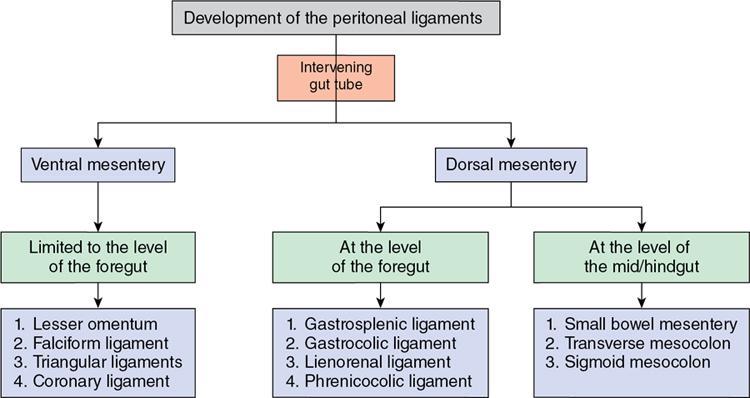
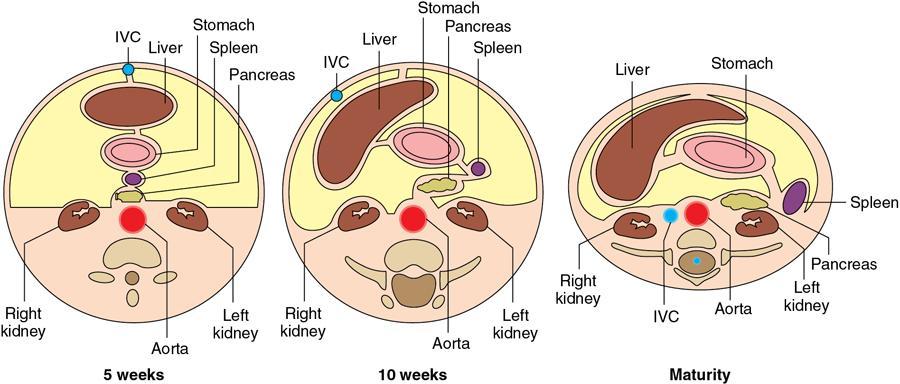
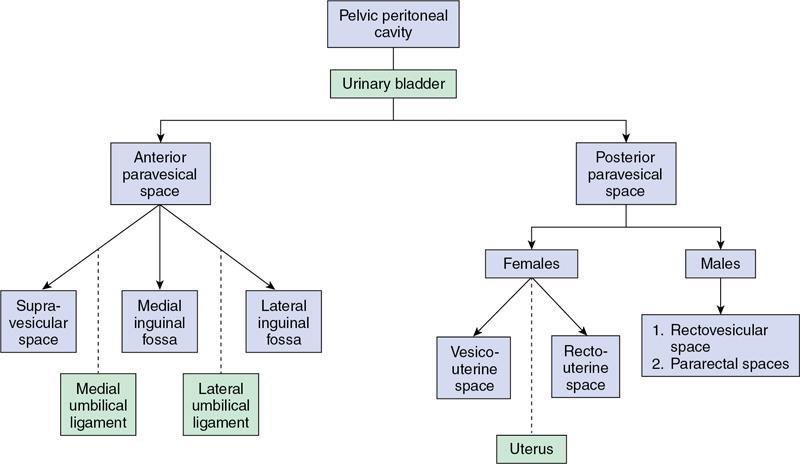
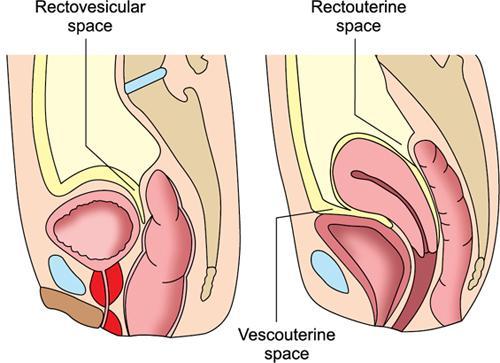
Shivsamb Jalkote, Slesha Bhalja, Disha Lokhandwala, Mansi Jantre, Karthik Ganesan Peritoneum is involved in a diverse group of primary as well as secondary pathologies ranging from the infective/inflammatory spectrum to malignancies. Peritoneal diseases are both common and potentially life threatening, and yet often get neglected in routine evaluation, due to the complex peritoneal anatomy and sometimes subtle imaging manifestations. The radiologist forms an integral part of the multidisciplinary team dealing with peritoneal pathologies and hence, having a lucid understanding of peritoneal anatomy, pathways of disease spread and different treatment options is paramount for an accurate radiological analysis. All three major serosal cavities (pleural, pericardial and peritoneal) of the body are derived from a single coelomic cavity, which is lined by a continuous mesothelium. At 2 weeks of gestational age, during the gastrulation stage, the embryo is a trilaminar disc, composed of the endoderm, mesoderm and ectoderm. During this stage, the development of the coelomic cavity commences as a consequence of morphological changes in the mesodermal cells leading to a coalition of the intercellular spaces. Peritoneum originates from mesoderm with its parietal layer developing from somatic mesoderm and visceral layer developing from splanchnic mesoderm. Initially, the peritoneum is an empty sac into which the growing gut tube protrudes (at 3 weeks of gestational age), resulting in the formation of the dorsal mesentery (formed by the fusion of two layers of the splanchnopleuric mesoderm) by which the gut tube is suspended lengthwise. The mesodermal cells opposed to the gut tube represent the future visceral peritoneum and those lining the body wall and the septum transversum form the future parietal peritoneum. Analogously, the ventral mesentery originates from the mesoderm of the septum transversum with the closure of the anterior abdominal wall, and, is short and not as extensive as the dorsal mesentery. The two mesenteries are aligned in the sagittal plane, subdividing the primitive peritoneum into right and left cavities, which are continuous anteriorly below the inferior edge of the ventral mesentery. With the development of various abdominal organs as buds from the gut tube, the capacity of the abdominal cavity diminishes, pushing the midgut loop into the umbilical cord. This physiologic herniation of the midgut takes place at the 6th week of gestation. Simultaneously, as the peritoneum drapes over the developing organs, there is compartmentalization of the cavity and a pattern of folds develops that eventually form the peritoneal ligaments. The majority of these ligaments arise from the ventral or dorsal mesentery. As the hepatic bud develops within the ventral mesentery, derivatives of the ventral mesentery can be divided based on their relation to the liver. The gastrohepatic ligament (lesser omentum) develops dorsally and the falciform, coronary, and triangular ligaments develop ventral to the liver. Conversely, the dorsal mesentery spans between the gut tube and the posterior abdominal wall, and the spleen and pancreatic tail develop within it. In congruence, it forms the gastrophrenic, gastrosplenic and splenorenal ligaments and peritoneal folds of the small and large intestines. As stated earlier, the dorsal mesentery has a relatively larger caudad extension and since the spleen develops in its upper portion, the rapidly growing mesentery beyond the spleen forms the greater omentum. Finally, the differential growth of the greater curvature of the stomach and the right hepatic lobe results in rotation of the abdominal organs and their mesentery, and the cephalad portion of the peritoneal cavity is divided into a larger ventral cavity, called the greater sac, which caudally communicates with the common peritoneal cavity, and a smaller dorsal cavity called the lesser sac. The key aspects of peritoneal development are summarized in Flowchart 8.6.1 and Fig. 8.6.1. Laparoscopy remains the gold standard in the assessment of peritoneal disease, however, it is an invasive technique, and has certain diagnostic limitations including the inability to assess parenchymal diseases, extraperitoneal diseases, and also has limited utility in the presence of adhesions. Hence laparoscopy is not recommended as the first-line modality and has been replaced by cross-sectional imaging in routine clinical practice. The role of imaging is primarily to assess disease burden in terms of volume and extent and to identify surgically nontreatable disease, which reduces open and shut laparotomies. Peritoneal metastatic disease burden is assessed using peritoneal carcinomatosis index (PCI) both surgically as well as using cross-sectional imaging, details of which will be discussed subsequently. CT, MRI and PET/CT are all commonly used imaging modalities, however, there is no singular universally accepted reference imaging standard for the peritoneal disease. In a recent meta-analysis evaluating the diagnostic performance of imaging in the detection of peritoneal metastasis (PM), authors concluded that MRI (DWI) and PET/CT show