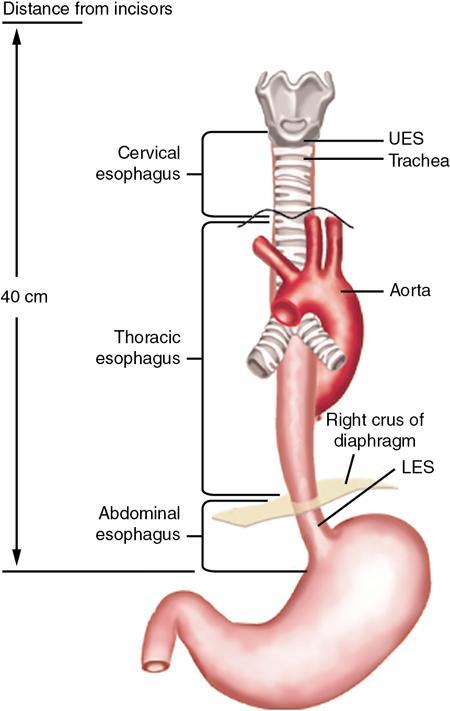
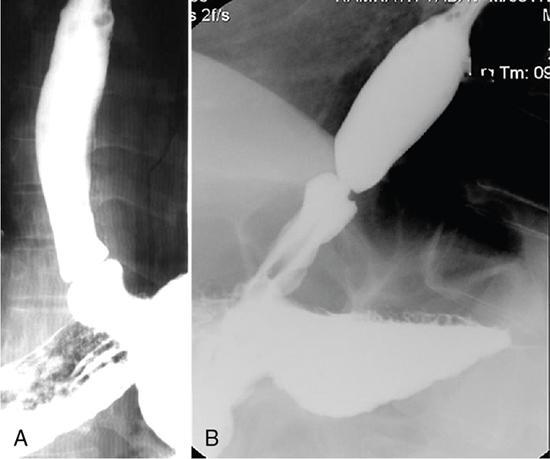
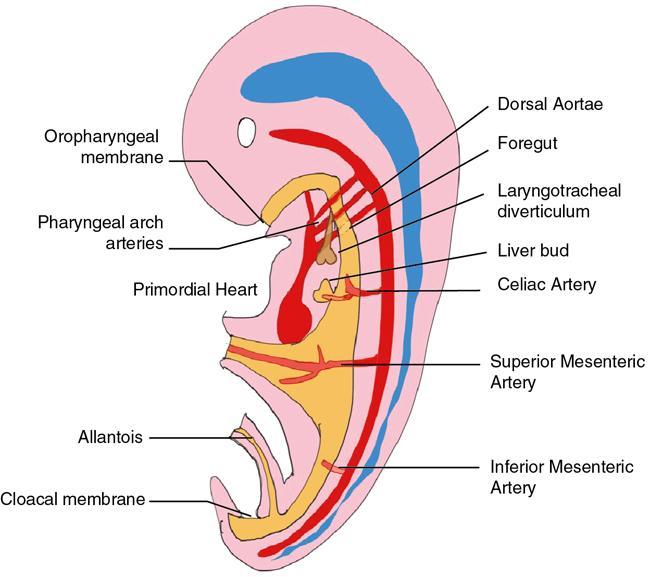
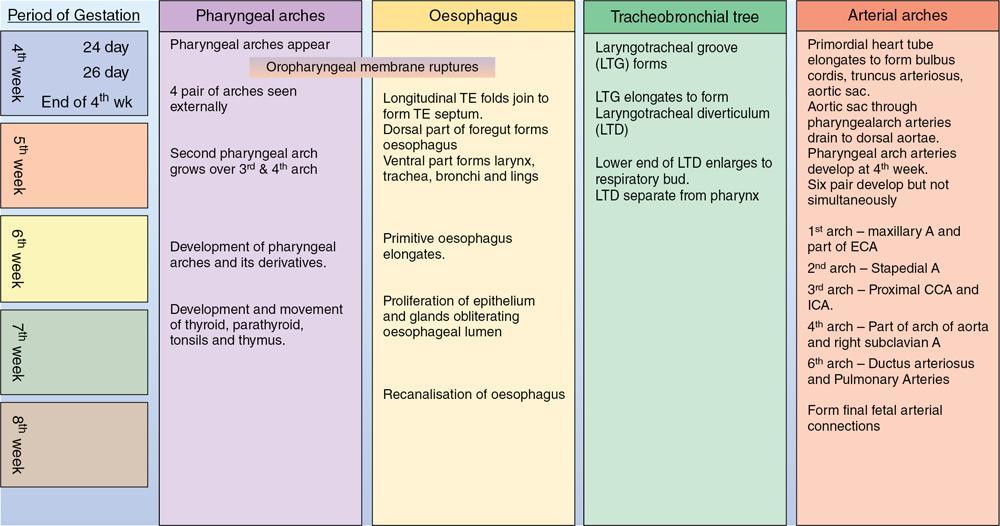
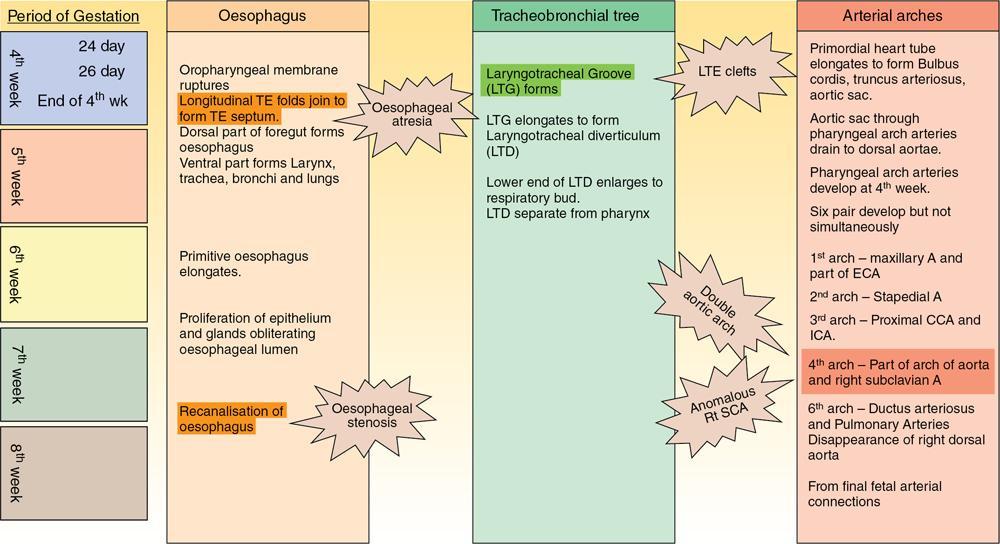
Gaurav Goswami, Gurdarshdeep Singh Madan, Ankita Dhawan Oesophagus is a fibromuscular tube responsible for conduction of food from oral cavity till the level of stomach. It is a hollow structure of approximately 25 cm. Proximal extent of oesophagus, that is cricopharynx begins at the level of C6 vertebral body. At this level, cricopharyngeus muscle merges with inferior constrictor muscle to form a functional zone known as upper oesophageal sphincter (UES). Oesophagus during its course through neck lies posterior to trachea and in thorax lies in posterior mediastinum. In mediastinum, oesophageal body courses to the left of the midline along left atrium, left main bronchus and aortic arch. It returns to the midline at level of T5. On plain radiograph, above T4 level, a sickle-shaped soft tissue shadow is seen overlying the tracheal air – where the trachea abuts the left lung – called the pleuroesophageal line. Similarly, azygoesophageal line is seen on the plain radiograph where the azygous vein and oesophagus abuts right lung (below T4 level). Oesophageal hiatus, present within right crus of diaphragm, is seen at the level of T10 vertebral body. Oesophagus through this hiatus enters the upper abdomen. An approximately 2–4 cm long segment of lower oesophagus is present in the hiatus. This segment shows asymmetrically arranged circular smooth muscles which forms the functional lower oesophageal sphincter (LES). This gastro-oesophageal junction is supported by various ligaments like phreno-oesophageal, gastrosplenic, phrenicolienal and gastrophrenic ligaments. These are responsible for strengthening of gastro-oesophageal junction. However, the most important phreno-oesophageal ligament (or membrane) is responsible for smooth longitudinal sliding of oesophagus through the diaphragmatic hiatus and it also acts as a demarcation between both thoracic and abdominal cavities. Oesophagus is thus anatomically divided into cervical, thoracic and abdominal parts. These parts have different arterial supply, venous supply and lymphatic drainage. Sensory, motor and parasympathetic fibres from vagus are responsible for nerve supply of all the parts of oesophagus. Vagal preganglionic nerve fibres in distal oesophagus are found to terminate within the Auerbach’s (myenteric) plexus. Similarly, Meissner’s plexus acts as a site for afferent impulses within the oesophageal wall. Structural relationship of oesophagus and its implications. These are depicted in Figure 8.1.1: For the purpose of staging of oesophageal cancer (AJCC), oesophagus is divided into upper, middle and lower parts on CT. Upper oesophagus comprises entire cervical oesophagus and thoracic oesophagus till the level of lower border of azygous vein, middle oesophagus from lower border of azygous vein till the lower border of inferior pulmonary vein and lower oesophagus from the lower border of inferior pulmonary vein to stomach including oesophago-gastric junction. The normal oesophageal wall is primarily made of four distinct layers, from inner to outer aspect: Oesophagus shows absence of serosa which is normally present in rest of the organs of GI tract. The distal oesophagus contains two rings – A ring and B ring (Schatzki’s ring) are seen at the proximal and distal to a mild fusiform dilatation of distal oesophagus called vestibule (Fig. 8.1.2). ‘A ring’ also known as muscular ring is present at the proximal extent of vestibule. It represents 4–5 mm band of hypertrophied muscle which can cause constriction at junction of vestibule and oesophageal lumen. It is normally lined by squamous epithelium. ‘A rings’ are rare seen. However, when symptomatic, they cause dysphagia to both solids and liquids. Symptomatic rings are treated endoscopically by dilators and botulinum toxin injection. ‘B ring’ is present at the distal end of the vestibule. It is relatively more common and in most cases seen in association with hiatus hernia. It is a thin membrane of squamous epithelium at its upper surface and columnar epithelium at lower surface. These rings comprise mucosa and submucosa. These rings can be congenital or acquired. Its relationship with GERD is documented. Usually these ‘B rings’ are asymptomatic but they can be symptomatic if they narrow oesophageal lumen to less than 13 mm. Symptomatic rings are treated with either bougie dilatation or mercury weight dilators. CLINICALLY RELEVANT EMBRYOLOGY OF OESOPHAGUS Gurdarshdeep Singh Madan Intrauterine life, especially the first 8 weeks, is extremely busy period in the development of the foetus. Any insult in this period will result in major congenital abnormality and a potentially fatal outcome. One should be mindful of the fact that different anatomically structures are developing simultaneously and not sequentially. Therefore, a specific teratogenic insult caused by drugs/radiation or IU infection like virus results in manifestations in more than one organ system. Over the course of this chapter, we will try to make embryology of oesophagus simple to understand and relate it to the simultaneous development of the respiratory system and embryonal arterial arches and also briefly mentioning their clinical relevance. At around 4th week of intrauterine life, a primordial gut is formed by the invagination of the dorsal yolk sac by lateral embryonal folds and folding of the head and the tail ends. Initially, the gut is a closed tube with oropharyngeal membrane at the cranial end and the cloacal membrane at the caudal end. The oropharyngeal membrane ruptures and orifice is formed around the 26th day. The cranial third of this alimentary system is called the ‘foregut’ (see Fig 8.1.3). Immediately following the formation of the primordial gut, a medial outgrowth develops caudal to the four pair of pharyngeal arches – known as the Laryngo-tracheal groove. By end of the first month (4th week) of intrauterine life, the laryngo-tracheal groove lengthen and protrudes caudally to form laryngo-tracheal diverticulum (LTD). The caudal end of LTD enlarges to form a ‘single’ globular respiratory bud from which both lungs develop. Simultaneously, the longitudinal tracheo-esophageal folds begin to form and start invaginating towards each other. By the end of the 5th week, the tracheo-esophageal folds fuse to form a complete partition, dividing the primitive foregut into ventral laryngo-tracheal tube and dorsal primordial pharynx and oesophagus. The laryngo-tracheal tube opens into the pharynx through a small opening known as the primordial laryngeal inlet (see Fig 8.1.4). Initially after its formation, the length of oesophagus is very short relatively as compared to the total length of primordial gut. However, the oesophagus elongates rapidly as compared to rest of the gut to attain postnatal relative length by the 7th week. During this time, the lumen of oesophagus is obliterated by proliferating epithelium and mucosal glands, which are derivatives of the endoderm. The recanalization of oesophagus is completed by end of the 8th week. The muscles of oesophagus are striated in superior third and derived from the mesenchyme of the fourth and the sixth pharyngeal arches. (Note: Fifth pharyngeal arch disappears.) The inferior third of oesophagus has smooth muscles developed from the splanchnic mesenchyme. The mid third of the oesophagus shows gradual change from striated to smooth muscles. The change from striated to smooth muscles in the oesophageal wall roughly corresponds to the level of arch of aorta in an adult. It must also be noted that both striated and smooth muscles of oesophagus are supplied by the vagus nerve (CN X) which is the nerve of fourth and sixth pharyngeal arches. The chart below illustrates the simultaneous development of pharyngeal arches, respiratory and alimentary system and briefly outlines the development of arterial tree (see Flowchart 8.1.1). The schematic diagram of the embryonal basis of congenital oesophageal abnormalities is attached below. The details of congenital anomalies are covered in relevant chapters (see Flowchart 8.1.2). IMAGING TECHNIQUES – OESOPHAGUS Gurdarshdeep Singh Madan Oesophagus like most other organs of the body may develop ailments – both benign and malignant. They may be congenital, presenting in neonatal period or later in life. The diseases may be structural affecting mucosa; they may also spare the mucosa or could be a transient motility disorder. Hence, the investigation chosen should have maximum cost-benefit to the patient as well as present minimum risk to the patient with judicial use of radiation. With advent of upper gastrointestinal endoscopy, it has replaced radiological studies as preferred investigation for multiple presentations. However, the utility of the radiological studies cannot be discounted due to their ability to provide information regarding motility as in upper gastrointestinal contrast studies and cross-sectional data for cancer staging. During the course of this chapter, we will discuss in brief various imaging modalities and techniques. Though endoscopy has largely displaced imaging studies as initial investigation of choice for dysphagia, their role in motility disorders is limited. Endoscopic ultrasound is investigation of choice for evaluating T1 and T2 oesophageal Ca. PET-CT is functional imaging which is gold standard in evaluating distant metastasis. CT scan is initial staging investigation for T3 and T4 disease and provides valuable information in preoperative planning and postoperative assessment. Contrast oesophagogram done with correct technique is an important investigation for evaluation of motility disorder and is a competent investigation in evaluation of structural/mucosal disorders. The choice of the imaging modality to evaluate a patient depends on patients’ condition, clinical suspicion and the risk benefit ratio to the patient. In a resource constraint setting, availability and affordability of the investigation is also equally important. Many international societies have propounded the guidelines for first investigation of choice. American College of Radiology appropriateness criteria (ACR-AC) for imaging studies in evaluation dysphagia has been