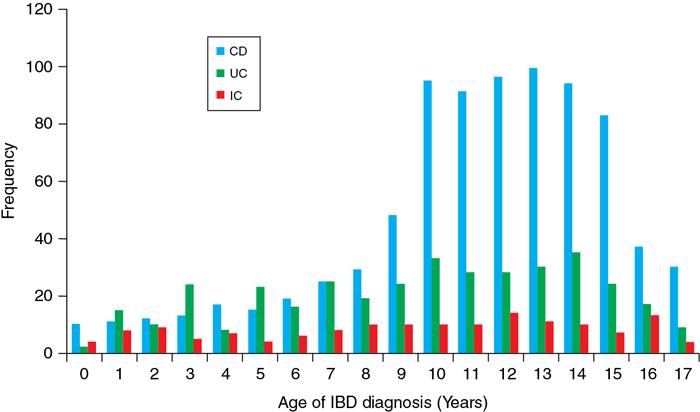
Kavita Saggar, Archana Ahluwalia, Devinder Pal Singh Dhanota, Geetika Khanna, Chander Gupta Inflammatory bowel disease (IBD) represents chronic idiopathic inflammation of the gastrointestinal [GI] tract. The common subtypes are Crohn’s disease, ulcerative colitis (UC) and indeterminate colitis (Fig. 7.11.1). Approximately 20%–25% of patients present in childhood or adolescence. Of these, nearly two-thirds of cases are caused by Crohn’s disease with an incidence of 4.56 per 1000,000 children (Fig. 7.11.2). IBD can affect any portion of the GI tract; however, children have a much higher incidence of small bowel and colonic involvement (Table 7.11.1). Currently, there is no single diagnostic test for the evaluation of IBD. Diagnosis is based on a combination of history, physical examination, serum inflammatory markers, endoscopy, histopathology and imaging. A host of imaging modalities is available for the evaluation of IBD in children. Traditionally, barium fluoroscopic studies, namely small bowel follow-through or small bowel enteroclysis (SBE) were used. However, barium studies depict only the mucosal abnormalities and do not evaluate the extraluminal extent of disease. Ultrasonography (US) is increasingly being used in the workup of patients with IBD. Small intestine contrast ultrasound (SICUS) and contrast-enhanced ultrasound (CEUS) are used for evaluation of IBD. CEUS requires intravenous administration of microbubble contrast such as Sonovue, and SICUS requires small bowel adequate luminal distension with ingestion of an iso-osmolar contrast agent such as polyethylene glycol. A recent meta-analysis of 33 studies showed that CEUS had better accuracy than US, CT and MR for the detection of inflammation and differentiation of fibrotic and inflammatory strictures. However, the extent of the disease was better evaluated by CT and MR. Currently, computed tomography enterography (CTE) and magnetic resonance enterography (MRE) have emerged as the most effective tools for imaging small bowel in patients with IBD. CTE and MRE can accurately depict the spectrum of imaging findings related to severity, extent and complications of IBD and thereby help in guiding management and assess the response to treatment. MRE is an attractive imaging modality due to lack of ionizing radiation, inherent multiplanar capability and excellent soft tissue contrast resolution. In addition, dynamic/cine imaging allows assessment of peristalsis and accurate identification of the diseased segment. The major disadvantage of MRE is high cost, lower spatial and temporal resolution and long examination time. Crohn’s disease is a transmural granulomatous disease that can affect any part of the GI tract from the oral cavity to anus, often involving multiple discontinuous segments of bowel. Patients have a genetic predilection to develop an abnormal immunologic response to environmental factors including food and gut flora, leading to a chronic inflammatory response. Small bowel is the major site of involvement. Terminal ileum is the most common location which is involved in approximately one-third patients. The colon is affected in 50% patients. In 15%–20% cases, there may be isolated involvement of the colon without affecting the small bowel. The clinical importance of small bowel Crohn’s disease is the impact that a diffuse small bowel disease is expected to have on child’s growth and development. Thus, objective evaluation of small bowel is essential in differentiating from other enteropathies and in directing the management of patients with IBD. Two methods used for imaging small bowel with barium using fluoroscopy are standard small bowel follow through (SBFT) and SBE. Small bowel evaluation with barium has long been considered the most common, noninvasive, inexpensive and easily accessible radiological method, but it has been replaced by cross-sectional imaging. In the current scenario, the only indication of barium studies is when CTE or MRE cannot be done because of nonavailability or are not feasible. Ultrasound is being increasingly used to assess the activity of Crohn’s disease, especially in paediatric patients. Greyscale ultrasound allows morphologic assessment of bowel wall and mesentery. Normal small bowel loops are easily compressible with the transducer and the wall thickness is <2 