comparable diagnostic performance in gastrointestinal and ovarian cancer malignancies however the widespread availability of MRI makes it the more likely imaging modality of choice in the future. The results of this study are summarized in Table 8.6.1. Conventional imaging including radiography and barium studies have a poor diagnostic yield in evaluating peritoneal pathologies. Abdominal radiographs are usually restricted for identifying pneumoperitoneum and bowel obstruction in the setting of acute abdominal pain. Diffuse increase in radio-opacity of the abdomen, poor definition of soft tissue shadows (psoas, liver and spleen), bulging flanks along with medial displacement of small bowel loops from the properitoneal fat stripe are indirect markers of large ascites which can prompt further evaluation with ultrasonography. Indirect features of peritoneal disease on barium studies include mass effect and displacement of adjoining bowel loop, narrowing of bowel loop with bowel dilatation along with nodularity, spiculation, or tethering of adjacent mucosal folds or haustra with sacculation of the uninvolved contralateral border. Barium studies have fallen out of favour in routine radiological practice owing to the widespread use of cross-sectional imaging. Ultrasonography (US) is a readily available and relatively inexpensive imaging tool. Traditionally its role in the evaluation of peritoneal pathologies is limited as US beam gets strongly reflected at the interface of air and visceral structures which leads to obscuration of underlying structures. US remains the primary imaging modality in the evaluation of an acute abdomen and is useful in detecting of ascites, focal lesions and any focal abnormality at the site of tenderness. US has the unparalleled advantage of providing real-time assistance in performing imaging-guided procedures such as paracentesis and peritoneal biopsies. US can be used in characterization of focal lesions (as solid, solid-cystic and cystic) and assessment of internal vascularity using colour Doppler. Transvaginal US provides a good overview of pathologies involving pouch of Douglas besides the uterus and adnexa. Experienced and skilled sonologists can detect omental thickening, peritoneal nodules and small surface deposits using high-frequency transducer (Fig. 8.6.2). Endoscopic US allows assessment of diseases involving peritoneal ligaments besides assistance in a guided biopsy. The reported sensitivity, specificity and accuracy of EUS-FNA versus CT/MRI in detecting PM was 91% versus 28%, 100% versus 85% and 94% versus 47%, respectively. CT is the most commonly employed imaging modality in the evaluation of peritoneal pathologies due to its widespread availability, fast acquisition and high spatial resolution. Current generation multidetector CT scanners (64 slices and above) offer very thin collimation, leading to the acquisition of near isotropic volumetric data which allow reformations in coronal and sagittal images without any added imaging time. It is the modality of choice for preoperative staging in most primary abdominal and pelvic malignancies (e.g. stomach, appendix, ovary) owing to its ability to detect local and regional infiltration, nodal involvement and distant metastases. CT is also used to evaluate PCI and detect disease in critical and occult locations, thus significantly altering further management strategies. CT is the most common imaging modality used in postoperative surveillance and evaluation of postoperative recurrence. CT protocol includes administration of both enteric contrasts (oral and rectal) and intravenous contrast (Table 8.6.2). Optimal bowel distension is key as collapsed loops can mimic disease and also mask the serosal and endoluminal disease. Often, a CT scan of the chest is performed as an adjunct to rule out pleuroperitoneal/transdiaphragmatic dissemination. Helical (≥64 slice scanner) Dual energy scanner if available As thin as possible (≤1 mm) Same as section thickness without gap From xiphisternum to pubic symphysis Chest included if suspicion of thoracic involvement 1 L of positive oral contrast over duration of 60 minutes and 500 mL of positive rectal contrast on table before administration of IV contrast 1.5–2 mL/kg of nonionic contrast with high iodine concentration (≥ 300) at rate of 3–5 mL/sec Unenhanced imaging not recommended. Monophasic acquisition at 60 seconds usually sufficient in majority cases Arterial phase at 20 seconds acquired for arterial assessment Delayed phase in cases of ureteric obstruction, slow bowel transit and suspected leak. Axial 1 mm thickness, multiplanar reformats in coronal and sagittal planes at 1 mm MIP or 3D volumetric sections for vascular assessment There is a wide range of reported diagnostic accuracy (sensitivity ranging from 25% to 100%, specificity ranging from 78% to 100%) of CT in the assessment of PM depending on the expertise of the reader and scanner type. In general, key factors affecting CT sensitivity include the size of lesion, the region of affection and presence of ascites. 