reflux, tracheomalacia and chest wall deformities. Dysphagia can occur post-operatively due to abnormal oesophageal motility. Oesophageal dysmotility can be caused by abnormal neural development of the oesophagus or may result from complication of atresia repair. Gastroesophageal reflux affects 40%–65% of patients following oesophageal repair. It may be due to an intrinsic deficiency in the motor function of the oesophagus; this may get exacerbated postrepair due to an alteration of the anatomical gastroesophageal junction. Oesophageal atresia key imaging finding – X-ray shows NG tube coiled in proximal oesophageal pouch. The stomach is divided into the fundus and body proximally and the antrum distally. The antrum is divided by the sulcus intermedius into a proximal pyloric vestibule and a distal pyloric antrum or pyloric canal. This canal terminates into the pyloric sphincter beyond which is the duodenum. Congenital anomalies of the stomach that manifest in the neonatal period are uncommon. Hypertrophic pyloric stenosis (HPS) is the most common surgical cause of vomiting in infants. The incidence is 2–5 per 1000 live births, with geographic and racial variations. Compared to Caucasians, HPS is less common amongst Asian populations. The male to female ratio is approximately 4:1, occurring more commonly in the first-born child. The precise aetiology is unknown. Overactivity or prolonged spasm of the circular muscle of the pyloric antrum is thought to be the primary problem in these infants. This spasm leads to muscle hypertrophy and obstruction. Multiple hypotheses have been proposed for this prolonged spasm. One is that some infants are genetically predisposed to increased gastrin production which leads to a vicious cycle of hyperacidity, increased gastric contraction and secondary development of muscular hypertrophy. Most infants present after the first week of life and before 3–4 months of age. Typical symptoms include projectile nonbilious vomiting, regurgitation and difficulty in feeding. The gastric outlet obstruction can lead to emaciation. The distended stomach with active peristaltic activity may be visible through the thin abdominal wall. In the past barium upper GI series was the mainstay diagnostic tool for HPS. Prior to the barium study, the stomach may need to be decompressed via a nasogastric tube. The classic sign on a barium study is the ‘string sign’ (Fig. 7.8.7) produced by a thin stream of barium in an elongated narrowed pyloric canal; the canal typically curves upward. In severe cases, complete obstruction will lead to the ‘beak sign’, which is produced by beaking of the contrast as it enters the pyloric canal. With lesser degrees of obstruction, the ‘double track sign’ is seen. This is produced when the pyloric canal is flattened and the barium accumulates in the crevices along either side of the flattened canal. Pylorospasm can transiently mimic the findings of HPS. Currently, ultrasound is the modality of choice for the diagnosis of HPS and upper GI studies are seldom used. Ultrasound is considered the ‘gold standard’ as it allows direct examination of the pyloric muscle and also provides the ability to perform a dynamic study. The ultrasound examination is performed with a linear high-frequency transducer (6–10 MHz). The examination begins with the baby in supine position. First, the antropyloric region is identified; the pylorus is usually located medial and posterior to the gall bladder. The normal pyloric muscle is a thin hypoechoic layer that measures 2 mm or less. The abnormal pyloric canal shows thickening of the muscle and the mucosa to varying degrees. In cross section, it is seen as a hypoechoic structure likened to a doughnut. The length of the canal is measured in long section where its appearance is likened to the cervix. The sonographic diagnostic criteria for HPS are a thickened muscle layer measuring ≥3 mm and an elongated pyloric channel ≥15 mm (Fig. 7.8.8). The diameter of the pylorus in cross section was also one of the originally used measurements. However, due to a significant overlap between normal and abnormal, this measurement is no longer utilized. Additional ultrasound findings of HPS are hypertrophy of the pyloric mucosa and a distended, actively peristalsing stomach. The hypertrophied redundant mucosa projects into the antrum analogous to the ‘nipple sign’ in barium studies (Fig. 7.8.8D.). Colour Doppler imaging shows increased flow in the muscle and mucosa. The above-described measurements of pyloric muscle thickness and canal length are vital to diagnosis. However, dynamic assessment is equally important. One must evaluate the antropyloric region over a period of time to ensure persistent stenosis without evidence of relaxation and opening of the canal. A distensible antropyloric region with normal passage of gastric contents excludes HPS. Transient contractions of the pylorus or pylorospasm can mimic pyloric stenosis in both measurement and appearance. With