adapted as Table 8.1.1. The investigation of choice in other clinical settings are covered in the respective chapters. Fluoroscopic biphasic oesophagogram Fluoroscopic single contrast oesophagogram Oesophageal transit nuclear medicine scan Fluoroscopic modified barium swallow CT neck and chest with or/and without IV contrast Fluoroscopy pharynx dynamic and static imaging Fluoroscopic biphasic oesophagogram Fluoroscopic single contrast oesophagogram Fluoroscopic modified barium swallow Oesophageal transit nuclear medicine scan CT neck and chest with or/and without IV contrast Fluoroscopy pharynx dynamic and static imaging Fluoroscopic single contrast oesophagogram CT neck and chest with IV contrast CT neck and chest without IV contrast Oesophageal transit nuclear medicine scan CT neck and chest with and without IV contrast Fluoroscopy pharynx dynamic and static imaging Fluoroscopic modified barium swallow CT neck and chest with IV contrast Fluoroscopic single contrast oesophagogram Fluoroscopic modified barium swallow Fluoroscopic biphasic oesophagogram Oesophageal transit nuclear medicine scan CT neck and chest with or/and without IV contrast Fluoroscopy pharynx dynamic and static imaging Mazzeo et al. and Carrascosa et al. have described technique of virtual oesophageal endoscopy with use of air insufflation via catheter inserted into the upper oesophagus and by use of effervescent agents like those used in fluoroscopic oesophagogram. However, despite these technological assessments, the sensitivity of CT scan in identifying T1 and T2 disease is limited. The CT scan is also limited in identifying normal-sized metastatic lymph node. The limitation can only be partially addressed by using morphological appearance like extracapsular spread and central necrosis. National Comprehensive Cancer Network (NCCN) guidelines for oesophageal and oesophagogastric Ca recommends CECT chest and abdomen with oral and IV contrast in staging assessment of cancers T3 and above. CT is indicated in assessment of early and delayed postoperative complications. Lantos et al. and Upponi et al. have demonstrated slightly better sensitivity of CT in detecting early postoperative complications. However, it is marred by poor specificity in the same period. In delayed postoperative dysphagia, use of CT oesophagogram has demonstrated increase in sensitivity in identifying recurrent disease, abnormality caused by surgical hardware/technique or a late onset seroma. CT oesophagogram (Table 8.1.3) can be performed by preparing 3% iodinated contrast in water or thickening agent like Simply Thick to form a honey- or nectar-like consistency swallowed on the table with last gulp taken immediately prior to the acquisition. The images are reconstructed in curved multiplanar reformats in soft tissue window. MRI and MR oesophagogram – Quint et al. was the first, however, unsuccessful attempt evaluation of oesophagus on a 0.35T MRI, 35 years back. Since then, there has been substantial advancement in the MRI technology with evolution of quicker and dynamic sequences, and advancement of k space filling technology leads to further shortening of scan times. Yamada et al. used MRI in vitro staging of Ca oesophagus with 4.7 T MRI in 1997. Subsequently, high-field MRI up to 7T have been used in evaluation oesophageal specimen and advanced MRI imaging techniques like diffusion tensor MRI and tractography have been used. In spite of these advancement in evaluation of ex vivo specimen, the advancement and utilization of MRI for clinical purposes has been gradual and sparse. In 2005, Riddell et al. used surface coil MRI in staging of oesophageal carcinoma. Sakurada et al. showed promising results in staging of T3 and T4 Ca oesophagus patients using T2WI and DWI. Over the recent years, the advancements in in vivo MRI of oesophageal Ca have been focused on identification of lymph nodal involvement (Alper et al.), staging postconcurrent radio-chemotherapy (Wang et al.), postoperative nodal recurrence (Shuto et al.) and gross tumour volume delineation (Hou et al.). Pavone et al. (1992) used Gadopentetate dimeglumine – barium paste in distending and opacifying and assessment of concentric/eccentric tumour growth in patients of Ca oesophagus and identifying a case of oesophageal leiomyoma in their study. In 1996, Ogawa et al. used ferric ammonium citrate-cellulose paste for oesophageal lumen opacification with excellent luminal opacification in 84% of the sagittal images and 79% of the axial images. Subsequently, Zhang et al. (2012) utilized high T2* signal of water to provide good contrast in MR oesophagogram. For easy intake of water, the head of patients was padded slightly high and water swallowing was done through a pipe/straw and patients were instructed to swallow the water continuously during the acquisition. Without respiratory triggering, sagittal and axial True fast imaging with steady state procession (True FISP) T2*weighted images were obtained. The diagnostic quality of water swallow was graded excellent in 97.7% of the sagittal images and in 81.8% of the axial images the cases. Even after the advances elucidated above, the present utility of MRI in diagnosis