mm. The bowel loops show regular peristalsis and are mobile. Colour Doppler shows minimal intramural or perienteric vascularity. The salient sonographic features of Crohn’s disease are: There is marked concentric or eccentric bowel wall thickening [>2.5–3 mm]. The mural echogenicity depends upon the degree of inflammation and fibrosis. In the acute phase, mural stratification is maintained (Fig. 7.11.3A and B). In patients with long-standing disease, a target or pseudo-kidney appearance may be seen. In long-standing burnt-out disease, there is submucosal fat deposition. The actively inflamed bowel segments show increased vascularity on Doppler ultrasound (Fig. 7.11.4A and B). CEUS with microbubbles can objectively assess the disease activity. A stricture is seen as a segment of bowel wall thickening with apposition of the luminal surfaces. There is persistent luminal narrowing with variable degree of upstream dilatation and hyperperistalsis. Doppler imaging reveals hyperemia in strictures with an active inflammatory component. Fibrotic strictures do not demonstrate increased blood flow. A major advantage of ultrasound is real-time imaging which allows the evaluation of bowel peristalsis. The diseased segments of the bowel (inflamed or fibrotic) show reduced peristalsis and often appear fixed in position. Mesenteric inflammation is commonly seen in active Crohn’s disease. Sonography reveals thickening of mesentery with heterogeneous echogenicity due to oedema. Doppler ultrasound shows increased vascularity due to engorgement of vasa recta. Fibrofatty proliferation of mesentery seen in chronic disease appears as abnormally thickened echogenic fat that displaces the bowel loops. Reactive mesenteric lymph nodes (up to 1.5 cm) can be seen in the active inflammatory phase of Crohn’s disease. These appear as hypoechoic round or oval structures with preserved fatty hilum. Sinus tracts and fistula often arise from or just proximal to a strictured segment and appear as linear areas of altered hypoechogenicity extending from serosal surface of gut. There may be tethering and/or angulation of the affected bowel segment. Abscesses are discrete well-defined fluid collections with internal debris, septation or nondependent echogenic gas. Colour Doppler reveals peripheral hyperemia with absent blood flow centrally. Ultrasound may be falsely negative if an abscess contains a large amount of air and can be mistaken for an air-filled bowel loop. Both CTE and MRE identify the transmural, extramural and mesenteric manifestations of small bowel Crohn’s disease. The imaging findings can be categorized as: Asymmetric long segment (>3 cm) bowel wall thickening is a consistent feature of Crohn’s disease. The mesenteric border is usually more severely affected. It is imperative that the bowel should be distended when assessing bowel wall thickening. Thickening may be mild (3–4 mm), moderate (5–10 mm) or severe (>10 mm) (Fig. 7.11.5A and B). This is the most consistent finding in the active inflammatory stage, defined as increased signal intensity or attenuation on contrast-enhanced scans in a noncontracted segment of bowel in comparison with adjacent small bowel segments. Hyperenhancement may be stratified into bilaminar and trilaminar patterns. In the bilaminar pattern, there is hyperenhancement of only the inner wall (often referred to as mucosal hyperenhancement), and in the trilaminar pattern, there is inner and outer wall hyperenhancement, with the submucosal oedema giving a halo effect. Hyperenhancement may also be homogeneously transmural or patchy. A potential pitfall in contrast enhancement is that normal jejunal loops enhance more than ileal loops and collapsed bowel segments can show apparent hyperenhancement (Figs. 7.11.6A,B and 7.11.7A–C). These are defined as multifocal (>3) segmental areas of involvement with normal intervening gut loops. This is an important feature that differentiates Crohn’s disease from UC when colon is predominantly involved (Figs. 7.11.5A,B and 7.11.7A–C). This is identified as an increased signal of the thickened bowel wall on T2W images. The inflamed walls also show restricted diffusion. The hyperintense signal due to intramural oedema persists on T2W fat-saturated images differentiating it from intramural fat seen in long-standing burnt-out disease (Fig. 7.11.8A and B; refer Fig. 7.11.20A–D).