64 slice scanners with multiplanar reformations allow 100% sensitivity and positive predictive value in detecting lesions measuring >5 cm, which partly diminishes for lesions between 0.5 cm and 5 cm in size with sensitivity and positive predictive being 83% and 93%, respectively. It receives the most criticism in the evaluation of lesions < 0.5 cm with significantly low sensitivity and positive predictive value (43% and 76%, respectively). There is also a great degree of variation in sensitivity based on the anatomical location of the disease. In a study by Koh et al., the detection rates ranged from 8% to 67%, depending on the region involved; only the epigastrium exceeded 60%, with the small-bowel disease being the most poorly visualized (8%–14%). The sensitivity for tumour detection in epigastrium, greater omentum and under surfaces of the diaphragms was 60%–90%, while it was 50%–70% in the retroperitoneum and pelvis, and small bowel-mesentery involvement was detected in merely 20%–50% of cases. These are noteworthy findings, as small-bowel involvement has major implications on outcome and is one of the limiting factors for complete cytoreduction. Poor soft tissue resolution of CT doesn’t allow accurate differentiation between mucinous deposits from ascites. Administration of positive enteric contrast can mask calcified serosal deposits. DECT uses two separate x-ray energy spectra and allows characterization of tissues based on their differences in attenuation properties at different energies. Though the role of DECT in the assessment of abdominal pathologies is increasingly reported in the literature, there are very limited data available on its application in assessment of peritoneal pathologies. The combination of iodine overlay with conventional imaging has shown a better specificity in differentiating PM from benign peritoneal entities, and hence can be particularly useful in the postoperative setting. MR imaging offers excellent soft tissue resolution, multiplanar capabilities and avoids exposure to ionizing radiation, making it an attractive tool for evaluation of peritoneal diseases. MRI is better suited and can be problem solving in visualization of smaller lesions (<1 cm) and assessment of relatively concealed areas such as subphrenic, mesenteric and bowel serosa. Unenhanced T1- and T2-weighted images can show larger peritoneal deposits and masses but are relatively insensitive for the depiction of smaller lesions. The combination of diffusion-weighted imaging (DWI) and delayed gadolinium-enhanced MR imaging is considered the most accurate technique for detecting peritoneal tumours (Fig. 8.6.3). MRI protocol for assessment of peritoneum has been detailed in Table 8.6.3. In a study by Low RN et al., MRI PCI correctly categorized tumour volume in 91% of patients compared to surgical PCI, as opposed to 50% with CT. In addition, MRI demonstrated per site sensitivity of 95%, specificity of 70% and accuracy of 88%, while CT showed a corresponding per site sensitivity of 55%, specificity of 86% and accuracy of 63%. MRI PCI has also shown to have better prediction of intraoperative burden of disease for invasive appendiceal and peritoneal mesothelioma histologies as compared to CT-PCI. *This general imaging protocol is based on 3 T MRI Siemens system, covers both abdomen and pelvis. Protocol is modified depending on primary disease and type of scanner. Low RN. Preoperative and surveillance MR imaging of patients undergoing cytoreductive surgery and heated intraperitoneal chemotherapy. Journal of gastrointestinal oncology. 2016 Feb;7(1):58. The higher cellularity of majority of peritoneal nodules results in restriction of water movement and corresponding high signal intensity on DW images. On the higher b-value images, bowel contents are suppressed and the serosal and peritoneal tumours become hyperintense. DWI is also useful to demonstrate associated lymphadenopathy, hepatic and osseous metastases. In a study by Zhang et al., in PC of colorectal carcinoma, DWI demonstrated sensitivity of 69.4%, 91.3% and 100% for lesions measuring <0.5 cm, 0.5–5.0 cm and >5 cm, respectively. The overall sensitivity, specificity and accuracy of DWI for the detection of peritoneal tumours were 80.3%, 84.5% and 82.1%, respectively. DWI/MRI is also superior in demonstrating lesions involving small bowel wall, with an accuracy of 92%–95% versus 48% for CT. Reported negative predictive value of DWI for small bowel serosal metastases is 100%. Whole-body DWI/MRI was highly accurate for the prediction of inoperability (PPV 100%, NPV 90.3%) and was capable of detecting metastases both inside and outside the abdominal cavity. Peritoneal tumours enhance slowly and are best depicted on the final set of images obtained about 5 minutes following gadolinium administration. The increased conspicuity of these enhancing peritoneal tumours improved detection of small tumours that are often missed on CT scans. For this reason, perfect breathhold is essential while obtaining the final set of images and if images are acquired later, contrast that has diffused into the ascitic fluid can obscure smaller lesions. The high-contrast conspicuity of fat-suppressed and delayed gadolinium-enhanced MRI makes it the imaging modality of choice in depicting subcentimetre deposits (including those measuring <5 mm), and also deposits in anatomically concealed sites (e.g., subphrenic, mesenteric and bowel serosa). Gadolinium-enhanced MRI has higher sensitivity in detecting peritoneal lesions <1 cm which is about 85%–90% compared to 22%–33% for CT. Despite the clear superiority of MRI over