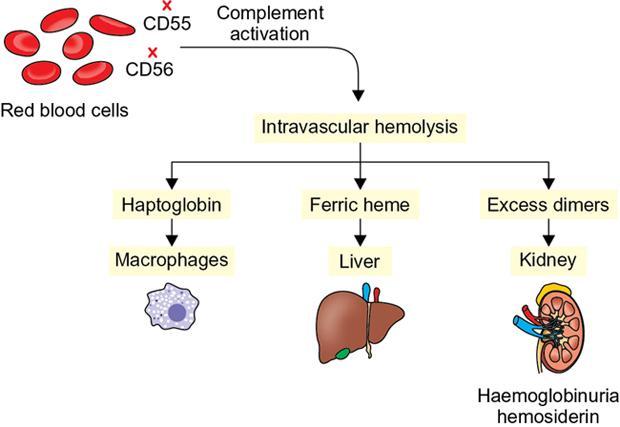
Paroxysmal nocturnal haemoglobinuria (PNH) is characterized by an increased sensitivity to complement-mediated erythrocyte lysis leading to cyclical haemoglobinuria. It is also known as Marchiafava-Micheli syndrome. Although commonly regarded as a type of haemolytic anaemia, PNH is in reality, a myelodysplastic, haematopoietic stem-cell disorder. It is most common in second decade of life with a male preponderance. DIAGNOSTIC PEARLS AND PITFALLS Most common presentation includes haemolytic anaemia with symptoms of fatigue, jaundice, and red or black urine. Some patients can present with thrombosis due to hypercoagulable state induced by complement activation. Rarely patients can have pain abdomen, dysphagia or erectile dysfunction due to depletion of nitric oxide by the free haemoglobin in the circulation. Recurrent episodes of haemoglobinuria lead to renal insufficiency and pulmonary hypertension over time. PNH occurs due to somatic mutation in the gene on the X-chromosome which codes for a protein involved in the assembly of phosphatidylinositol glycan (PIG), which anchors many proteins to the surface of cell membranes. Due to lack of PIG, the two protective proteins which inactivate complement complexes, decay accelerating factor (DAF; CD55) and membrane inhibitor of reactive lysis (MIRL; CD59) are not present in PNH red cells, which then become sensitive to lysis by complement. PNH can lead to chronic renal failure due to deposition of haemosiderin in the proximal convoluted tubules in the renal cortex or due to repeated microinfarctions secondary to microvascular thrombosis or direct nephrotoxic effect of iron. Fig. 10.12.4.2.1 depicts the pathogenesis of PNH. Ultrasound can only demonstrate morphological alterations in the kidneys due to secondary chronic renal failure but does not demonstrate iron deposits. CT without intravenous contrast can show high attenuation of the renal parenchyma. MRI shows low signal on both T1- and T2-weighted images with inversion of the signal intensity of the renal cortex (i.e. reversed renal cortex-medulla differentiation) due to ferric iron present in the haemosiderin which deposits in the renal cortex. T2-weighted imaging has higher magnetic susceptibility and shorter examination times and is hence superior to T2-weighted imaging in detection of renal cortical haemosiderosis. In case of PNH, bone marrow signal on MRI is normal. Bone marrow intensity shows fatty degeneration with ageing. The levels of iron deposits in the liver and spleen are usually normal in PNH which is in contrast to other haemolytic anaemias. Decrease in signal intensity of liver and spleen on MRI is seen in patients with PNH who have received multiple blood transfusions. In acute attack, the use of steroids is still debatable. Transfusion therapy helps in correcting anaemia and suppresses the production of PNH cells by the bone marrow, thereby reducing the severity of haemolysis. Anticoagulation with warfarin decreases the risk of thrombosis. Monoclonal antibody that targets complement protein C5, Eculizumab, is FDA approved for the treatment of PNH. It significantly reduces haemolysis and leads to improvement in anaemia. MR is the best imaging method to demonstrate iron overload in the renal cortex in patients with PNH. Bone marrow signal on MRI is normal in PNH. RENAL DISORDERS OF MIGRATION AND RENAL ANOMALIES Priscilla Joshi, Anand Mukund Rahalkar, Nagesh Seth, Mangal Mahajan, M.D. Rahalkar, Vandana Jahanvi, Aparna Katdare, Palak Bhavesh Popat, Nilesh P. Sable, Ganesh Bakshi Renal anomalies encompass a spectrum which ranges from lethal renal agenesis to incidentally detected renal anomalies secondary to aberrant embryonic migration. Imaging helps in the early diagnosis, detection of complications, surgical planning and follow-up of these patients. On an embryological basis, anomalies of the kidney and urinary tract can be divided into: Imaging modalities used for evaluation include: A number of anomalies can be detected antenatally on ultrasound. An anomaly scan includes evaluation of the kidneys – number, size, appearance, dilatation of the collecting system and presence of a distended urinary bladder. An over distended urinary bladder with bilateral hydronephrosis and hydroureter would indicate presence of a bladder outlet obstruction. Abnormalities if any, can be confirmed with a foetal MRI so that appropriate timely management can be instituted. These foetuses merit postnatal evaluation after birth and 4–6 weeks later. Plain radiographs may be done in calculus disease and give a clue as to the presence of an anomaly.
2. Paroxysmal nocturnal haemoglobinuria
Introduction
Clinical features
Aetiopathogenesis
Diagnostic imaging
Differential diagnosis
Categories
Conditions
MRI Features
Haemolysis
Paroxysmal nocturnal haemoglobinuria
Low signal intensity in the renal cortex
Mechanical haemolysis
Low signal intensity in the renal cortex
Sickle cell disease
Low signal intensity in the renal cortex and spleen
Infection
Haemorrhagic fever with renal syndrome
Well-defined zone of low signal intensity in the outer medulla
Vascular disease
Acute renal vein thrombosis
Renal swelling, indistinct corticomedullary differentiation on T1-WI, low signal intensity of the renal cortex and medulla
Renal cortical necrosis
Low signal intensity of the inner renal cortex. Swelling of both kidneys and loss of corticomedullary differentiation on T2-WI
Arterial ischaemia and infarction
Infarcted area shows low signal intensity on both T1- and T2-WI. Signal intensity may be higher in haemorrhagic infarcts
Rejection of a transplanted kidney
Loss of corticomedullary differentiation. Low signal intensity is seen if combined with diffuse haemorrhagic necrosis or cortical necrosis
Acute nonmyoglobinuric renal failure
Initially high signal intensity on T1-WI and variable signal intensity on T2-WI. Low signal intensity on both T1- and T2-WI after the 7th day
Treatment
Conclusion
Introduction
Imaging modalities
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree