Abstract
Parvovirus B19 is a single-stranded deoxyribonucleic acid (DNA) virus that has the potential for transplacental transmission in susceptible mothers. Congenital infection has been associated with severe fetal anemia, myocarditis, fetal hydrops, and death. Serologic testing is used to confirm the diagnosis of maternal parvovirus infection. Fetal infection is diagnosed by detection of viral DNA in fetal blood or amniotic fluid. Once maternal infection is confirmed, the fetus should be evaluated for sequelae of congenital infection. Serial ultrasounds to assess for signs of anemia and hydrops fetalis are recommended every 1–2 weeks for 8–12 weeks following exposure. Middle cerebral artery peak systolic velocity via Doppler velocimetry is considered the optimal method for detection of fetal anemia. Cordocentesis is performed if fetal anemia is suspected and, if confirmed, is followed by intrauterine blood transfusion. In general, long-term outcomes for infants who survive intrauterine infection are favorable.
Keywords
parvovirus B19, fetal anemia, hydrops fetalis
Introduction
Parvovirus B19 infection during pregnancy is rare, but potentially dangerous. In susceptible mothers, transplacental transmission of parvovirus B19 is possible. If acquired in utero , parvovirus B19 infection can lead to severe fetal anemia and myocarditis. In some instances, the infection may result in fetal death in the second or third trimester. Although in utero fetal infection is usually self-limited, severe cases are treated with intrauterine fetal blood transfusion to maintain fetal red blood cell levels until the fetus recovers from the infection.
Disorder
Prevalence and Epidemiology
Parvovirus B19, a single-stranded deoxyribonucleic acid (DNA) virus, is most commonly associated with erythema infectiosum (also referred to as fifth disease ). Although one-third of infections are asymptomatic, the virus may also cause maternal arthritis, aplastic crisis, and hydrops fetalis.
Parvovirus B19 is primarily transmitted by respiratory droplets and infected blood products. The patient is considered infectious 5 to 10 days after exposure and before the onset of a rash or other symptoms. When a rash erupts, the patient is no longer infectious. Approximately 50% to 60% of women of reproductive age have evidence of prior infection. The secondary attack rate for the household is approximately 50%. Susceptible individuals exposed in a classroom setting have a 20% to 30% chance of acquiring the disease. Of pregnant women, 40% to 50% are immune to parvovirus B19. The incidence of this acute infection in pregnancy ranges from 3%–4%. The risk of fetal transmission, which does not change with gestational age, is approximately 33%. The rate of fetal loss ranges from 2%–9% among women with serologically proven parvovirus B19 infection. However, fetal loss rate appears to be significantly higher if the disease is acquired before 20 weeks’ gestation (11% versus <1%). Although immunity is lifelong, there have been reports of reinfection.
Etiology and Pathophysiology
The incubation period for parvovirus is 10 to 20 days. Manifestations of erythema infectiosum include low-grade fever, malaise, myalgias, arthralgias, and a “slapped cheek” facial rash. An erythematous, lacelike rash may extend onto the torso and upper extremities. In children, parvovirus infection can cause a transient aplastic crisis. An aplastic crisis may also occur in adults with an underlying hemoglobinopathy.
When maternal parvovirus infection occurs during pregnancy, the virus can cross the placenta and infect red blood cell progenitors in the fetal bone marrow. The virus attaches to the P antigen on red blood cell stem cells and suppresses erythropoiesis, resulting in severe anemia and high-output congestive heart failure. This same antigen is also present on fetal myocardial cells, and in some fetuses, the viral infection causes a cardiomyopathy that contributes further to heart failure. As long as fetal anemia is treated, spontaneous recovery is expected. Fetal complications are unlikely to occur more than 8 to 10 weeks after the initial exposure to the virus. Epidemiologic studies have failed to establish any association between parvovirus B19 infection and congenital malformations.
Manifestations of Disease
Clinical Presentation
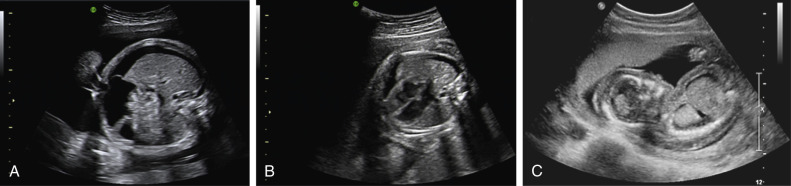
Hydrops fetalis is the most obvious and serious manifestation of an intrauterine parvovirus B19 infection, which is seen in approximately 4% of all affected pregnancies ( Fig. 167.1 ). The risk of hydrops depends on the gestational age at which maternal infection occurs. If infection develops during the first 12 weeks of gestation, the risk of hydrops ranges from 5%–10%. If infection occurs during weeks 13 through 20, the risk decreases to 5% or less. Infection after the 20th week of gestation is associated with a fetal hydrops risk of 1% or less. In addition to fetal anemia, parvovirus B19 has been linked to late second-trimester and third-trimester intrauterine fetal death. Most of these fetuses are not hydropic.