CT, the major detractors against MRI are the long exam times and the higher expense burden. Longer acquisition times can lead to motion artefacts related to respiration and bowel peristalsis compromising detection of smaller lesions. MRI is contraindicated in patients with pacemakers, cochlear implants and claustrophobic patients with some exceptions. DW-MRI is less sensitive in special histological subtypes such as signet ring cell and mucinous adenocarcinoma due to their limited restricted diffusion. The combined use of FDG-PET with anatomical CT imaging, provides the added benefit of qualitative and semiquantitative metabolic analysis (Fig. 8.6.4). PET/CT has an established role in staging of most of the primary tumours of the chest, abdomen and pelvis. It is also helpful in assessing malignant ascites or PM of an unknown primary. In disseminated malignancies, it can localize the optimal site for biopsy depending upon metabolic activity of the lesion. PET/CT should be used selectively in preoperative assessment of patients with large tumour volumes and poor prognostic histologies being considered for CRS and HIPEC to rule out extra abdominal metastases. It can also detect extraabdominal metastases, including mediastinal and supraclavicular nodes, and other organ sites, such as bone and brain. It can also detect nodal involvement in normal-sized lymph nodes seen on conventional cross-sectional imaging. PET/CT also plays a role in disease surveillance and assessment of response to neoadjuvant chemotherapy. This is particularly relevant in malignancies where anatomical changes are so small that measurement of size change alone becomes unreliable and also in differentiating between residual/recurrent disease and postoperative changes after CRS and HIPEC. Besides oncological indications, PET/CT is also helpful in assessment of benign and inflammatory diseases to evaluate disease extent and also localize the site for biopsy. In cases of pyrexia of unknown origin, PET/CT has often been employed to demonstrate unsuspected peritoneal disease. Utility of PET/CT in peritoneal disease assessment is limited in developing counties due to its higher cost burden and lower accessibility. Certain histological subtypes such as mucinous and signet ring cell carcinomas show low FDG uptake making PET/CT less sensitive. Low metabolic uptake in these subtypes is thought to be due to low tumour cell density, higher content of metabolically inert mucin and lack of expression of glucose transporter Glut-1. This is more relevant in context of colorectal malignancy where mucinous carcinoma and signet ring cell carcinoma more frequently metastasize to the peritoneum compared to adenocarcinoma (48%, 51% and 20%, respectively). PET/CT is prone to artefacts caused by misregistration between the CT and PET images which can lead to inaccurate assessment of certain regions such as dome of diaphragm and structures adjoining the urinary bladder, making the detection of pelvic implants difficult due to partial volume effects. Implants against the small bowel and mesentery may be obscured by the physiological metabolic activity of bowel. Pretreatment PET/CT demonstrates a higher accuracy than CT for nodal and metastasis staging but has limited sensitivity to detect PM, especially when there is small-volume disease as only 13.6% of the nodules less than 0.5 cm were correctly identified by PET/CT. Table 8.6.4 summarizes role of different imaging modalities for assessment of peritoneal diseases. Peritoneum is the largest serosal membrane in human body with surface area of about 2.2 m2. It is composed of parietal and visceral layers which are continuous with each other, with parietal peritoneum lining the coelomic cavity and visceral peritoneum lining the visceral organ (Fig. 8.6.5). The peritoneum essentially minimizes friction, resists infection and stores fat, allows free movement of the abdominal viscera which gets restricted in presence of adhesions. Peritoneum acts as first line of defence in controlling disease spread, and, it exudes fluid and cells in response to injury or infection (peritonitis), to wall off or localize infection. In natural states, intraperitoneal spread of infection is limited by lymphoid aggregates and stomata on both the peritoneal layers. Omentum is commonly known as policeman of abdomen as it migrates to the site of infection and isolates affected area from healthy tissues. Iatrogenic violation of peritoneum following surgery can lead to recurrence along the operative incision scar sites and along the vaginal cuff following hysterectomy in pelvic malignancies. The parietal peritoneum is supplied by nerves (e.g. phrenic and thoracoabdominal) to the adjacent body wall, and is predominantly very sensitive to pain, which explains the typical referred shoulder pain from upper abdominal pathologies such as gall stones and pancreatitis. The visceral peritoneum is insensitive to pain and responds mainly to stretching. These two layers encompass a potential space, called the peritoneal cavity which contains approximately 50–75 mL of clear serous fluid. Peritoneal cavity is a closed space in males while in females it communicates with extraperitoneal pelvis through ostia of fallopian tubes allowing transport of ovum. This communication serves as natural route for ascending spread