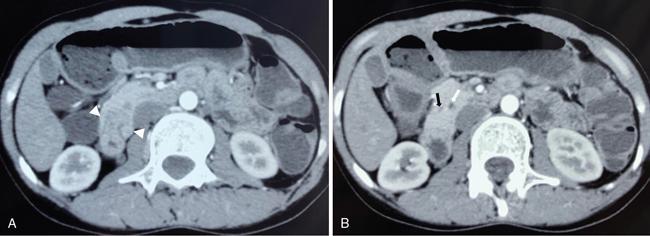
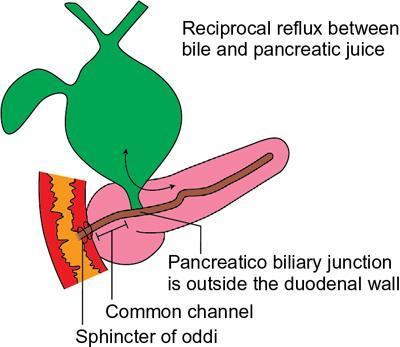
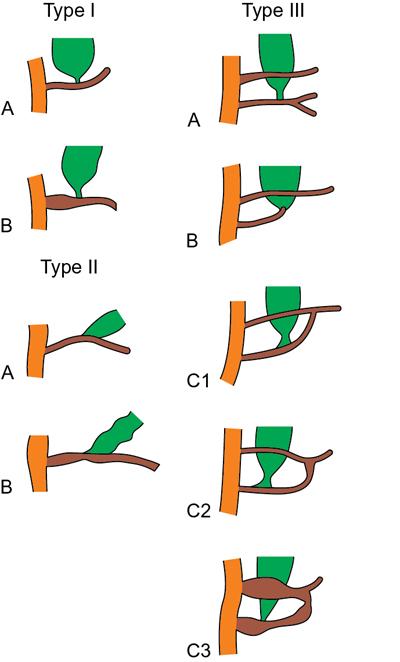
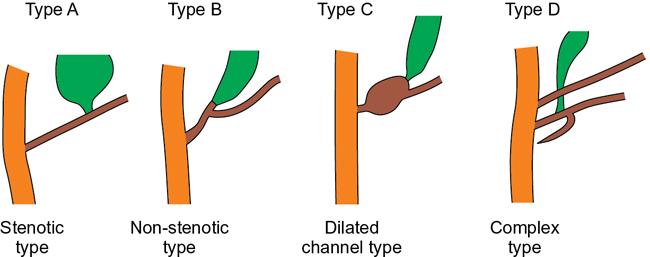
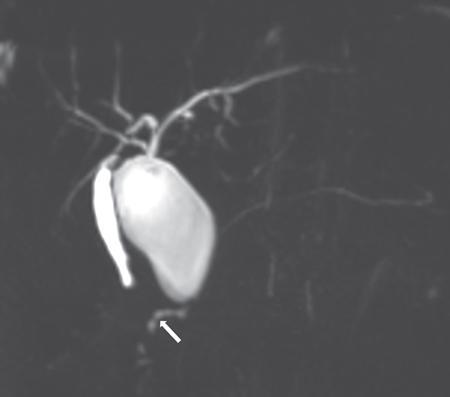
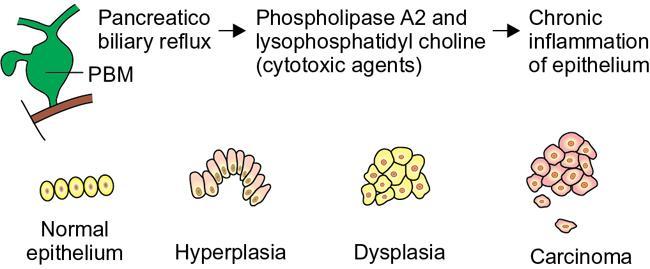
Priscilla Joshi, Mangal Subhash Mahajan, Vandana Jahanvi Imaging is important in paediatric patients with pancreatic pathologies which can be congenital, pancreatitis or neoplastic. Congenital pancreatic anomalies predispose the children to recurrent attacks of pancreatitis and other complications. Paediatric pancreatic tumours are uncommon but when they do occur imaging is essential for their diagnosis, staging and posttreatment follow-up. Pancreas divisum is the most common congenital pancreatic anomaly. It occurs in approximately 4%–14% of population. Failure of fusion of dorsal and ventral pancreatic ducts results in pancreatic divisum. Three variants of pancreatic divisum are known: In most cases, pancreatic divisum is asymptomatic. However, it may give rise to recurrent episodes of pancreatitis. Pancreatitis is believed to occur due to inadequate drainage of pancreatic secretions by the duct of Santorini. Pancreatic divisum is easily identified on MRCP (magnetic resonance cholangiopancreatography) which is a noninvasive technique and therefore is now increasingly being performed over ERCP to avoid ERCP-induce pancreatitis. Secretin-induced MRCP improves the visualization of the pancreatic ducts. The dorsal duct can be seen passing the CBD anteriorly and superiorly on imaging. This entity can also be identified on MDCT if the pancreatic duct is visualized. Rarely, it may be associated with cystic dilatation of terminal portion of duct of Santorini which is called Santorinocele. Failure of rotation of ventral bud along with the duodenum results in encasement of duodenum by a rim of pancreatic tissue and this entity is known as annular pancreas. The band of pancreatic tissue encircling the duodenum could either completely or partially surround the duodenum and is in continuity with pancreatic head. It occurs in approximately 1 in 20,000 population. Not all cases of annular pancreas present in childhood. In paediatric age group, it can present as neonatal duodenal obstruction because of associated duodenal stenosis. Children and adults with annular pancreas may also present with recurrent pancreatitis, postbulbar duodenal ulcerations or biliary obstruction. On upper gastrointestinal studies (barium meal), characteristic narrowing of second part of duodenum is seen. CT and MRI may show rim of pancreatic tissue encircling the duodenum. In partial annular pancreas (Fig. 9.16.2), pancreatic tissue is seen extending posterolateral or anterolateral to the duodenum with the pancreatic tissue giving a ‘crocodile – jaw’ configuration. This in the presence of gastric outlet obstruction helps in the diagnosis of incomplete annular pancreas. MRCP shows the pancreatic duct encircling the second part of the duodenum and entering it over its right lateral aspect. The pancreatic duct draining the annular segment usually drains into the main pancreatic duct. It can also drain into the intrapancreatic portion of common bile duct, the duct of Wirsung or the duct of Santorini. Secretin-induced MRCP because of its ability to demonstrate ductal anatomy well may become the best noninvasive imaging modality for diagnosis of pancreatic ductal variants including annular pancreas. Other causes of paediatric duodenal obstruction should be kept in mind while diagnosing annular pancreas. Other than annular pancreas, the presence of duodenal narrowing on barium study in neonates, should raise the possibilities of duodenal atresia, duodenal