of oesophageal disease is more as an occasional adjunct for resolving specific clinical dilemmas rather than as a primary investigation. However, the future of the modality is bright and promising with continuous improvement in acquisition technology and the hardware. EUS is a specialized endoscope with an ultrasonographic probe to evaluate hollow visceral organs. It is useful to visualize oesophagus and stomach lining. Adjacent organs can also be visualized like liver, pancreas, GB, para-aortic region and mediastinum. EUS is done under mild sedation. EUS is available in two types of probes, that is Linear and Radial Probe. Gastrointestinal tract is visible in a healthy patient as five layers. These are first layer – superficial mucosa, second – mucosal, third – submucosal, fourth – muscularis propria and fifth – oesophageal adventitia. EUS is helpful mainly in characterizing the lesion as benign or malignant. On a standard endoscopy study, these lesions are usually seen as a bulge within wall. EUS helps in determining the location of lesion, whether it is within wall or outside the wall. Also it helps in determining the extent of involvement of lesion and its relationship with surrounding structures. It also has a role in looking for secondary causes of motility disorder like achalasia. However, its diagnostic as well as therapeutic role in oesophageal cancer is noteworthy. Endoscopic ultrasound plays a vital role in local staging of oesophageal cancer. Staging of oesophageal cancer is essential to choose appropriate therapy. Surgery is the gold standard for staging of oesophageal cancer. Usually in clinical practice, most patients undergo preoperative therapy as chemoradiation which is followed by surgery. Thus, for pretreatment staging, EUS findings are complementary with radiological imaging like MRI, CT scan and PET scan. Standard protocol is to perform EUS in most cases of oesophageal cancer. EUS is useful in early stages of oesophageal cancer, that is T1a and T1b. It is helpful in evaluating lymph node enlargement. Whenever a patient is assessed for feasibility of early cancer for mucosal resection via endoscopic procedure, it is important to rule out deeper tissue invasion and lymph node metastasis. EUS also plays essential role in lymph node FNA and biopsy in doubtful lesions. Role of EUS in advanced oesophageal cancer is limited. Usually EUS cannot be used to differentiate between T3 and T4. Distant metastasis is usually well diagnosed on cross-sectional imaging. EUS can only detect invasion into aorta as T4 which is better visualized in CT scan. EUS has limitations like it cannot look into metastases where CT scan and PET scan are useful. Role of EUS in postradiation or chemotherapy is under evaluation. EUS is technically difficult and has a significant learning curve. ALGORITHMIC APPROACH TO A PATIENT WITH OESOPHAGEAL DYSPHAGIA Gurdarshdeep Singh Madan, Ankita Dhawan Swallowing is essential not only for maintaining the nutrition for life but also a major determinant in one of the pleasures of life. The voluntary component of swallowing is the initial 10% and is initiated by collecting the food bolus onto the tongue and voluntarily propelling the bolus into the oropharynx. This initiates a wave of involuntary contraction and relaxation of pharyngeal muscles which pushes the food through cricopharynx into the oesophagus. Closure of glottis and cessation of respiration are reflex activities. Thereafter, the ‘peristaltic’ ring of oesophageal muscle contraction pushes the food towards the stomach at a rate of approximately 3.0–4.5 cm/s. The semisolid and liquid food falls by gravity, ahead of the peristaltic wave when the individual is upright. In cases where there is incomplete clearing of the oesophagus, secondary peristaltic waves are generated which are weaker than the primary peristaltic wave and aid in clearing the oesophagus. Unlike the rest of oesophagus, lower oesophageal sphincter (LES) is contracted at rest and relaxes on swallowing, which prevents reflux of gastric contents into the stomach while permitting the food bolus into the stomach (Fig. 8.1.7). Difficulty in swallowing is a common problem affecting approximately 3%–15% of general population. This prevalence increases in elderly population and in patients with other comorbidities. Understanding the symptomatology and pathophysiology of dysphagia is essential for the radiologist to plan the correct investigation and to remain clinically relevant in management of the patient. The symptoms of difficulty on swallowing has been further classified into various terminology, each highlighting a specific association and hence clinically leading to a smaller set of differential diagnosis (Table 8.1.4). Depending on location, dysphagia may be localized to oral, oro-pharyngeal or oesophageal regions. Dysphagia can also be subclassified as Structural dysphagia – caused by mismatch between the size of food bolus and the lumen of the aerodigestive track/oesophagus that is a large food bolus or the narrow lumen. It is classically described as difficulty in swallowing solid food. Motor dysphagia – it is caused by abnormalities of peristalsis/impaired