observation over a period of time, opening of the pyloric canal may be visualized. Hence the key to diagnosis of HPS is persistent non-relaxing thickened musculature. Gas in the stomach may obscure the pylorus. To avoid this, place the infant in an oblique position, right side down which will allow the antrum to fill with fluid. Conversely, an overdistended fluid-filled stomach can displace the pylorus dorsally. In such cases, turning the infant left side down will displace the fluid towards the fundus and will allow the pylorus to rise to a more anterior position. At times, equivocal examinations may occur where the muscle measures 2–3 mm in thickness and does not relax. In these patients, careful monitoring with repeat ultrasound is warranted. In a few cases, the abnormalities advance overtime to fully developed HPS. Hence in such equivocal cases if vomiting continues, follow-up ultrasound is advisable. Surgical pyloromyotomy is the treatment for HPS. On ultrasound, the myotomy site can be seen as an interruption in the hypoechoic doughnut. It is important to be aware that postsurgery, the pyloric muscle may remain thickened for up to 5 months with a gradual return to normal thickness. In the first week after surgery, the muscle can be the same thickness as pre-op or even thicker. One should not be alarmed by this appearance. The dimensions gradually return to normal over the course of a few months. Postop if the child is thriving well, there is no need for imaging to be performed. HPS key imaging finding: On high-frequency ultrasound muscle thickness ≥3 mm and elongated pyloric channel ≥15 mm. Pyloric atresia is a rare anomaly comprising less than 1% of all intestinal atresias. Pyloric atresia is classified into three types (Fig. 7.8.9): The exact aetiology remains controversial. In the past, it was postulated that failure of recanalization of the gastrointestinal tract was the cause. Recent evidence suggests that pyloric atresia is the result of localized vascular occlusion. This vascular theory suggests that the atresia is due to focal ischemia secondary to intrauterine stress, vascular insult or anoxia. In complete obstruction, the presentation is in the neonatal period with nonbilious vomiting within the first few hours of life. Incomplete obstruction due to a diaphragm has a variable presentation depending on the size of the orifice. These patients can present later in life with intermittent vomiting or postprandial vomiting. In complete pyloric atresia, a dilated stomach is present on radiographs with absence of gas in the distal bowel. This is known as ‘single bubble’ appearance (Fig. 7.8.10). This appearance is diagnostic and in such cases, contrast studies are generally not required. In the membranous type, there is an opening in the centre of the diaphragm and hence, the obstruction is usually incomplete. On radiographs, the stomach is distended and lesser than normal volumes of air is present in the small bowel (Fig. 7.8.11). On upper GI barium studies, a diaphragm is seen as a thin (2–4 mm) linear filling defect that spans across the antrum with a normal pyloric canal. One needs to determine on fluoroscopy whether the membrane is causing significant obstruction or not. If obstructing, the diaphragm will balloon out with gastric peristalsis. In incompletely obstructing diaphragms, the edges of the diaphragm will be visible but there will no obstruction or ballooning. Pyloric atresia key imaging finding: X-ray abdomen – ‘single bubble’ appearance. Microgastria is an extremely rare congenital abnormality wherein the stomach is small, midline in position and typically associated with a dilated oesophagus. The stomach is represented by a small tubular structure without recognizable differentiation into fundus, body, antrum and pylorus. Agastria is the most extreme form of microgastria. Microgastria is frequently associated with other anomalies such as polysplenia-asplenia syndrome, malrotation, gastrointestinal atresia and vertebral, cardiac, renal and limb reduction anomalies (VACTREL association). Microgastria occurs due to arrest in early development of the foregut. In the 5th week of gestation, dilatation of the region of the future stomach occurs. Ninety-degree rotation of the stomach occurs in the 6th week, followed by development of the greater and lesser curves and growth of the fundus in the subsequent weeks. Arrest of this process leads to microgastria, with severity dependent on the stage at which arrest occurs. The development of spleen within the dorsal mesogastrium of stomach explains the association of microgastria and splenic anomalies. The association of limb, cardiac, tracheoesophageal, vertebral and renal anomalies has been attributed to impairment of early mesodermal development. The symptoms are related to the effects of the inadequate stomach and dilated oesophagus or due to the associated severe anomalies. These babies may present with feeding difficulties, failure to