web and Ladd’s bands. In duodenal atresia, the contrast does not pass beyond the obstruction and no air is seen on radiograph beyond the atretic segment. Whereas, in annular pancreas, intestinal gas is seen distal to the involved segment. And on upper gastrointestinal studies, the duodenal bulb is distended with slow transit of oral contrast through the stenosed duodenal segment distally. In duodenal web classical ‘wind–sock’ diverticulum is seen due to distal stretching of the web forming an intraluminal pseudodiverticulum. On barium studies, thin radiolucent membrane is seen due to filling of barium within the lumen and around the diaphragm. The mutation in the developmental protein IPF1, results in pancreatic agenesis. It is very uncommon as it is incompatible with life. Anomalies associated with this condition are: foetal growth restriction, agenesis of gallbladder and polysplenia. Hypoplasia or partial agenesis occurs due to nondevelopment of dorsal or ventral pancreatic bud. Partial agenesis of the dorsal pancreas is more common than agenesis of the ventral portion. However, complete dorsal pancreatic agenesis is rare. Dorsal pancreatic agenesis is often associated with heterotaxia syndrome. Partial dorsal pancreatic agenesis is more common than partial ventral agenesis. Complete dorsal pancreatic agenesis is however rare. In partial dorsal agenesis, short rounded head of pancreas is seen adjacent to the duodenum with absence of pancreatic neck, body and tail; however, remnant of duct of Santorini and minor duodenal papilla are generally present. In complete dorsal agenesis, both the minor duodenal papilla and the duct of Santorini are absent. The distal pancreas contains the islet cells, hence those with dorsal agenesis are at a higher risk of developing Diabetes Mellitus. This uncommon condition is diagnosed when there is an accessory lobe of pancreatic parenchyma originating from the main gland and draining through an aberrant duct. The accessory lobe may be of varying size and may drain into the main pancreatic duct through a narrow or a wide communication. Association with gastric duplication cyst is known and if present the aberrant duct communicates with the gastric duplication cyst and the main pancreatic duct. Accessory pancreatic lobe may present as acute pancreatitis which is believed to result from obstruction of the pancreatic duct by viscus secretions, ulcer bleeding or biliary sludge. Ectopic pancreatic tissue can be seen in 0.6%–13.7% of population. It is most commonly seen in the stomach or duodenum. The ectopic tissue is generally located in the submucosa. Although, usually asymptomatic, it presents as stenosis, ulceration, gastrointestinal haemorrhage or intussusception. Pancreaticobiliary maljunction is a congenital abnormality in which the main pancreatic and common bile ducts join outside the duodenal wall that forms a common channel. It was first described by Arnolds in 1906. Women are more frequently affected than men with a female to male ratio of 3:1. It is predominantly present in young women. Its incidence is 0.9%–6.2% on ERCP and 61.8%–70% on autopsy. Although few familial cases have been reported whether pancreaticobiliary maljunction is hereditary remains unclear. It is diagnosed on the basis of radiological findings or anatomical findings at surgery or autopsy. Hence, radiologist play crucial role in making diagnosis of this entity. Pathogenesis of pancreaticobiliary maljunction is controversial. Several studies have proposed that anomalous development of the ventral pancreas with abnormal fusion between the bile duct and branches of the ventral pancreatic duct is responsible for development of