upper oesophageal sphincter relaxation/closure and impaired lower oesophageal sphincter relaxation. It is difficulty in swallowing both solid and liquid foods. There are certain limitations to such generalization of symptoms. For example, Scleroderma, which is primarily an oesophageal motility disorder, presents initially with mild dysphagia on swallowing solid bolus. Also, in patients with oropharyngeal structural pathology, patient may be more symptomatic and concerned due to inability to handle fluids and causing aspiration. In other subset of patients, more than one mechanism may be responsible causing both improper propulsion of food and associated secondary luminal narrowing by stricture formation. Odynophagia generally arises due to passage of bolus along inflamed or ulcerated mucosa causing irritation, which may be due to foreign body ingestion, infective, chemical or inflammatory oesophagitis. Associated symptoms like cranial nerve abnormalities, hoarseness and other general and systemic symptoms and signs may point towards the location and cause of dysphagia. The patient may be able to localize the location of dysphagia. However, the sensation of dysphagia in oesophagus (especially in distal two-thirds) may be proximally referred in a third of the cases. Inability to produce sufficient saliva (Xerostomia) may also be perceived as dysphagia, especially common in elderly (see Fig. 8.1.8). Adult oesophagus measures approximately 17–26 cm in length and extend from cricopharynx to the lower oesophageal sphincter. Oesophagus can be anatomically divided into cervical oesophagus (cricopharynx to suprasternal notch) and thoracic oesophagus. The structural causes of the dysphagia manifest when the lumen of the oesophagus has been reduced to one-third (~13 mm in size). Dysphagia in a setting of Gastro-oesophageal Reflux Disease is very common and is multifactorial in pathophysiology – increased mucosal sensitivity, impaired distensibility and motor function, formation of strictures. Propulsive/motor disorders can be due to abnormality of peristalsis (both excessive and reduced) or relaxation of lower oesophageal sphincter (see Figs. 8.1.9–8.1.13). Few common causes of oesophageal dysphagia are enumerated below: Presentation and causes of dysphagia in neonates and infants tend to be different from adults. They may present as aspirations, incessant crying in addition to choking and inability to feed. In neonates, the causes may be neuromuscular or central in origin affecting swallowing or could be congenital in nature affecting oral or pharyngoesophageal phase of swallowing (Fig. 8.1.14). In older children and toddlers, ingestion of foreign body is one of the most common cause of dysphagia/choking. In these cases endoscopy is therapeutic. Ingestion of caustic agents can cause severe caustic oesophagitis which can be potentially fatal. In children, high index of suspicion should be present and relevant history for ingestion of caustic agents should be elicited. Other causes of dysphagia (Fig. 8.1.15) in paediatric age group are covered in detail in relevant chapters. Which investigation to choose: The armamentarium available for a clinician while evaluating dysphagia are mentioned below: Before the advent of flexible upper gastrointestinal endoscopic techniques, contrast (barium) upper gastrointestinal radiological studies especially barium swallow were the cornerstone in evaluating dysphagia. However, endoscopic examination of the oesophagus is presently the first line investigation of choice for oesophageal dysphagia. Endoscopy offers the advantage of mucosal assessment and characterization, ability to identify Barrett’s metaplasia and offer diagnostic and therapeutic interventions in a single sitting. Endoscopy is relatively deficient in diagnosing motility disorders and identifying benign conditions which cause dysphagia but do not cause mucosal abnormalities. Barium swallow should be done as a first line investigation of choice where dysmotility or oesophageal ring/webs/subtle strictures are suspected. A well-planned barium study with barium-coated appropriate bolus such as bread/marshmallow/barium pill may demonstrate oesophageal ring causing hold up of contrast. Barium swallow is also the first line investigation of choice for Achalasia. It may also be used for characterization of hiatal hernia. Oesophageal manometry/HRIM (high resolution impedance manometry) is specifically done in patients in whom endoscopy and barium studies are noncontributory. They show high pressure at upper and lower oesophageal sphincter with mid oesophagus showing waves of peristalsis. It is the gold standard to diagnose achalasia with certainty. It is also of immense value in diffuse oesophageal spasm and nutcracker oesophagus. Endoscopic ultrasound combines the advantage of an endoscope with high-frequency ultrasound, providing improved spatial resolution, which is useful for staging of oesophageal carcinoma and allows taking directed biopsies for histopathology. It can also be used to assess Barrett’s metaplasia and evaluating other submucosal tumours/paraoesophageal lymph nodes. PET-CT scan is a preferred modality of oesophageal