thrive, vomiting, recurrent aspiration and malnutrition due to severe gastroesophageal reflux. Radiographs may show an absent stomach bubble; in some cases, the shadow of the dilated oesophagus may be seen in the lower chest. On an upper GI study, the stomach appears small, tubular and midline in position. There is often associated severe gastroesophageal reflux and a very dilated oesophagus. The oesophagus dilates to take over the storage function of the inadequate stomach. Additional imaging studies are required to identify the associated anomalies. Key imaging finding: Upper GI study – Small, tubular stomach located in the midline. Congenital anomalies of the duodenum present with obstructive symptoms. The primary considerations for bilious vomiting in the neonate are duodenal atresia, duodenal stenosis, annular pancreas and malrotation with midgut volvulus. The most important investigation in the majority of these patients is an upper GI series as it accurately demonstrates the level and nature of duodenal obstruction. Duodenal atresia may be seen in isolation or in the setting of trisomy 21 (about 30% of patients have Down’s syndrome). Associated anomalies are frequent and include malrotation, oesophageal atresia, annular pancreas, biliary atresia, congenital heart disease and vertebral anomalies. Duodenal atresia is due to failure of gut recanalization during embryologic development leading to complete obstruction. Normal recanalization of the duodenum occurs between the 9th and 12th weeks of gestation. Failure of this process results in duodenal atresia. In most cases (roughly 80%), the atresia is distal to the ampulla of Vater. Neonates with duodenal atresia present with bilious vomiting in the first few hours of life. In those patients where the atresia is proximal to the ampulla, the vomitus will be nonbilious. In duodenal atresia, air is present in the stomach and duodenum with no gas in the distal intestinal tract. Dilatation of the stomach and the duodenum produces the characteristic ‘double bubble’ appearance on radiographs (Fig. 7.8.12). This appearance is diagnostic and rarely requires further imaging. Key imaging finding: X-ray abdomen – ‘double bubble’ appearance Duodenal stenosis and duodenal web cause partial obstruction due to a narrowed segment or thin membrane, respectively. As with duodenal atresia, both these conditions show a strong association with malrotation, annular pancreas and a preduodenal portal vein. Duodenal web and stenosis is due to incomplete recanalization of typically the second part of the duodenum. In babies with duodenal stenosis, the presentation and clinical findings depend on the degree of stenosis. Those with less severe forms of stenosis may present later in life. Radiographs in duodenal stenosis and web show a distended stomach and duodenum. Since the obstruction is incomplete, gas will be present in the distal loops. On upper GI study, duodenal stenosis is seen as a focal narrowing in the second part of the duodenum (Fig. 7.8.13). In duodenal web, a thin membrane/diaphragm is seen that partially obstructs the lumen, usually in the second segment of duodenum. The membrane is often difficult to demonstrate (Fig. 7.8.14). A ‘windsock deformity’ may also be seen; this appears as a proximal dilated duodenal segment with contrast outlining a thin web that bulges into a distal nondilated segment. This windsock duodenum is seldom seen in the neonatal period as this appearance is due to stretching of the membrane over time. Key imaging finding: Upper GI study – narrowing in second part of duodenum. Annular pancreas is an anomalous band of pancreatic tissue that encircles the second portion of duodenum. Annular pancreas may result in extrinsic duodenal obstruction. Often there is associated intrinsic duodenal abnormality viz. some degree of duodenal atresia, web or stenosis. As in duodenal atresia, associated anomalies such as trisomy 21, cardiac defects, malrotation, oesophageal atresia and anal atresia can occur. The pancreas arises as two endodermal outgrowths, ventral and dorsal buds, from the duodenum. The head of the pancreas derives from the smaller ventral bud. Annular pancreas occurs if the ventral bud becomes tethered to the duodenum or it fails to rotate completely before fusion with the dorsal bud. The clinical presentation is similar to duodenal atresia or stenosis. If the obstruction is complete, presentation is in the neonatal period with bilious vomiting. Cases of incomplete obstruction may remain asymptomatic until adulthood. Annular pancreas is the second most common condition to produce a ‘double bubble’ appearance on radiographs. Plain films will show dilatation of the stomach and duodenum. Upper GI contrast study will show narrowing of the second part of duodenum (Fig. 7.8.15). On ultrasound pancreatic tissue may be seen encircling the second part of duodenum (Fig. 7.8.16). At times the appearance may resemble a mass at the level of head of pancreas.