pancreaticobiliary maljunction. Normally, the main pancreatic duct and common bile duct open either separately or join to form a common channel that opens in second part of duodenum. Length of the common channel varies between 1 and 12 mm normally. Sphincter of Oddi is present at the distal end of pancreatic and common bile ducts. It regulates outflow of pancreatic and bile juice. The sphincteric control at the pancreaticobiliary junction is important for the regulated drainage of bile and pancreatic juice. In maljunction, as the union of the bile duct and the pancreatic duct is outside the duodenal wall, the sphincter of Oddi is unable to regulate the flow of pancreaticobiliary juices resulting in bidirectional regurgitation of juices (Graphic 9.16.1). It produces various pathological conditions in the biliary tree and pancreas. On the basis of dilatation of common bile duct, pancreaticobiliary maljunction is divided into that with biliary dilation and without biliary dilatation. The former is more common and present in 77% of cases. The latter is less common and present in remaining 23% of cases. New Komi classification (Graphic 9.16.2) is based on how the pancreatic and common bile ducts join. It takes into account presence or absence of the common channel dilatation and the concept of pancreatic divisum. Type I union is bile duct type, in which the common bile duct joins the pancreatic duct at right angle. It has single papilla. It is subclassified into type IA and IB based on absence or presence of the common channel dilatation respectively. Type II union is pancreatic duct type, in which the pancreatic duct joins the common bile duct at an acute angle. It also has single papilla. It is subclassified into type IIA and IIB based on absence or presence of the common channel dilatation, respectively. Type III is complex type, in which the two ducts join in complex configuration. It has two papillae. It is subclassified into type IIIA, IIIB and IIIC. Type IIIA is similar to pancreatic divisum with biliary dilatation. Type IIIB is characterized by absence of duct of Wirsung. Type IIIC is further subclassified into IIIC1, IIIC2 and IIIC3. A thin communicating duct between the main and accessary ducts is present in type IIIC1. A common channel is made up of common and accessary ducts of equal calibre in type IIIC2. There is complex network of enlarged ducts that join each other by total or partial dilatation of the ductal system in type IIIC3. Japanese Study Group on Pancreaticobiliary Maljunction proposed a new classification (Graphic 9.16.3) in 2015 on the basis of formation of pancreaticobiliary maljunction. According to this new classification, pancreaticobiliary maljunction is divided into four types: (1) Stenotic type: The common channel is joined by the distal CBD with stenosis, (2) Nonstenotic type: The common channel is joined by the distal CBD without stenosis, (3) Dilated common channel type: As the name suggests, the common channel is dilated and (4) Complex type: A complicated pattern is formed by the pancreaticobiliary junction. Pancreaticobiliary maljunction with congenital biliary dilatation have higher incidence of symptoms in neonatal and infantile period and manifest as jaundice and abdominal mass. Less common symptoms include abdominal pain, vomiting and fever. Dilatation of common bile duct is an important finding on ultrasonography and further imaging with magnetic cholangiopancreatography is recommended to look for the status of common channel. Pancreaticobiliary maljunction without congenital biliary dilatation is more difficult to diagnose because of less frequent symptoms and less remarkable imaging findings. These patients