of sexual transmitted disease from genital organs into peritoneal cavity leading to pelvic inflammatory disease and uncommonly perihepatitis (Fitz–Hugh–Curtis syndrome). The invaginations of the peritoneum result into formation of three different types of structures namely ligaments, omentum and mesentery (Figs. 8.6.6–8.6.9) which serve as bridges that permit blood, lymph vessels and nerves to reach the viscera. Ligaments are double folds of peritoneum that connect one organ to another organ or to the abdominal wall and are named usually after the respective viscera. The liver, for example, is connected to the diaphragm by the falciform ligament, coronary ligament and right and left triangular ligaments. Mesenteries are double folds of peritoneum that suspend intestine to the posterior abdominal wall (retroperitoneum) such as the small bowel mesentery, transverse and sigmoid mesocolon. Omentum are double folds of peritoneum that extends from the stomach to other organs and include the greater and lesser omentum. Table 8.6.5 summarizes the peritoneal ligaments in abdomen and pelvis. In medical school days, the anatomy textbooks traditionally divided abdomen and pelvis into tricompartmental model comprising of peritoneal cavity, retroperitoneum and extraperitoneal spaces. This model is good for basic understanding of anatomy and confinement of certain disease processes but fails to explain spread of diseases from retroperitoneum to peritoneum besides spread of some disease processes within peritoneal cavity itself. Considering these shortcomings of conventional description of anatomy, the unifying concept of subperitoneal space has evolved in literature and is getting accepted increasingly. As per this concept, abdomen and pelvis are divided in two spaces peritoneal cavity and subperitoneal space which are separated from each other by peritoneum. Subperitoneal space (Fig. 8.6.10) is large anatomically continuous potential space which connects peritoneal cavity with retroperitoneum. It constitutes the subserosal areolar tissue and fat tissue lining the inner surfaces of the peritoneum and the abdominopelvic musculature. The omentum, ligament and mesentery are the interconnecting peritoneal folds building the subperitoneal space. All the abdominal and pelvic organs along with their vascular, lymphatic and nervous supplies are situated within subperitoneal space. With this concept, we want to correct the common misconception of intraperitoneal organs as in strict terminology there is no organ within the peritoneal cavity itself. It is imperial to understand differences between peritoneal and subperitoneal spaces as these are mutually exclusive spaces having distinct patterns of disease spread which we will discuss in subsequent sections. In resting state, peritoneal cavity is collapsed and thus not visible on routine imaging. Peritoneal cavity usually gets distended due to disease processes composed of fluid, air or masses. The peritoneal cavity is divided into two parts: the greater sac and the lesser sac which communicate with each other via an epiploic foramen (of Winslow). The greater sac forms main peritoneal cavity and extends from the diaphragm down into the pelvis. The lesser sac is small diverticulum and lies between stomach and pancreas. For better understanding, abdominal peritoneal cavity is divided into supra- and inframesocolic compartments, communicating with pelvic peritoneal cavity which is further divided by other peritoneal folds and reflection (Flowchart 8.6.2, Figs. 8.6.10 and 8.6.11). Supramesocolic compartment (Fig. 8.6.12) is situated between the diaphragm and transverse mesocolon. It is arbitrarily divided into right and left supramesocolic peritoneal spaces, which can be further subdivided into a number of subspaces by upper abdominal organs and peritoneal ligaments. Right supramesocolic space is divided into three subspaces: right subphrenic space, right subhepatic space and lesser sac. Right subphrenic space is partially separated from right subhepatic space by right triangular ligament. Right subphrenic space is situated between right hemidiaphragm and right lobe of liver (superior, anterior and right lateral surfaces). It is limited posteriorly by the coronary ligament and medially by the falciform ligament with the latter separating it from the left subphrenic space. Right subhepatic space is situated inferior to right lobe of liver and has anterior and posterior components. Anterior component is limited inferiorly by transverse mesocolon whereas the posterior component extends anterior to right anterior perirenal fascia. Posterior component of right subhepatic space is commonly known as hepatorenal pouch (of Morrison) and forms most dependent site in supine position, thus being a common site of fluid accumulation. The right subhepatic space freely communicates with right subphrenic space superiorly and right paracolic gutter inferiorly. Lesser sac (also known as omental bursa) is situated superior to the transverse mesocolon, anterior to pancreas and posterior to the lesser omentum, stomach and gastrocolic ligament. Lesser sac (Fig. 8.6.13) communicates with right subhepatic space (of greater sac) via epiploic foramen which is bounded by IVC posteriorly, free margin of hepatoduodenal ligament anteriorly, D1 (first part of duodenum) caudally