7.8: Neonatal gastrointestinal disorders
Introduction
Air seen in stomach
At birth
Air in duodenum
30–60 minutes
Air in jejunum
2–4 hours
Air in ileum
4–6 hours
Air in colon
12–18 hours
Air in rectum
24 hours
Oesophagus
Oesophageal atresia and tracheoesophageal fistula
Aetiology
Classification
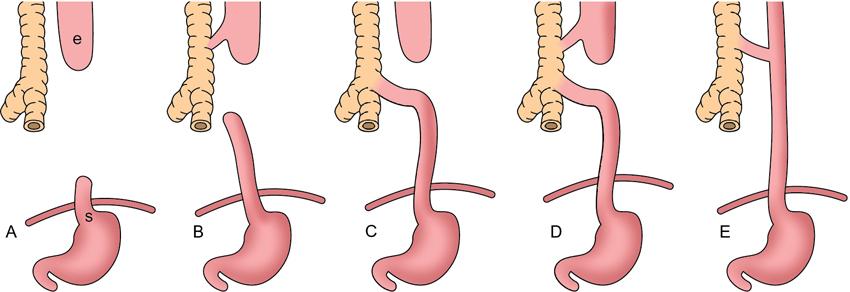
Presentation
Imaging
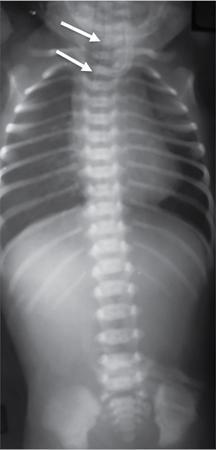
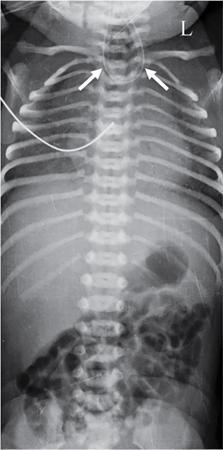
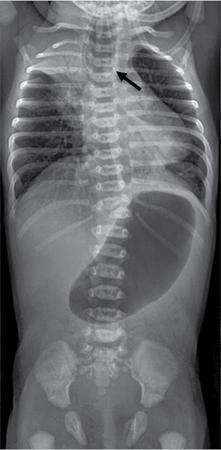
Postop complications
Stomach
Hypertrophic pyloric stenosis
Aetiology
Presentation
Imaging
Pitfalls.
Post-treatment imaging
Pyloric atresia and antropyloric membrane
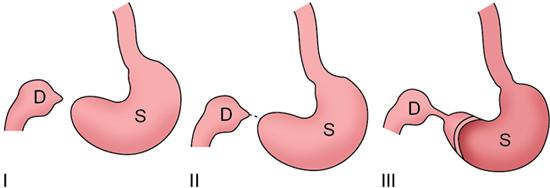
Aetiology
Presentation
Imaging
Microgastria
Aetiology
Presentation
Imaging
Duodenum
Duodenal atresia
Aetiology
Presentation
Imaging
Duodenal web and stenosis
Aetiology
Presentation
Imaging
Annular pancreas
Aetiology
Presentation
Imaging
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