are frequently asymptomatic but may presents as abdominal pain and hyperamylasaemia in adulthood. Many adult patients may show signs of biliary cancers. Focal or diffuse gallbladder wall thickening may be an early clue for the diagnosis. Hence, pancreaticobiliary anatomy should be assessed by magnetic resonance cholangiopancreatography when no specific cause of gallbladder wall thickening is established. The diagnosis of pancreaticobiliary maljunction is made when there is an abnormally long common channel of the pancreatic duct and the CBD; or there is an abnormal union between the pancreatic and bile ducts seen on an investigation which directly images these structures. These include endoscopic retrograde cholangiopancreatography, percutaneous transhepatic cholangiography or intraoperative cholangiography, magnetic resonance cholangiopancreatography or three-dimensional drip infusion cholangiography computed tomography (CT). However, when the common channel is relatively short, it is crucial to confirm that the effect of the papillary sphincter does not extend to the junction with direct cholangiography. There is no clear definition of a long common channel. Few authors suggest 8 mm or longer while others suggest 15 mm or longer as a long common channel. It can also be diagnosed if the pancreaticobiliary junction is demonstrated outside the duodenal wall on endoscopic ultrasound or multiplanner reconstruction images of multidetector-row CT. Pancreaticobiliary junction must be present outside the duodenal wall or the union of pancreatic or bile duct must be abnormal at surgery. Elevated amylase levels in the bile within the bile duct and gallbladder obtained immediately after laparotomy and presence of extrahepatic biliary dilatation strongly suggest the existence of pancreaticobiliary maljunction. Rarely, the amylase levels are close to or below the normal serum value in these patients. When cystic, fusiform, or cylindrical dilation is present in the extrahepatic bile ducts, detail workup is necessary to determine whether pancreaticobiliary maljunction is present. To diagnose biliary dilatation, maximum calibre of the common bile duct is measured. Diagnosis of bile duct dilatation is age-dependent (Table 9.16.4). Ultrasound is used as screening tool and alone may not be enough to detect an anomalous pancreaticobiliary junction. Extrahepatic bile duct dilation or gallbladder wall thickening are the clues to make early diagnosis of pancreaticobiliary junction. Endoscopic ultrasound demonstrates the pancreatic and bile duct junction outside the duodenal wall and help to diagnose pancreaticobiliary maljunction. Magnetic resonance cholangiopancreatography is an accepted noninvasive imaging tool to demonstrate the pancreaticobiliary anatomy. It is preferred over ERCP. Source images and maximum intensity projection images of 3D MRCP are very useful to delineate the pancreaticobiliary anatomy. Its sensitivity is 75% in adult and 44%–65% in children. Fig. 9.16.3 shows a case of Pancreaticobiliary maljunction with Choledochal cyst. Secretin-stimulated dynamic MRCP and time-spatial labelling inversion pulse (Time-SLIP) can be used to identify the pancreaticobiliary reflux. Secretin stimulates the exocrine pancreas to secrete fluid. This increase of fluid content within the lumen of pancreatic duct improves visualization. In pancreaticobiliary maljunction, there is enlargement and retrograde increase in signal intensity of the common bile duct. Time-SLIP MRI allows direct visualization of pancreaticobiliary flow by placing the inversion pulse at the head and body of pancreas and suppressing the background. Gadoxetic acid-enhanced MRI is used to identify