and caudate lobe of liver rostrally. Division of lesser sac into smaller superior compartment (superior recess) and larger inferior compartment (inferior recess and splenic recess) is accomplished by prominent crescentic gastropancreatic fold (peritoneal fold overlying common hepatic and left gastric artery) which extends from neck of the pancreas to the upper part of lesser curvature of stomach. The superior recess is situated to the right of midline above pancreas and posterior to lesser omentum. It surrounds the medial aspect of the caudate lobe and superiorly into the fissure for the ligamentum venosum. Splenic recess extends to splenic hilum, and is limited by gastrohepatic ligament (anterior), gastrosplenic ligament (lateral) and splenopancreatic ligament (posterior). Inferior recess is located to left of midline, separates stomach from the pancreas and transverse mesocolon. In few cases, an infrapancreatic part can be seen due to persistence of inferior recess within the greater omentum or due to downward bowing of transverse mesocolon. Left supramesocolic space is wider than right supramesocolic space and is limited above by left hemidiaphragm and below by transverse mesocolon and phrenicocolic ligament. Left supramesocolic space (subphrenic part) is medially separated from right supramesocolic space by falciform ligament and inferiorly from left paracolic gutter by phrenicocolic ligament. It is divided into four communicating subspaces: anterior left subphrenic space, posterior left subphrenic space, anterior left perihepatic space and posterior left perihepatic space. All these spaces communicate with each other. The definitions of the boundaries of these spaces vary in literature. Anterior left subphrenic space is situated between gastric fundus and left hemidiaphragm. Posterior left subphrenic space (also known as perisplenic space) surrounds spleen. Anterior left perihepatic space is situated between anterosuperior aspect of left lobe of liver and left hemidiaphragm. Posterior left perihepatic space (also known as gastrohepatic recess) is situated between inferior aspect of left lobe of liver and lesser omentum. Inframesocolic compartment (Fig. 8.6.14) extends below transverse mesocolon to the level of pelvic brim and is divided into large central part and smaller paracolic part (lateral recesses). Central portion of inframesocolic compartment is unequally divided by obliquely oriented small bowel mesentery leading to formation of smaller right inframesocolic compartment and larger left inframesocolic compartment. Right inframesocolic space terminates at the ileocaecal junction and does not communicate with pelvis. Left inframesocolic space is in communication with pelvic peritoneal spaces except at the sigmoid mesocolon attachment which offers partial barrier. Peritoneal recesses on the posterior abdominal wall lateral to the ascending and descending colon form right and left paracolic gutters respectively. Both paracolic gutters communicate freely with pelvic peritoneal spaces. Right paracolic gutter is deeper than left gutter and is in free communication with right supramesocolic space. Left paracolic gutter is partially limited from left supramesocolic space due to presence phrenicocolic ligament. Pelvis constitutes about one-third of the total volume of the peritoneal cavity and is most dependent part in both supine and erect positions. Inferiorly peritoneum drapes over fundus of urinary bladder, anterior and posterior surface of uterus and upper posterior vagina in females and anterolateral surface of the upper rectum leading to formation of different peritoneal spaces. The urinary bladder subdivides pelvis into anterior and posterior paravesical spaces. The anterior paravesical space is further compartmentalized by remnant of the urachus (median umbilical ligament), the obliterated umbilical arteries (medial umbilical ligament), and the lateral umbilical ligaments (inferior epigastric vessels) into five fossae: the right and left lateral and medial inguinal fossae and the supravesical fossa. The supravesical space is seen in midline with medial and lateral inguinal fossae placed on either side. The supravesical space lies above urinary bladder between the two medial umbilical folds, is usually occupied by small bowel loops and fundus of distended urinary bladder. Just lateral to the supravesical space is the medial inguinal fossa, located between the medial and lateral umbilical folds. The femoral ring is located inferolaterally and hence ascitic fluid in the medial inguinal fossa can extend into the femoral canal. Finally, the lateral inguinal fossa is located between the lateral umbilical fold and parietal peritoneum (which reflects over the vas deferens or round ligament). It usually contains the cecum or ileum on the right and sigmoid colon on the left. Extension of ascitic fluid into the inguinal canal can occur through the deep inguinal ring, located anteromedial to the lateral inguinal fossa. The posterior paravesical space is a larger recess situated between the urinary bladder and rectum. In women, uterus further divides it into smaller vesicouterine and larger rectouterine spaces. The vesicouterine space is located between the urinary bladder and uterus and is formed as the peritoneum drapes the uterine body and fundus and then runs over the urinary bladder (along its posterosuperior surface). It is continuous with the supravesical space anteriorly and its depth varies with the degree of urinary bladder distension. The rectouterine pouch (of Douglas) is delimited anteriorly by the uterus and broad ligaments and posteriorly by the rectum. Lateral to this cul-de-sac on either side are the uterosacral ligaments that run from the sacrum to the uterine cervix that separate the rectouterine pouch from the laterally located ovarian fossae. In men, the rectovesical pouch is formed as the peritoneum runs from the rectum to the posterior surface of the bladder. Posteriorly, the peritoneal reflection from the upper rectum runs to the pelvic sidewalls, forming right and left pararectal fossae, which vary in size with the degree of rectal distension. Analogous to the uterosacral ligaments in women, the rectovesical space is restricted by sacrogenital folds on either side, which extend from the urinary bladder to the sacrum. The rectovesical space and rectouterine space (pouch of Douglas) form most dependent areas in both in supine and standing positions in males and females, respectively (both in supine and standing positions). The key aspects of pelvic peritoneal spaces are summarized in Flowchart 8.6.3, Figs. 8.6.15 and 8.6.16. Peritoneal recesses represent small pockets or fossae in the peritoneal cavity bounded by folds of peritoneum and possibly represent developmental aberrations. Knowledge of these recesses is important as they can serve as pathways for internal hernias. Multiple peritoneal folds may surround fourth part of duodenum (D4) and duodenojejunal junction forming duodenal recess. Six major duodenal recesses include superior, inferior, paraduodenal, retroduodenal, duodenojejunal and mesentericoparietal recess. The superior duodenal recess is situated to the upper left side of fourth part of duodenum (D4) behind the superior duodenal fold at L2 level. Inferior mesenteric vein is situated behind lateral end of superior duodenal fold. Inferior duodenal recess is situated on left of D4 below DJ flexure and behind the inferior duodenal fold at L3 level. Inferior duodenal recess often exists in association with superior duodenal recess. Reported incidence of superior and inferior duodenal recesses is about 40%–50% and 70%–75%, respectively. Paraduodenal recess is seen on left side and slightly behind D4 bounded anteriorly by paraduodenal (falciform fold). Right free margin of paraduodenal fold contains the inferior mesenteric vein and an ascending branch of the left colic artery. This recess is seen in about 2%–3.8% cases and is site for left paraduodenal hernia. Retroduodenal recess as the name suggests situated behind the third and fourth parts of the duodenum and in front of abdominal aorta. Its orifice is bounded anteriorly by duodenoparietal fold. Retroduodenal space is largest among all the duodenal recesses and is seen in about 2.5% cases. Duodenojejunal recess is situated between the duodenojejunal flexure and root of transverse mesocolon, hence also known as mesocolic recess. Its boundaries include pancreas (superior), left renal vein (inferior) and left kidney (left). Its opening lies between two peritoneal folds, is directed inferiorly and to the right. Duodenojejunal recess is seen in about 20% cases and is almost never associated with other duodenal recesses. Mesentericoparietal recess is situated below third part of duodenum and behind the upper part of the mesentery. Its opening is bounded anteriorly by the fold of mesentery raised by the superior mesenteric artery. This recess is most rare among all with incidence of about 1%. It is the site for right paraduodenal hernia. The key aspects of duodenal recesses are summarized in Table 8.6.6 and Fig. 8.6.17. Peritoneal folds adjoining cecum result in formation of four main recesses: superior ileocaecal recess, inferior ileocaecal recess, retrocaecal recess and paracolic recess (Fig. 8.6.18). Surgically these recesses are important as they can be sites for internal herniation (except for paracolic sulci) or site of abscess formation in complicated appendicitis. Superior ileocaecal recess is bounded in front by the vascular fold of the cecum (containing anterior caecal vessels), behind by the ileal mesentery, below by the terminal ileum and on the right by the ileocaecal junction with its opening on left. Inferior ileocaecal recess is bounded in front by the ileocaecal fold (bloodless fold of Treves), above by the terminal ileum and its mesentery, to the right by the caecum, and behind by the upper part of the mesoappendix with its opening on left. Retrocaecal recess is bounded in front by posterior wall of cecum (and sometimes the proximal ascending colon), behind by the parietal peritoneum (posterior abdominal wall), and on either side by two caecal folds (parietocolic folds). Retrocaecal recess is largest among all the caecal recesses with its opening inferiorly and often contains appendix. Paracolic sulci represent variable one or more shallow lateral depressions in the peritoneum investing the cecum usually within the right paracolic gutter. This recess lies behind and inferior to the apex of inverted V-shaped root of sigmoid mesocolon with orifice opening inferiorly. Posteriorly, it is limited by parietal peritoneum of covering the left ureter at its iliac vessel crossing. This recess can be variable in size ranging from dimple to a small fossa in majority of the cases usually admitting little finger. It forms most common site of sigmoid mesocolon hernia. It can also be involved by peritoneal fluid and metastatic disease. Peritoneal and subperitoneal spaces are mutually exclusive spaces having distinct patterns of disease spread. Different pathways of the intraabdominal and extraabdominal disease spread are summarized in Table 8.6.7.
8.6: Peritoneum
Embryology of peritoneum
Imaging techniques
Modality
Sensitivity
Specificity
Diagnostic Odds Ratio
CT
0.68 (0.46–0.84)
0.88 (0.81–0.93)
15.9 (4.38–58.01)
MRI
0.79 (0.092)
0.90 (0.80–0.96)
36.5 (6.7–200.0)
PET/CT
0.91 (0.96)
0.85 (0.78–0.91)
63.3 (31.5–127.3)
Data in parentheses are 95% confidence intervals.
Conventional imaging
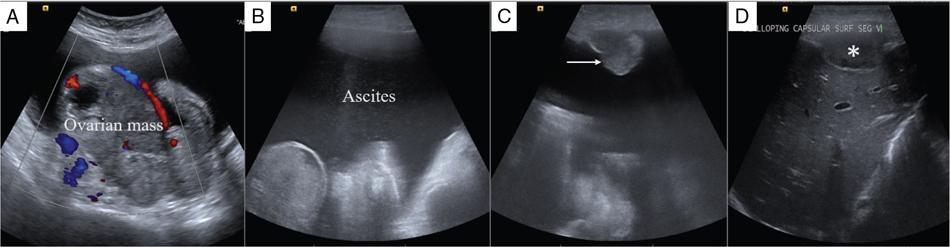
Ultrasonography
Computed tomography (CT)
Type of scanner
Section thickness
Interval
Scan area
Enteric contrast
Intravenous contrast
Scan acquisition timing
Modifications
Image reconstruction and displays
Limitations of CT in the assessment of peritoneal disease
Dual-energy CT (DECT)
MRI
3 Plane localiser
Coronal SSFESE
Axial SSFESE
Axial SSFESE FS
3 D Axial Dual Echo
Axial DWI [b 50,500,1000]
3 D Axial LAVA pre-contrast
3 D Axial LAVA post-contrast
3 D Coronal LAVA post-contrast
3 D Sagittal LAVA post-contrast
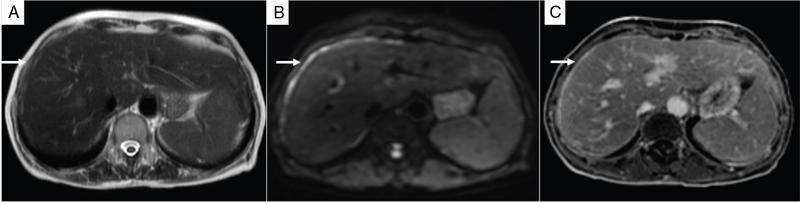
Diffusion-weighted MRI imaging
Contrast-enhanced MRI (CE MRI)
Limitations of MRI
PET/CT
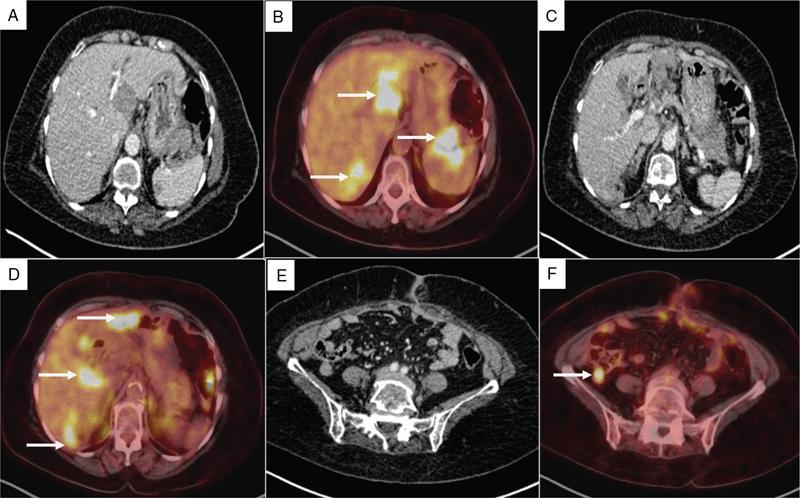
Limitations of PET/CT
Modality
Indications
Advantages
Disadvantages
USG
First-line imaging modality in acute abdomen, assessment for ascites, preferred modality for interventions
Portable, real time, widely available and cost effective
Insensitive to smaller lesions. Underestimates the disease burden
CT
First-line imaging modality of choice for assessment of malignancies of chest, abdomen and thorax
Widespread availability, faster acquisition
Limited soft tissue resolution limiting assessment of <1 cm size nodules and difficult resection areas such as mesentery, bowel wall, subphrenic space
MRI
Alternative to CT, assessment of difficult resection areas, allergy to iodinated contrast
Higher spatial resolution allowing better detection of smaller lesions
Longer acquisition time, contraindications of MRI
PET/CT
Pretreatment staging, Postoperative surveillance
Combines benefits of CT and PET
Higher cost and high radiation doses, limited availability
Anatomy of peritoneum
Peritoneal ligaments, omentum and mesentery
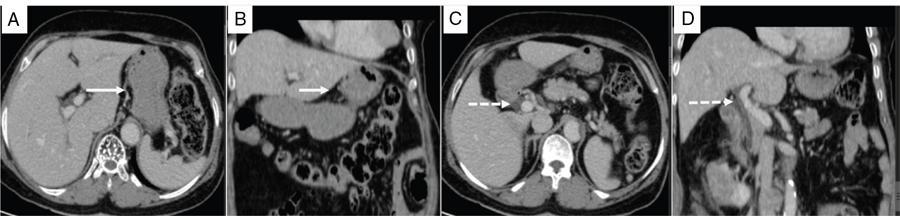
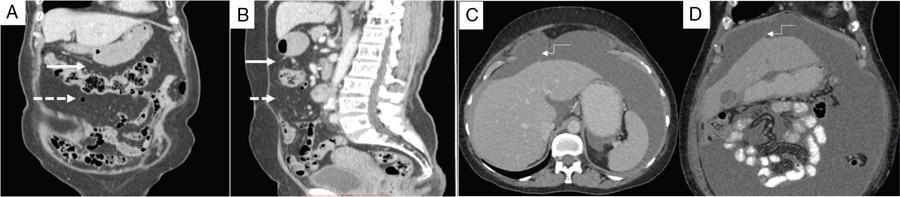
Peritoneal Ligaments
Connected Structures
Key Vasculature
SUPRAMESOCOLIC COMPARTMENT
Lesser curvature to fissure of ligamentum venosum
Hepatic hilum to upper margin of first part of duodenum
Greater curvature and inferior margin of duodenal bulb to lower border of pancreatic capsule and transverse colon
Greater curvature to diaphragm
Anterosuperior aspect of liver to supraumbilical anterior abdominal wall
Lies in free edge of falciform ligament
Left portal vein to IVC (near the entry of left hepatic vein)
Right lateral hepatic surface to diaphragm
Left hepatic lobe to diaphragm
Splenic hilum to left anterior pararenal fascia
Greater curvature (near the fundus) to splenic hilum
Splenic flexure to diaphragm
INFRAMESOCOLIC COMPARTMENT
Duodenojejunal flexure to ileocaecal junction.
Ascending colon to mesenteric root
Pancreatic capsule to transverse colon
Descending colon to left superior duodenal fold
Sigmoid colon to origin of inferior mesenteric artery.
Subperitoneal space
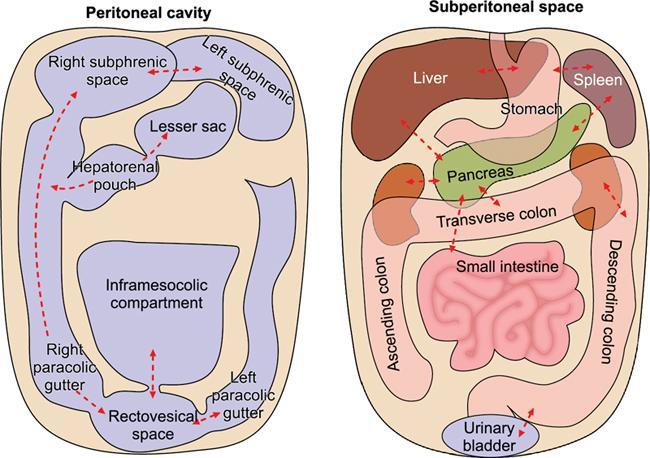
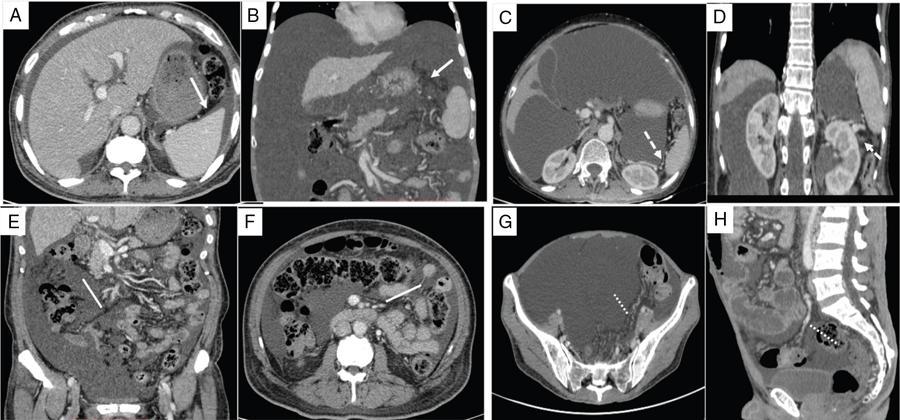
Peritoneal spaces
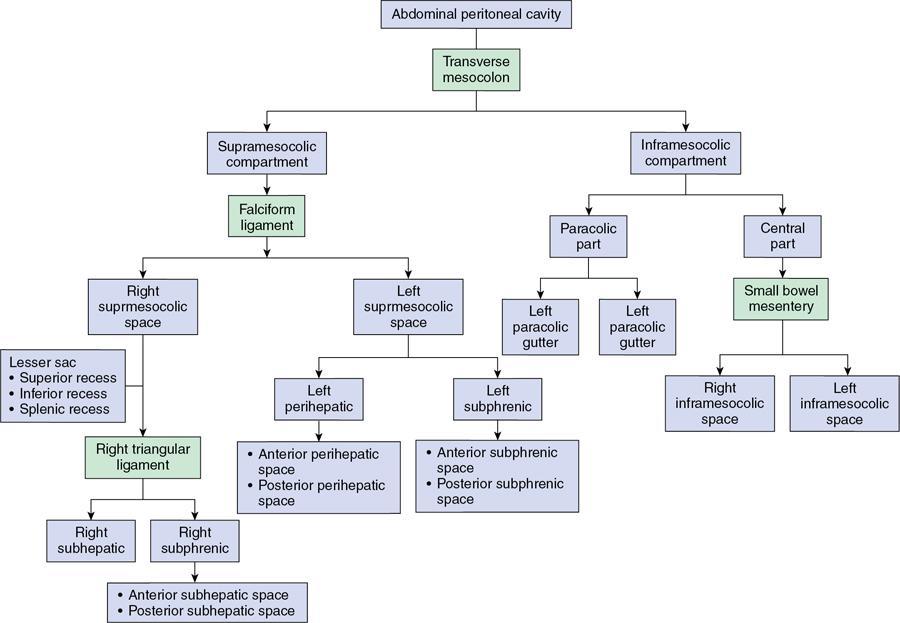
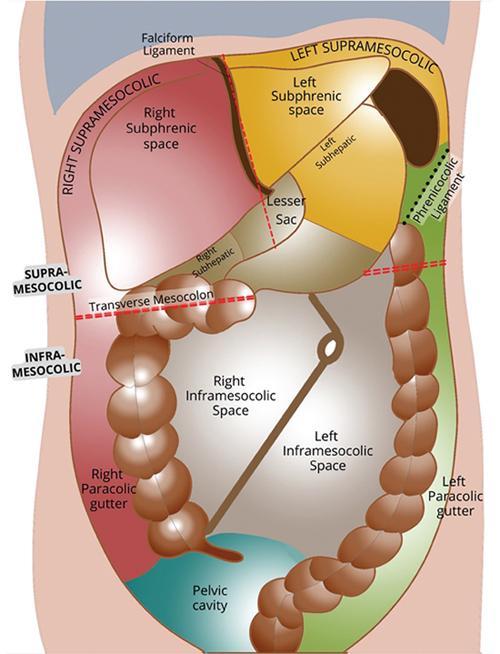
Supramesocolic compartment
Inframesocolic compartment
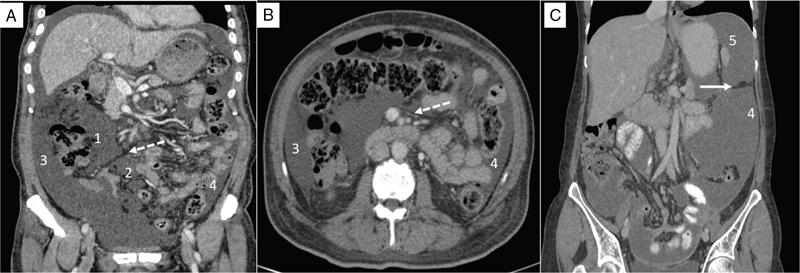
Pelvic peritoneal spaces.
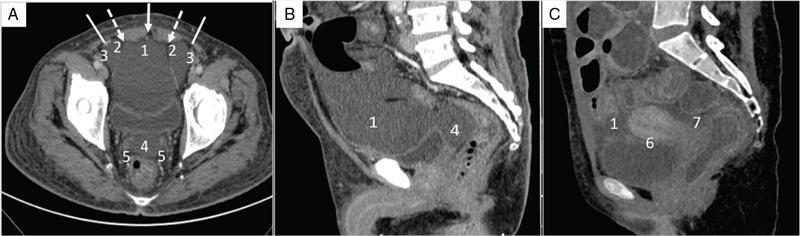
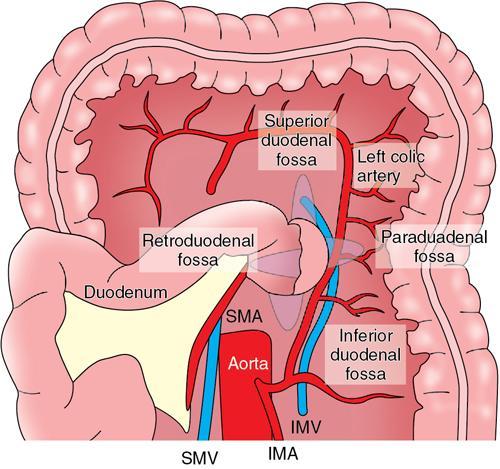
Peritoneal recesses
Duodenal recesses.
Duodenal Recess
Location
Associated Peritoneal Fold
Incidence
Additional Features
Superior duodenal recess
On upper left side of fourth part of duodenum (D4)
Superior duodenal fold. IMV is situated behind its lateral end.
40%–50%
Superior and inferior recesses often coexist
Inferior duodenal recess
To left of D4 below DJ flexure
Inferior duodenal fold (avascular)
70%–75%
Most common recess
Paraduodenal recess
On left side and slightly behind D4
Paraduodenal fold containing IMV and ascending branch of left colic artery
2%–3.8%
Site for left paraduodenal hernia
Retroduodenal recess
Behind D3 and D4 and in front of abdominal aorta
Duodenoparietal fold
2.5%
Largest among all
Duodenojejunal (mesocolic) recess
Between DJ flexure and root of transverse mesocolon
Orifice between two peritoneal folds
20%
Almost never associated with other duodenal recesses
Mesentericoparietal recess
Below third part of duodenum and behind upper part of mesentery
Fold of mesentery raised by SMA
1%
Rarest, site for right paraduodenal hernia
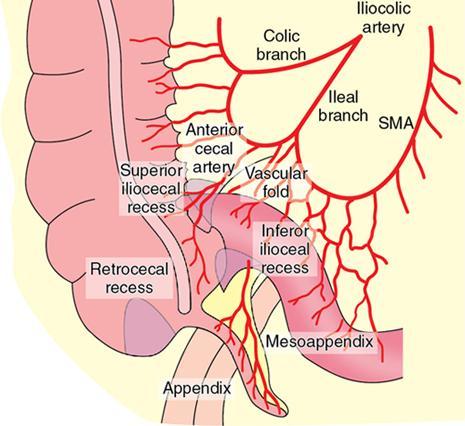
Caecal recess.
Intersigmoid recess.
Implications of peritoneal anatomy on the pattern of disease spread
Peritoneum
MRI Imaging Protocol for Peritoneal Pathology in our Institution *