and oesophago-gastric junction Ca and includes PET CT from skull base to mid-thigh in absence of M1 disease. Early disease may not be detectable on PET-CT as depth of invasion cannot be accurately assessed. Assessment of locoregional lymph node is limited as uptake from oesophageal lesion cannot be differentiated from uptake from periesophageal lymph nodes due to limited spatial resolution of PET. Nonregional or distant lymph node involvement can be well identified and assist in accurately upstaging and planning treatment of Ca oesophagus. CT scan has limited value in identifying early oesophageal Ca and identifying nonregional lymph nodes. It can be used in assessing recurrence and to check for integrity postoesophagectomy. Ambulatory reflux monitoring is useful in diagnosing GERD without oesophagitis. Endoscopy is normal in these patients with hypersensitive oesophageal mucosa – causing pain. The electrode tip is placed in distal oesophagus and pH is measured over 24/48 hours. The outcome is expressed as percentage duration when the distal oesophageal pH was less than 4. This can be combined with impedance monitoring to increase sensitivity. They also serve as a guide to evaluate poor response to therapy for GERD. A reading of more than 6% is considered abnormal. Additional newer advances like use of Functional Lumen Imaging Probe (FLIP) has been evaluated to assess oesophagogastric junction distensibility and distension-mediated peristalsis in oesophageal motility disorders. The investigations are often guided by clinician’s preferences, availability of the investigation and likelihood of a probable diagnosis on history taking and clinical examination. OESOPHAGEAL MOTILITY DISORDERS Ankita Dhawan Oesophagus is around 25–40 cm muscular tube in posterior mediastinum. It comprises both striated and smooth muscles. It extends from the level of upper oesophageal sphincter (UES – at the level of cricopharyngeal muscle) and lower down till the level of lower oesophageal sphincter (LES – present at the junction of lower end of oesophagus and cardia of stomach). Normal function of oesophagus is to transport both solids and liquids to stomach, which is mainly attributed to oesophageal peristalsis and gravitational force. Therefore, radiographic evaluation of oesophageal peristalsis is done in recumbent position to remove the contribution from gravity.
8.1: Oesophagus
Parts of Oesophagus
Approximate Length (cm)
Arterial Supply
Venous Drainage
Lymphatic Drainage
Nervous System
Cervical and upper thoracic
8
Inferior thyroid artery
Inferior thyroid vein
Lower deep cervical lymph nodes
Branches of recurrent laryngeal nerve
Mid thoracic
8
Aortic branches
Azygous system
Mediastinal nodes
Oesophageal (myenteric) plexus.
Lower thoracic and abdominal
8
Left gastric artery
Left gastric vein
Left gastric nodes
Para-sympathetic fibres from vagus; sympathetic fibres from T1 to T4/6 spinal segments
Histology of oesophagus
Rings of oesophagus
Embryology of oesophagus

Introduction
Which investigation to choose?
ACR Appropriateness Criteria
Category/Presentation(Initial Imaging)
Usually Appropriate
May Be Appropriate
Not Appropriate
Retrosternal dysphagia in immunocompetent patient
Retrosternal dysphagia in immunocompromised patient
Early postoperative dysphagia (oropharyngeal/retrosternal)
Delayed postoperative dysphagia (>1 month) (oropharyngeal/retrosternal)
Imaging techniques


CT and CT Oesophagogram
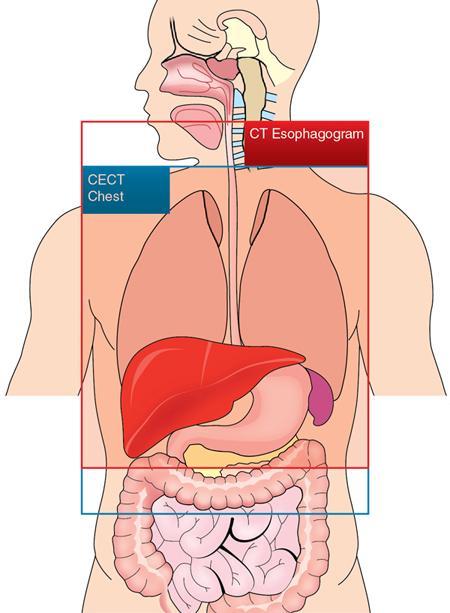
CECT Chest
CT Oesophagogram
Position
Supine
Supine
Respiration
Breath hold (inspiration)
Breath-hold preferable
FOV
Patient thorax to include ribs and chest wall
Same as CECT. However, narrow FOV could be done
Effervescent agents
Not required
May be used prior to oral contrast
Oral contrast
Not required
3% of water-soluble contrast in water or in flavoured food-thickening agent made to nectar/honey thick consistency
IV Contrast
Required
Not required
Role of endoscopic ultrasonography in oesophageal lesions (Fig. 8.1.6)
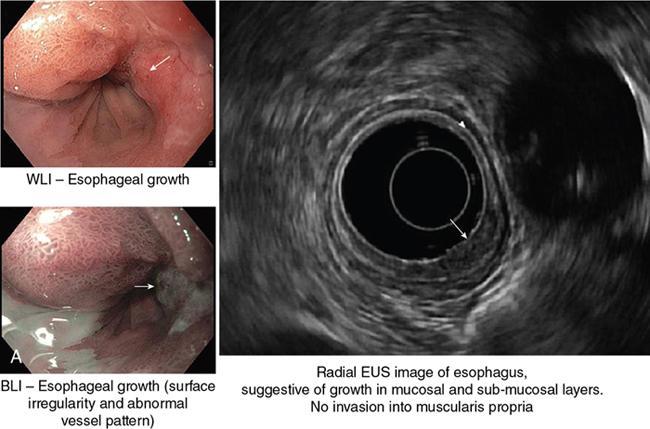
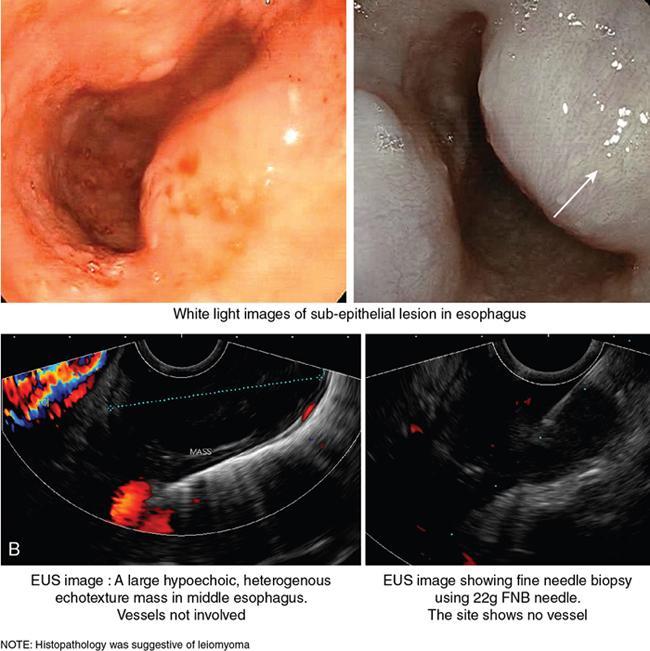
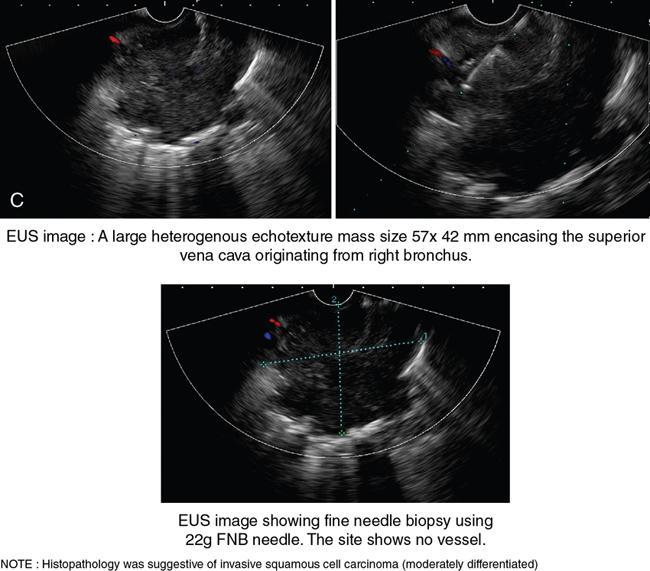
EUS in oesophageal cancer
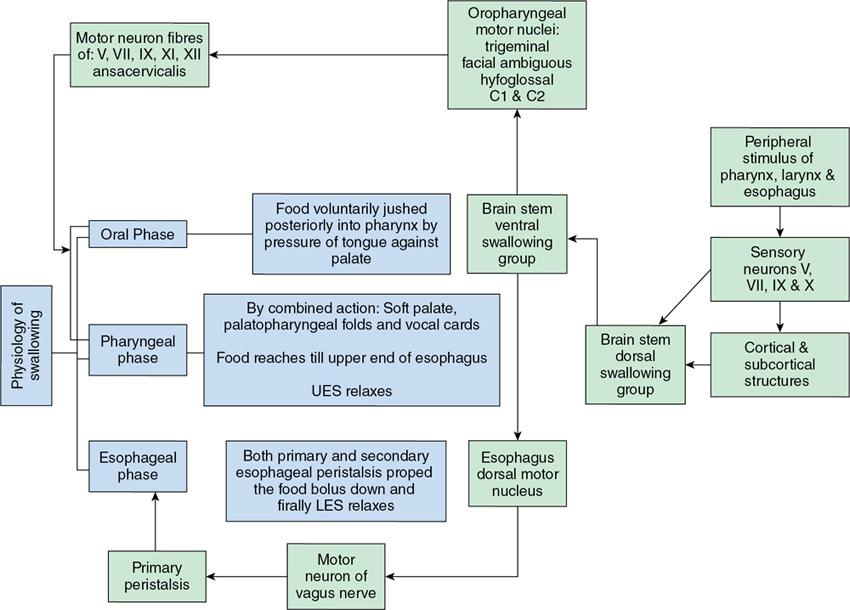
Dysphagia
Symptom of difficulty on act of swallowing and is frequently associated with sensation of hold up of bolus of food. It may or may not be associated with pain.
Odynophagia
Painful swallowing. It is generally associated with dysphagia. However, the reverse is not always true.
Aphagia
Complete obstruction to the pharynx or oesophagus.
Phagophobia
Morbid fear of swallowing – may be psychogenic or secondary to anxiety about aspiration or food impaction in organic oropharyngeal/oesophageal abnormalities including neuromuscular/motility disorders.
Globus pharyngeus
It is a sensation of foreign body stuck in neck which at times is relieved temporarily by act of swallowing. It is commonly associated with pharyngeal abnormalities.
Transfer dysphagia
It is a disorder pertaining to transfer of food bolus from oral/oropharyngeal phase to oesophageal phase of swallowing. It may or may not result in nasal or tracheobronchial aspiration. Commonly associated with neuromuscular/motility disorders.