biliopancreatic reflux. Gadoxetic acid is a hepatobiliary specific contrast agent. It is taken up by hepatocytes and excreted into the bile. In pancreaticobiliary maljunction, there is retrograde increase in signal intensity of the pancreatic duct. Multiplanar reconstruction images of the contrast-enhanced high-resolution multidetector CT scan can demonstrate the communication of the pancreatic and bile ducts and help to diagnose pancreaticobiliary maljunction. Its sensitivity is 58%–100% in adults and 20% in children. Drip infusion CT cholangiography involves intravenous injection of biliary contrast agent. It demonstrates details of pancreaticobiliary anatomy; however, it sometimes fails to demonstrate the pancreatic duct preventing the diagnosis of pancreaticobiliary anatomy. Endoscopic retrograde cholangiopancreatography confirms lack of effect of the sphincter of Oddi on the pancreatic and bile duct junction. Its sensitivity is 75%. Main advantage of ERCP is that it allows bile and tissue sampling and therapeutic procedures. In pancreaticobiliary maljunction, the Oddi sphincter fails to regulate the pancreaticobiliary junction. Normally, hydrostatic pressure within the pancreatic duct is higher than that in the bile duct. Hence pancreatic juice often refluxes into the bile duct. There is increased pressure in the bile and pancreatic duct secondary to obstruction of the common channel or primary stricture of distal bile duct. Obstruction or stricture is caused by impaction from a protein plug. It is often a temporary process because these protein plugs are fragile and resolve spontaneously. It causes transient and intermittent symptoms like abdominal pain, vomiting, and jaundice. Protein plugs consist of lithostathine. It is a soluble protein secreted by pancreas. They are often depicted at ERCP but not at CT or MRCP. Incidence of acute pancreatitis in pancreaticobiliary maljunction is 30% in children and 9% in adults. Chronic pancreatitis is present in 3% of these patients. In majority, the changes are less severe but can be recurrent. Protein plugs are believed to be one cause of acute pancreatitis. Pancreatic calcifications are usually absent in chronic pancreatitis with pancreaticobiliary maljunction. Common bile duct and gallbladder calculi occur more often in adults than in children. Its incidence is 23% in adults and 9% in children. Calculi formation appears to be related to bile stasis because pigmented calculi are more commonly detected than cholesterol calculi in patients with pancreaticobiliary maljunction.
9.16: Paediatric pancreatic pathologies
Introduction
Pancreas divisum
Aetiopathogenesis
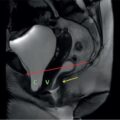
Imaging findings (Fig. 9.16.1)
Annular pancreas
Aetiopathogenesis
Demographics
Clinical presentation
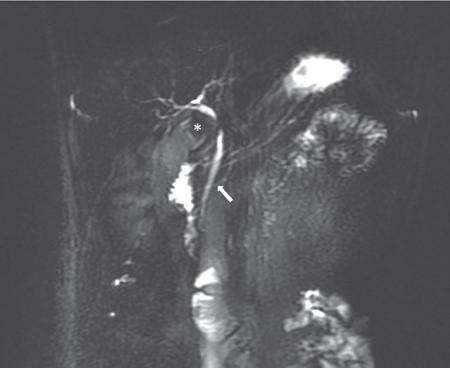
Imaging findings
Differential diagnosis
Agenesis of pancreas and pancreatic hypoplasia
Accessory lobe of pancreas
Ectopic pancreas (Table 9.16.1)
Serial No.
Sites
1.
Stomach
2.
Duodenum
3.
Jejunum
4.
Meckel’s diverticulum
5.
Colon
6.
Oesophagus
7.
Gallbladder
8.
Bile ducts
9.
Liver
10.
Spleen
11.
Umbilicus
12.
Mesentery
13.
Mesocolon
14.
Omentum
Pancreaticobiliary maljunction
Introduction
Aetiopathogenesis
Types (Table 9.16.2)
Diagnosis
Diagnostic criteria (Table 9.16.3)
Imaging diagnosis
Anatomical diagnosis
Supplementary diagnosis
Age in Years
Dilatation in Millimetres
0
3
5
3.9
10
4.5
15
5.0
20–29
5.9
30–39
6.3
40–49
6.7
50–59
7.2
60–69
7.7
70
8.5
Imaging modalities
Complications
Regurgitation of pancreatic juice and formation of protein plugs
Pancreatitis
Biliary calculi
Biliary cancers (Graphic 9.16.4)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree