PAROTID INFECTIONS
KEY POINTS
- Imaging, especially computed tomography, sometimes provides critical information in the management of parotid infections and is key in determining whether a parotid abscess is present.
- Imaging can establish the source of a parotid-region infection when it may not be clear clinically.
- Computed tomography and magnetic resonance imaging may identify changes suggestive of autoimmune sialoadenitis before that diagnosis is established clinically.
The vast majority of parotid inflammatory conditions do not come to imaging. Imaging may be used to help in medical decision making when the source of a parotid-region infection is in question or if a parotid abscess might be a complicating feature. Clinical examination of an acutely infected parotid gland is limited by severe pain and toughness of the parotidomasseteric fascia. Sometimes, low-grade, chronic infections can mimic tumor and present predominantly as a parotid mass. Occasionally in chronic repeated infections, imaging will help to confirm the diagnosis and etiology and decide whether parotid surgery should be part of the management plan.
Noninfectious inflammatory parotid diseases may be manifestations of systemic autoimmune disease, other systemic inflammatory disease, or localized disease of unknown cause. These are discussed in Chapter 178 and have important implications with regard to their creating increased risk for lymphoma and their association with human immunodeficiency virus (HIV) infection.
ANATOMIC AND DEVELOPMENTAL CONSIDERATIONS
Applied Anatomy
Parotid Gland
The important anatomic considerations in parotid infections confined to the gland mainly take into account the variations in the gland’s normal relative fat and glandular tissue and the ductal system anatomy. When infection might spread to the surrounding structures, the anatomic focus expands to the muscles, bones, vessels, and nerves that come in contact with the gland. Also, the parotid capsule attachments will direct the spread of infection. These two latter considerations are also important when the source of infection may be other than the parotid gland. This anatomy should be reviewed in detail at this point, if necessary, in Chapter 175 and with regard to the facial nerve in Chapter 179. The following anatomic relationships must be understood to properly ensure that imaging data is incorporated into the assessment and ultimately the treatment of parotid infections:
- Ductal system within the gland and then through the buccal space to its point of drainage in the upper gingivobuccal sulcus across from the second molar
- Attachments of the parotid capsule to the external auditory canal and its commonality with masseteric fascia
- Facial nerve and the anatomic landmarks that relate to the surgical approach to facial nerve preservation
- Deep relationships of the gland to the parapharyngeal spaces
- Parotid lymph nodes and their drainage patterns
- For spread beyond the gland capsule, the surrounding musculoskeletal relationships of the temporomandibular joint, masticator space, mandible, maxilla, temporal bone, and more central skull base to the parotid gland
IMAGING APPROACH
Techniques and Relevant Aspects
Computed Tomography
Specific computed tomography (CT) protocols for various indications appear in Appendix A and are discussed in more detail in Chapter 175. Theoretically, calcification might be masked by contrast use, but this does not justify the cost or radiation burden of routine pre- and postcontrast studies.
Magnetic Resonance Imaging
Specific magnetic resonance (MR) protocols for various indications appear in Appendix B. A steady state image may be included that allows for MR depiction of the ductal system, sometimes referred to as MR sialography.
Radionuclide Studies
Radionuclide studies are not used routinely for the evaluation of parotid gland masses. Those using technetium, iodine, and fluorine-18 2-fluoro-2-deoxy-D-glucose (FDG) activity are used in highly selected circumstances.
Ultrasound
Standard high-resolution scanning techniques are used as described in Chapter 4. Ultrasound (US) and conventional sialography can be used as an adjunct to sialoendoscopy.
Conventional Sialography
Conventional sialography has been used very selectively since the 1980s and most recently in conjunction with endoscopic treatment of main duct pathology.
Pros and Cons
General Approach
Magnetic Resonance and Computed Tomography
MR and CT are the most-used imaging studies to evaluate an acute parotid-region infection. CT is preferred for its simplicity and to avoid the insensitivity of magnetic resonance imaging (MRI) to stones both within the ductal system and the parenchyma (Fig. 177.1). The dimension of ductal system visualization added by MR sialography is typically not of enough critical value in cases of acute inflammation to supplant CT. In chronic inflammatory conditions, MR might be a first choice. Both CT and MRI might be deferred in favor of US and sialoendoscopy in the hands of experienced groups when main duct pathology is believed to be the most likely contributing factor.