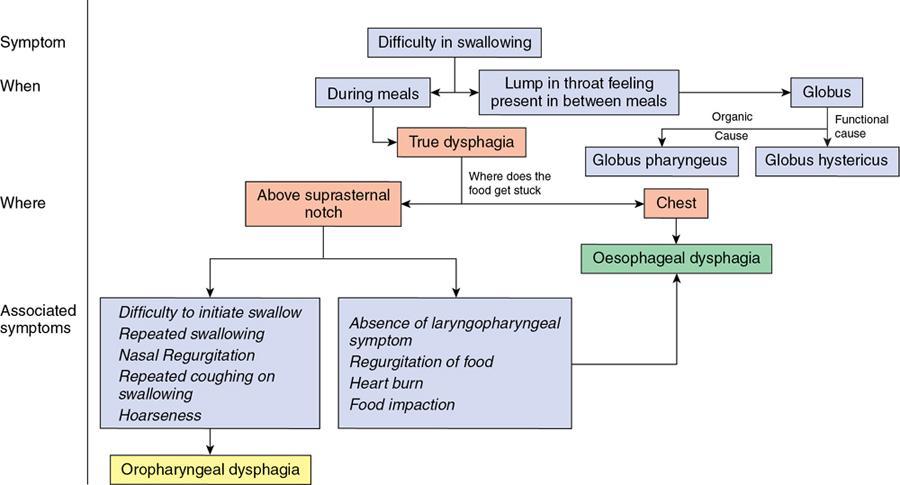
Oesophageal dysphagia
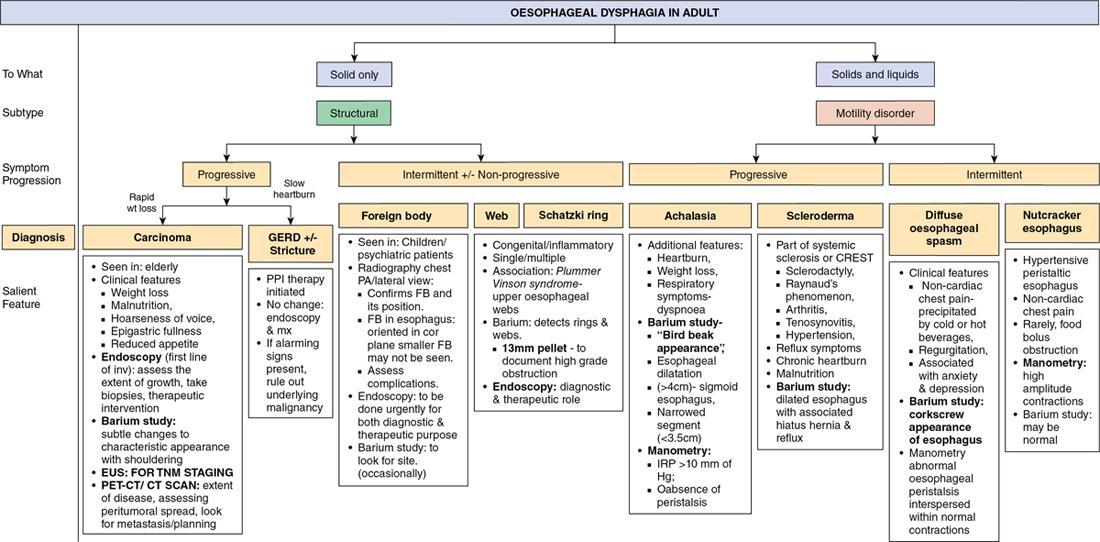
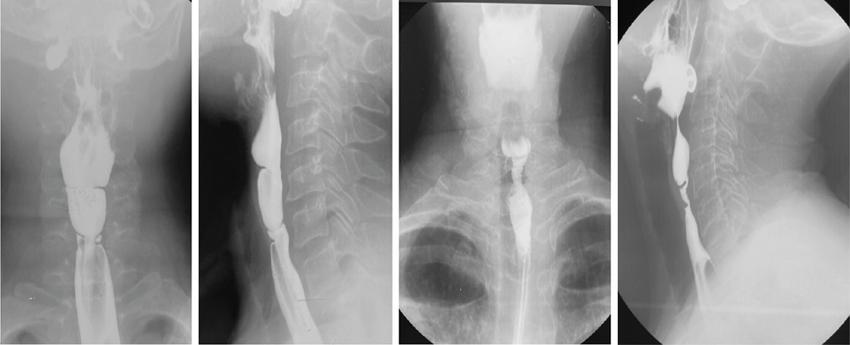
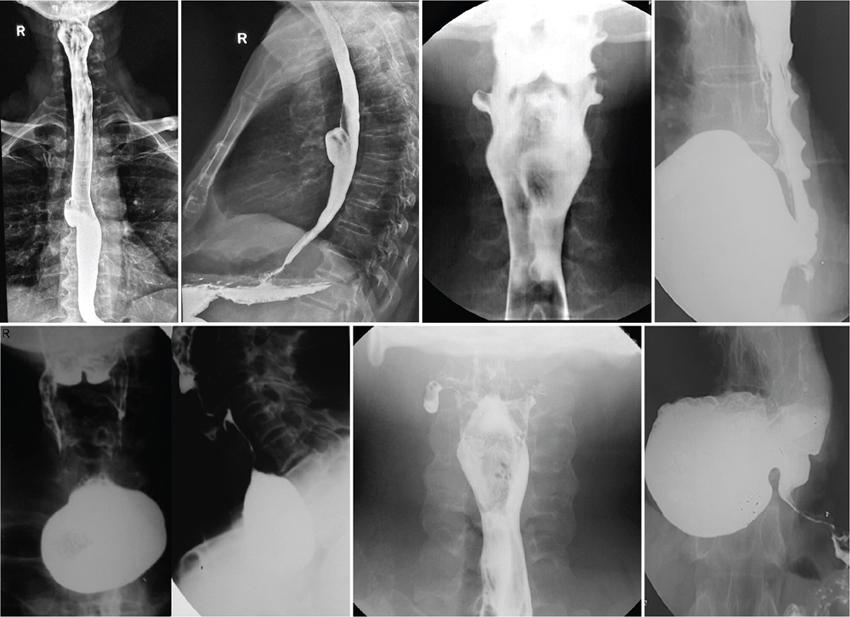
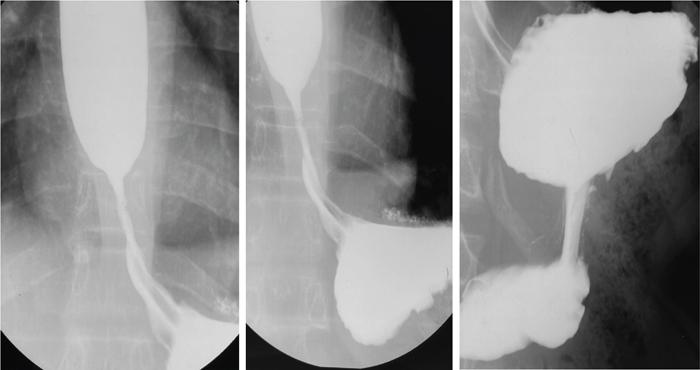
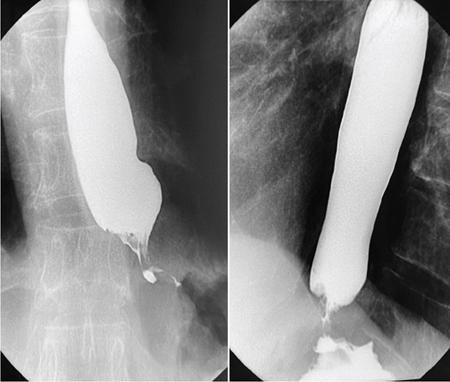
Motor Dysphagia
Structural Dysphagia
Odynophagia
GERD with weak peristalsis
Schatzki ring
Infective oesophagitis
Achalasia
Peptic strictures
Pill oesophagitis
DES/Corkscrew oesophagus
Neoplasia
Caustic injury
Connective tissue disorders
Hiatal hernia
Inflammatory bowel disease
Special considerations
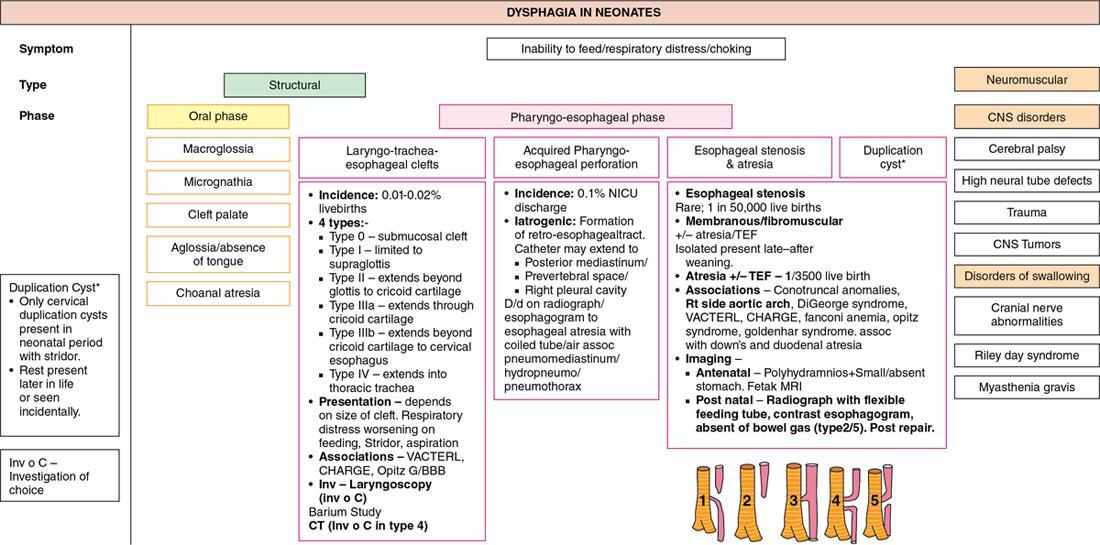

Endoscopy +/− Biopsy
Barium swallow (13 mm tablet/marshmallow/bread soaked in barium) with video fluoroscopy
Oesophageal manometry (high resolution impedance manometry)
High-frequency intraluminal endoscopic ultrasound (EUS)
Routine chest radiograph
PET CT
CT scan with contrast
Ambulatory reflux monitoring
Bernstein testing (obsolete)
Advantages
Disadvantages
Assessment of mucosal abnormalities
Cannot assess motility disorders
Offer diagnostic (biopsy) and therapeutic interventions (endoscopic ablation/resection)
Difficult to evaluate beyond stenotic segment
Can be combined with endoscopic ultrasound
Lesions with no mucosal abnormalities may be missed
Poor patient comfort
Advantages
Disadvantages
Inexpensive, easily available
Appropriateness limited on use of spot images and by proficiency/experience of technician/doctor
Can be tailored depending on patient presentation
Negative barium study does not rule out need of endoscopy
Video-fluoroscopy allows adequate/physiological assessment of oesophageal motility
No cross-sectional information obtained