7.11: Paediatric inflammatory bowel disease
Learning objectives
Introduction
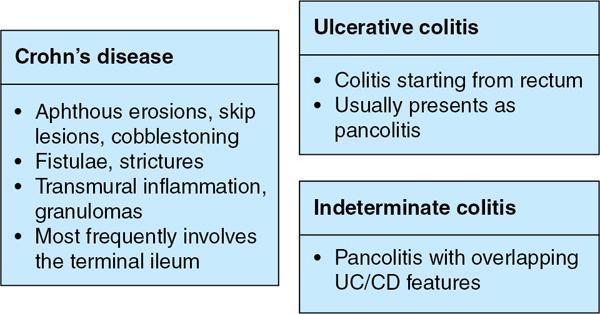
Crohn’s disease
Barium studies
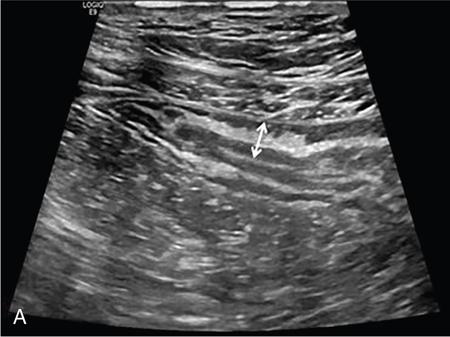
Ultrasound
Bowel wall thickening
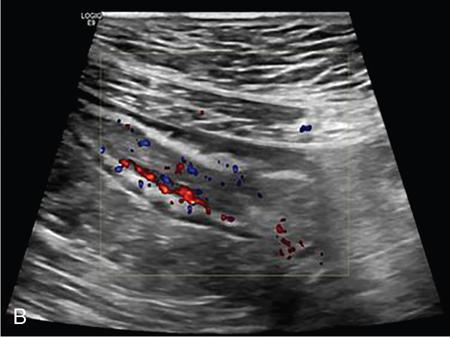
Hyperemia
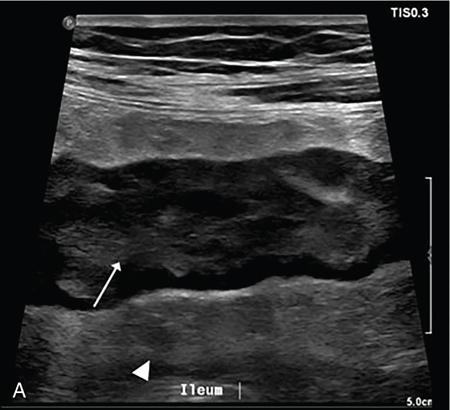
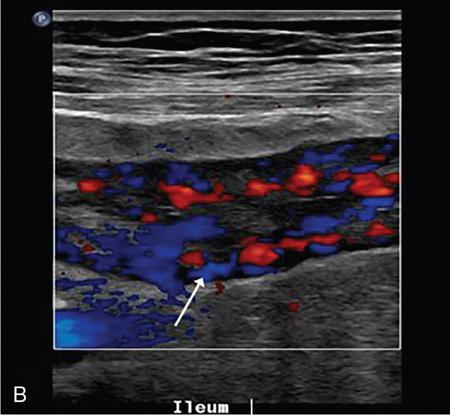
Stricture
Altered peristalsis
Mesenteric changes
Regional lymphadenopathy
Sinus tracts, fistula and abscess
Computed tomography and magnetic resonance enterography
Wall findings
Thickening
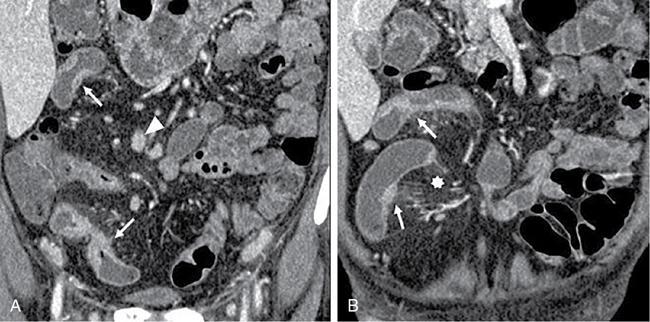
Segmental mural hyperenhancement
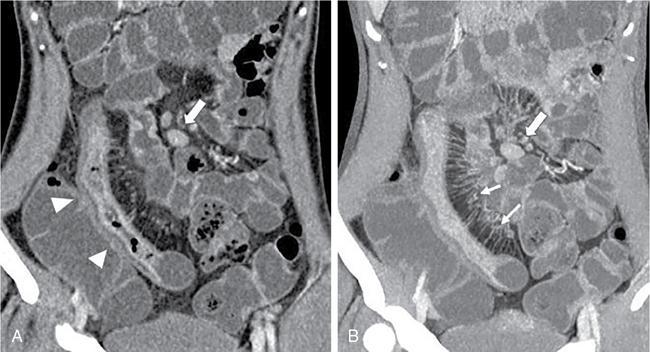
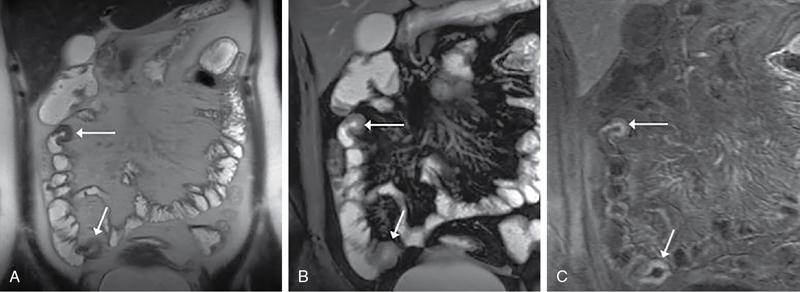
Skip lesions
Intramural oedema
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