Serologic testing is used to confirm the diagnosis of maternal parvovirus infection. Enzyme-linked immunosorbent assay, radioimmunoassay, or Western blot can be used to determine maternal serologies. The sensitivity of the serologic immunoglobulin (Ig)G and IgM testing is approximately 80%. IgM results should be evaluated cautiously because false-positive results can occur. IgM may be present in the maternal circulation for 3 months or longer. Possible serologic test results are presented in Table 167.1 .
| IgM | IgG | Interpretation |
|---|---|---|
| Negative | Negative | Susceptible |
| Negative | Positive | Immune |
| Positive | Negative | Acute infection (within previous 7 days) |
| Positive | Positive | Subacute infection a |
a Caution: Occasionally low levels of IgM antibodies may persist for >12 months after infection. Possible exposure time should be determined before further surveillance is planned.
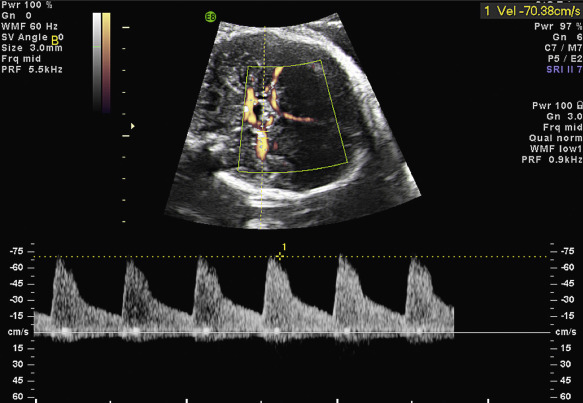
Fetal infection can be diagnosed by detection of parvovirus-specific IgM in fetal blood by enzyme immunoassay, immunofluorescence test, or radioimmunoassay and by detection of viral DNA in fetal blood or amniotic fluid by the polymerase chain reaction technique. When maternal infection is confirmed, the fetus should be evaluated for evidence of anemia. Assessment of the middle cerebral artery (MCA) peak systolic velocity via Doppler velocimetry is considered the optimal method for detection of fetal anemia ( Fig. 163.2 ). Serial ultrasound (US) examinations should be performed every 1 to 2 weeks for 8 to 12 weeks after exposure.
The most obvious US manifestation of fetal anemia is hydrops (see Fig. 167.1 ). By the time US evidence of hydrops is present, the fetal hematocrit is likely to be less than 20%.
Imaging Technique and Findings
Ultrasound
Grayscale Ultrasound.
The fetus should be evaluated for the presence of hydrops. A complete anatomic survey of the fetus should be performed to rule out any defect that may account for hydrops, including vascular lesions, which may lead to high-output cardiac failure, structural heart defects, and cardiac arrhythmias.
Middle Cerebral Artery Doppler Ultrasound.
Historically, measurement of amniotic fluid bilirubin levels with spectral analysis at 450 nm (ΔOD 450 ) was used to assess the severity of erythroblastosis in utero . However, because the anemia in parvovirus B19 infection is caused by bone marrow suppression, this approach is unreliable in assessing the status of the fetal anemia.
Doppler velocimetry in the fetal MCA is a precise, noninvasive way to detect fetal anemia (see Fig. 167.2 ). With this approach, moderate or severe anemia is predicted by values of peak systolic velocity in the fetal MCA above 1.5 times the multiples of median for gestational age ( Table 167.2 ), with a sensitivity of 100% and a false-positive rate of 12% in fetuses with red blood cell alloimmunization. Increased MCA peak systolic velocity can also reliably predict anemia in fetuses with parvovirus B19 infection. If velocimetry indicates fetal anemia, cordocentesis should be performed to determine the fetal hematocrit, unless the fetus is mature enough for delivery. An intrauterine blood transfusion should be performed when anemia has been confirmed. It has been shown that MCA peak systolic velocity can be reliably used even in fetuses with prior transfusions.