Barium is not absorbed and hence allergic reactions are rare
Barium can elicit severe mediastinitis when used in oesophageal perforations
Uses
Done primarily in case of dysphagia after noncontributory endoscopy; in evaluation of GERD and noncardiac chest pain.
Limitations
May miss transient relaxations of LES in GERD; and transient contractions of oesophagus in nutcracker oesophagus/DES.
Procedure may be uncomfortable for some patient.
Advantages
Good spatial resolution and identification of oesophageal wall layers and assess depth of invasion.
Evaluation of loco-regional lymph nodes.
Investigation of choice for follow up in postendoscopic ablation/resection patients.
Limitations
Operator dependent.
Use of high-resolution probe limits evaluation of deeper structures.
Distant lymph node metastasis needs to be evaluated separately.
Advantages
Disadvantages
Provides metabolic imaging. Preferred investigation in patients of oesophageal Ca in absence of M1 disease.
Assessment of local lymph nodes is limited by spatial resolution of the PET component.
PET combined with CT scan provides good anatomic images for planning.
Availability and cost of PET-CT might be a limitation.
High sensitivity in identifying distal solid organ/lymph node metastasis.
Advantages
Disadvantages
Easy availability
Low sensitivity in picking up small metastatic lymph nodes
Preferred investigation in posttreatment follow up of oesophageal Ca patients
No metabolic information
Used for assessment of immediate postop complications after oesophagectomy
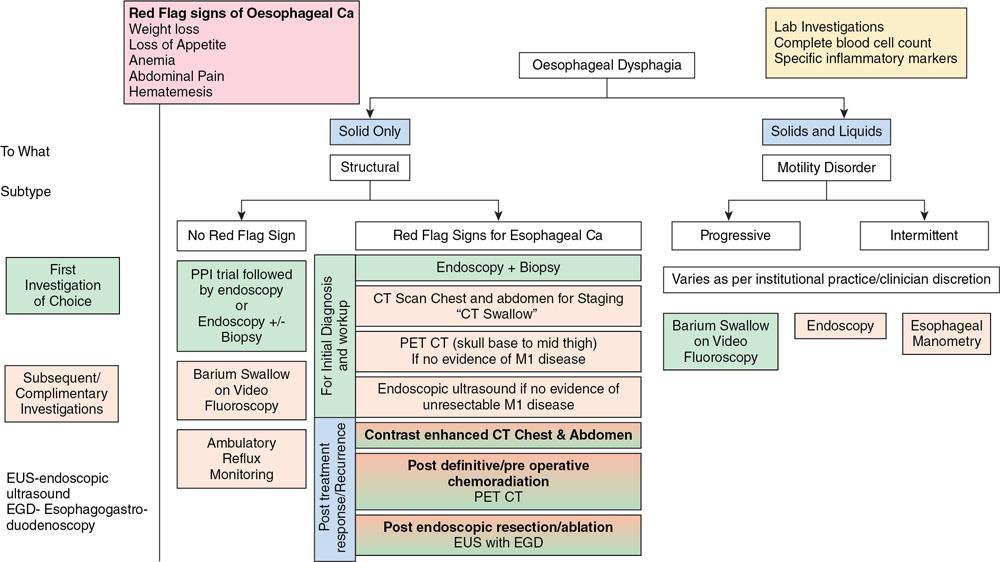
Introduction
Normal oesophageal physiology
Resting phase
Types of oesophageal peristalsis
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