Ultrasound and Conventional Positive Contrast Sialography
US may be used to determine whether there is ductal dilatation or stones or a parotid abscess. US alone may be enough imaging for triage to appropriate care in selected cases in the hands of experienced sonographers. This is especially true where teams that triage with US for potential follow-up sialoendoscopy are in place. Sialography may also fit well into such a triage scheme for main duct disease possibly following a nondefinitive US in the diagnostic hierarchy (Fig. 177.1). However, contrast should be placed in the parotid duct with great caution if there is evidence of active inflammation and especially if the infectious agent may be a pyogenic bacterium.
Radionuclide Studies
Radionuclide studies are not used routinely for the evaluation of parotid gland infections.
Specific Indications
Specific indications for parotid region infection include the following:
- Define the origin and extent of a parotid region infection, determining whether it arises from within the parotid gland or from an extrinsic source (Fig. 177.2).
- Define the presence and extent of a parotid abscess and its relationship to the facial nerve and surrounding structures and spaces outside the parotid if an abscess is present (Figs. 177.2 and 177.3).
- Identify ductal changes or sialolithiasis that might contribute to a diagnosis and treatment plans (Fig. 177.1).
- Establish whether the changes may be part of a generalized major salivary gland condition and/or possibly related to a systemic inflammatory process (Fig. 177.2 and Chapter 178).
Controversies
US may be promoted as a starting point in the evaluation of parotid disease; however, the sonographic evaluation of the deep lobe is fundamentally limited by the bony confines of the mandible and mastoid. Using US as a starting point is a very good approach but only in experienced hands. Outside of that context, US becomes potentially cost additive and nondefinitive.
MR sialography had been promoted as a screening tool for Sjögren disease in the past. However, there are more cost-efficient and reliable means of establishing such a diagnosis. Sjögren disease is primarily a clinical diagnosis that is confirmed by serologic studies. In serologically negative patients with highly suspicious findings of dry eyes and mouth, imaging may aid in medical decision making. Unless there is a risk of significant reaction to iodinated contrast, MRI should defer to CT if an advanced imaging technique is necessary for medical decision making.
The value of using a combination of sialography and endoscopic treatment of ductal pathology is becoming more widespread. This is not controversial but does require a careful coordinated effort between the sialendocopist and the imaging team.
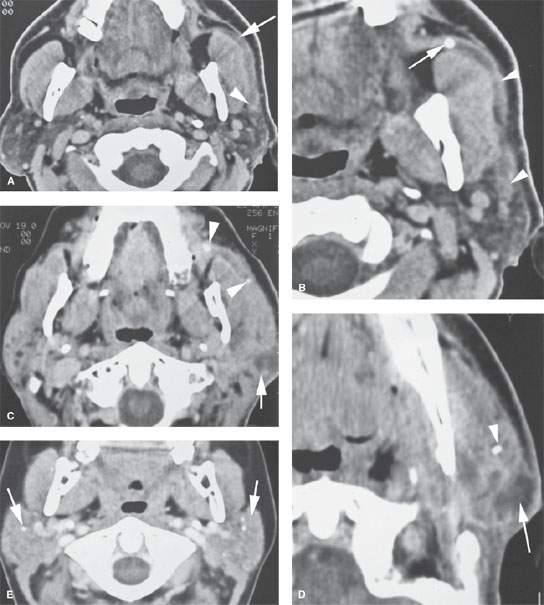
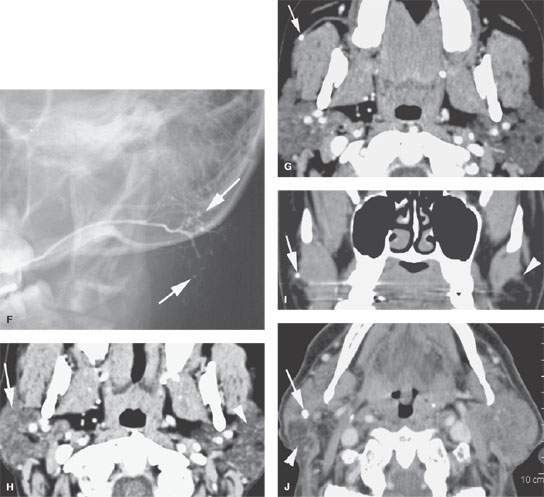
FIGURE 177.1. Five patients showing various degrees of ductal pathology and glandular calcification as part of infectious inflammatory parotid disease. A, B: Patient 1 presenting with acute and subacute pain and tenderness in the left parotid region. In (A), the parotid duct is dilated as it overlies the masseter muscle (arrow) and extends to its major branch point within the substance of the parotid gland (arrowhead). The duct wall enhances. In (B), the obstructing stone is visible just beyond the buccal space, and the ductal dilatation and wall enhancement is seen extending well back into the gland parenchyma (arrowheads). C, D: Patient 2 presenting with pain and tenderness over the left parotid gland. In (C), stones can be seen impacted within the dilated main parotid duct (arrowheads). The duct wall enhances. Farther back in the gland, there is a developing parotid abscess (arrow). In (D), a section more inferiorly shows stones likely within the ductal system and a discreet parotid abscess secondary to infection at the tail of the gland (arrow). E, F: Patient 3 had contrast-enhanced computed tomography (CECT) in (E) showing generalized increased density of the parotid gland, which can be a normal variant; however, multiple stones are present (arrows). This glandular pattern, while possibly normal, can also be an indication of chronic sialoadenitis. In (F), a view from a sialogram shows the pattern of the chronic punctate sialoadenitis (arrows) and a relatively normal main ductal system. The more distal ducts are narrowed, irregular, and pruned. G–I: Patient 4 presenting with pain in the right ear. An otalgia workup was performed, and the cause of the pain was chronic right sialoadenitis manifesting on CECT in (G) and (H) by some slight but generalized parenchymal enhancement on the right (arrow) compared to the normal left gland and a small stone in the duct (arrow). A partially obstructing stone (arrow) with some periductal swelling is confirmed in the coronal section (I) compared to the normal duct on the left (arrowhead). J: Patient 5 with a CECT showing bilateral chronic sialoadenitis due to chronic repeated infections. A stone can be seen on the right side (arrow), and atrophy is present in the tail of the gland (arrowhead) due to chronic obstructive disease.
ACUTE AND SUBACUTE PYOGENIC BACTERIAL INFECTIONS, VIRAL INFECTIONS, AND CHRONIC INFECTIONS
Etiology
Viral infections are common but usually do not come to imaging except for unilateral disease that mimics bacterial infection that might be thought to require imaging (Fig. 177.4). The most well known is the mumps virus, but other forms including Epstein-Barr virus and HIV can present as primarily parotid problems (Fig. 177.5). On occasion, viral or bacterial conjunctivitis, blepheritis, or cutaneous infections will cause reactive or suppurative lymphadenitis of periauricular lymph nodes mimicking parotid infections.
Infections of the parotid that come to imaging are often acute or subacute pyogenic bacterial infections, with these studies being mainly done to look for a complicating abscess, an underlying cause, or if the parotid origin is not certain. Chronic persistent or recurrent bacterial infections are usually studied to identify a cause or complicating factor such as duct obstruction or stones. Occasionally, a chronic inflammatory process will present as a “mass.” Fungal infection is very rare. Parotid tuberculosis and unusual infections such as actinomycosis are rare and difficult to anticipate prior to tissue sampling and culture.
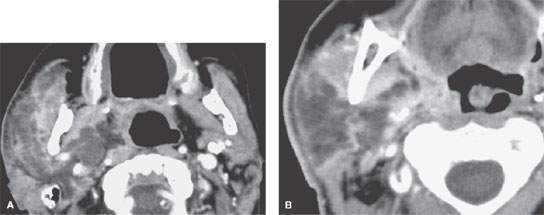
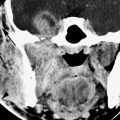
FIGURE 177.2. Computed tomography study of a patient with diabetes who presented with generalized swelling involving the right cheek. Clinically, it was not certain whether this was a dental or other type of infection. In (A), the infection was due to an acute suppurative parotid infection spreading to the parapharyngeal space. In (B), the infection can be seen to be confined to the parotid gland and the parapharyngeal space. The pterygoid and masseter muscles are secondarily inflamed, but the disease was clearly not of dental origin. This was due to a rapidly spreading anaerobic infection in a poorly controlled diabetic patient.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree